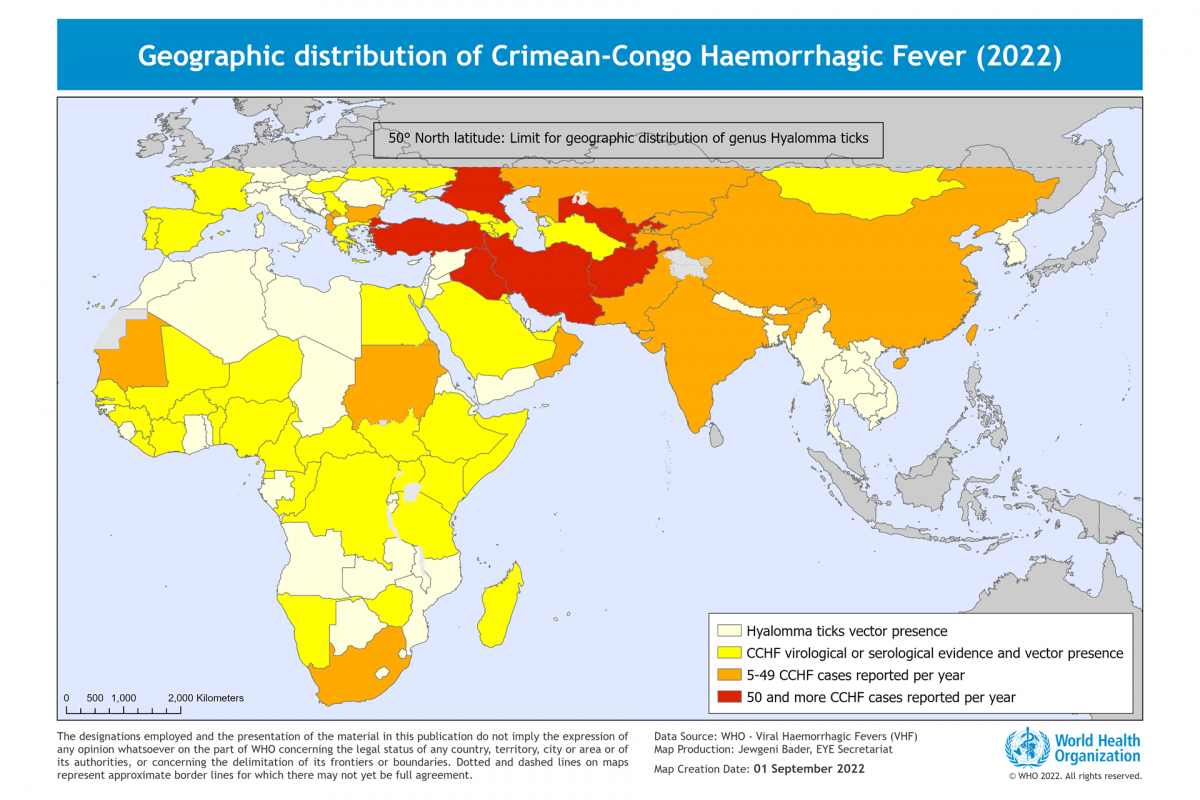
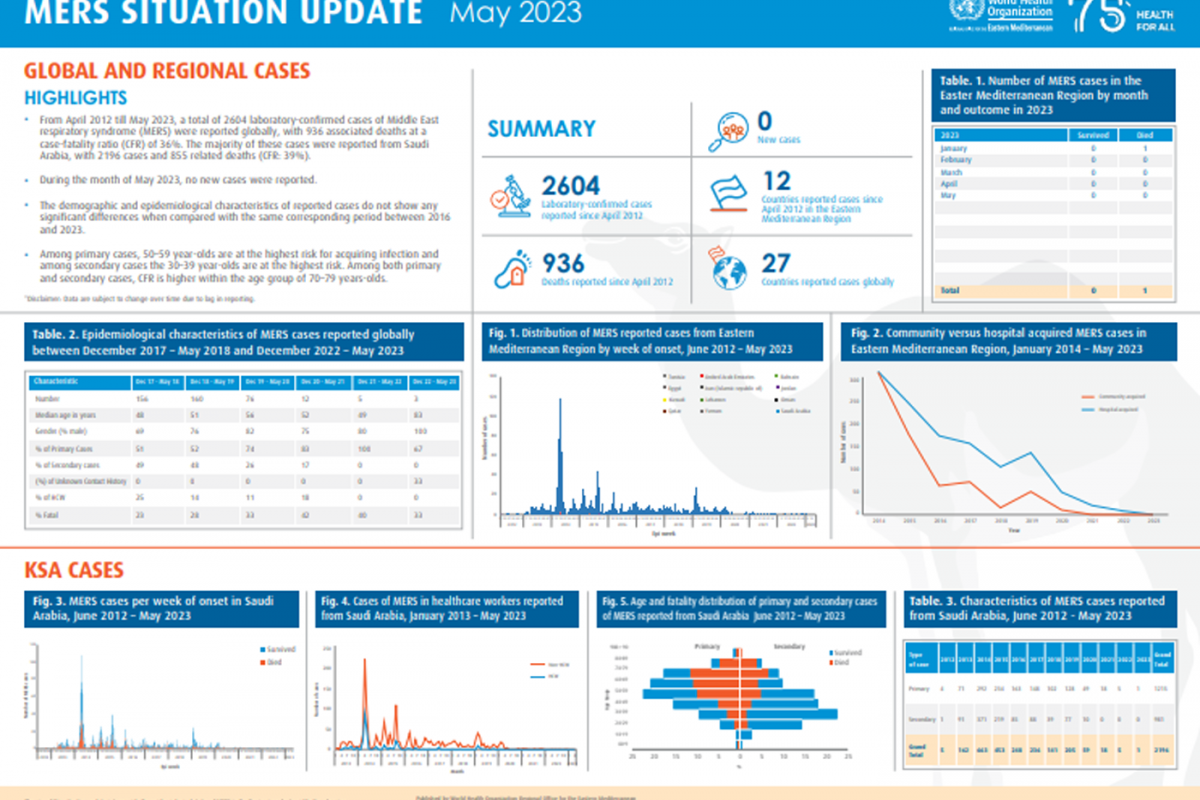
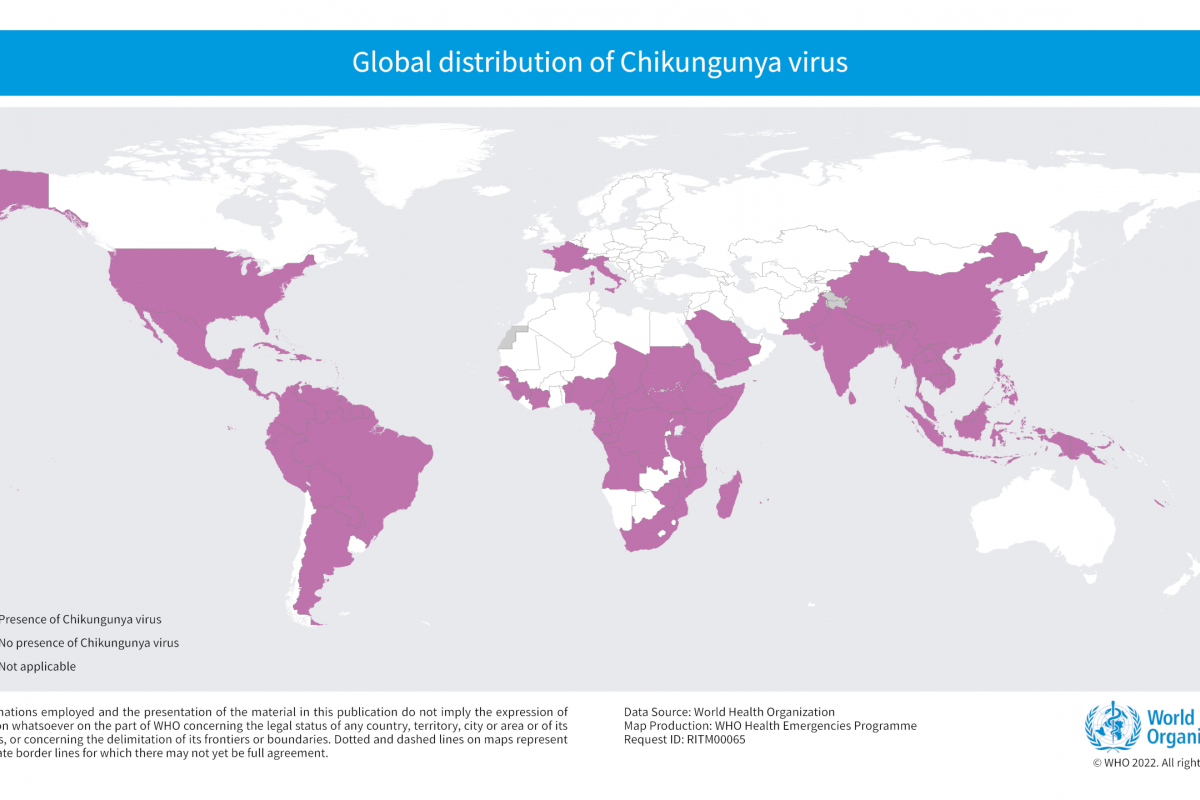
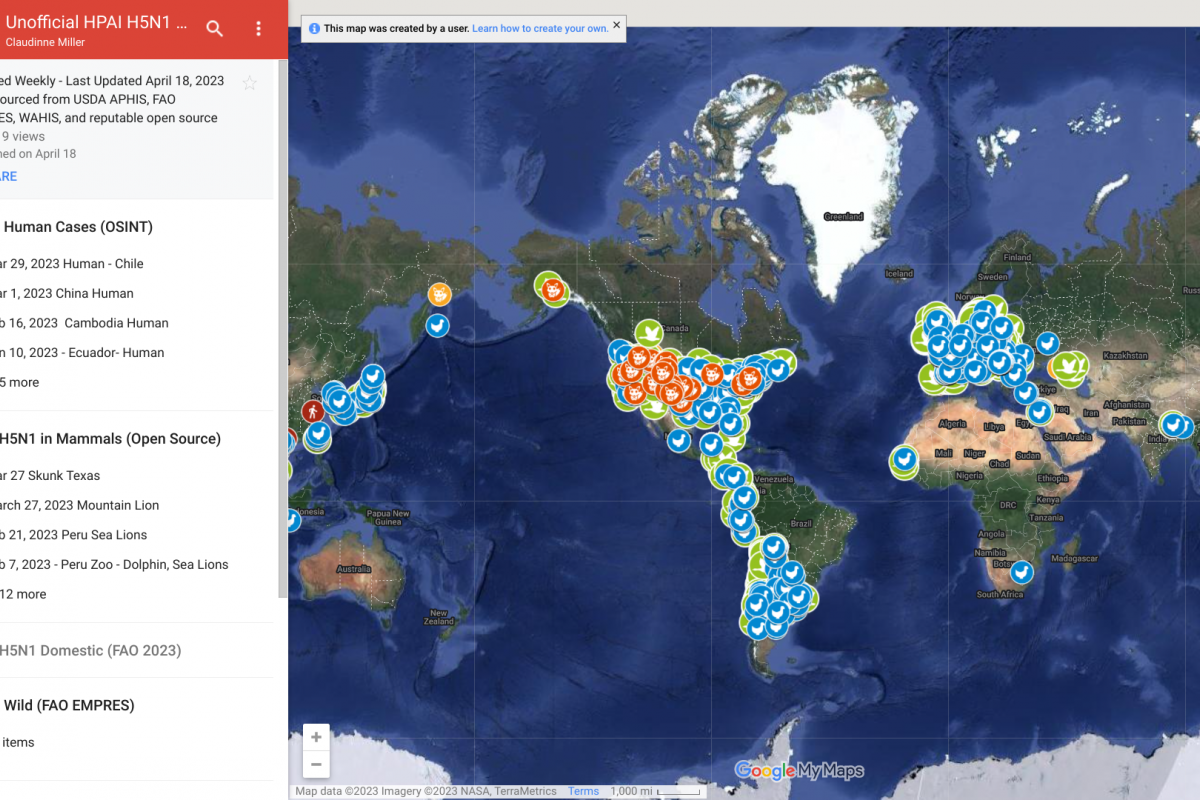
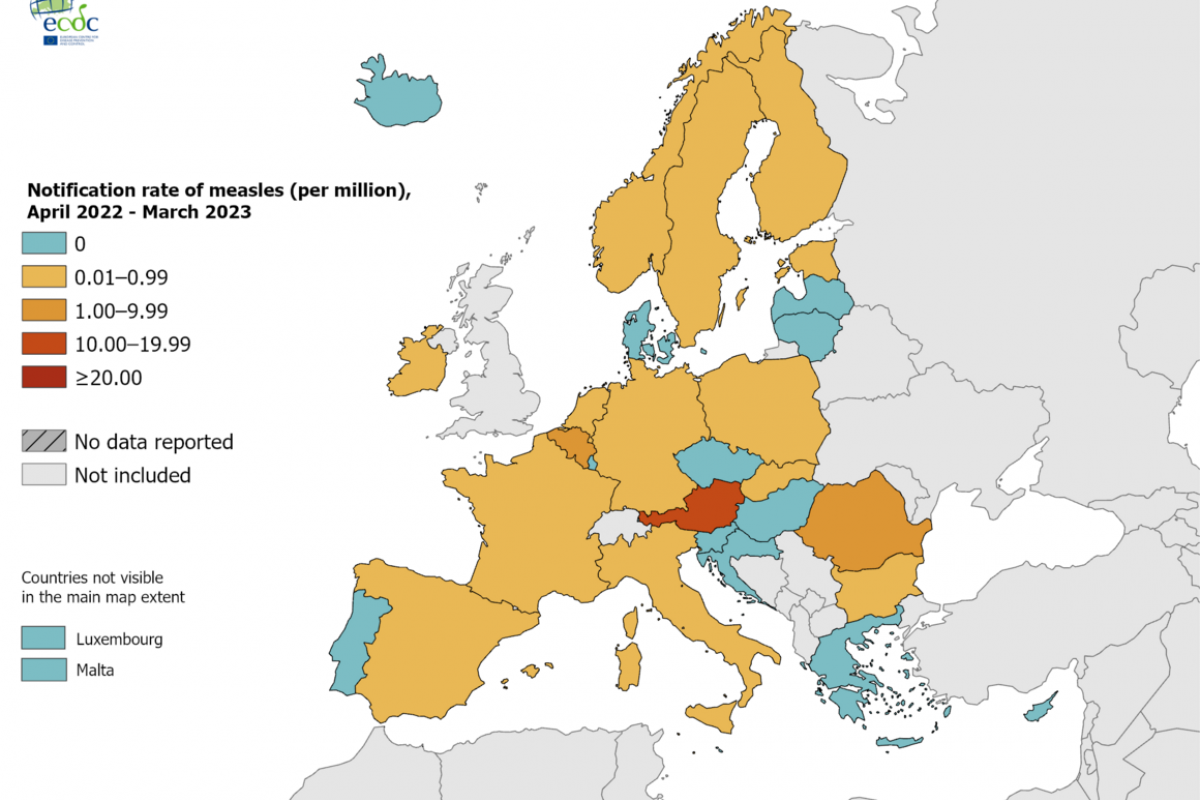
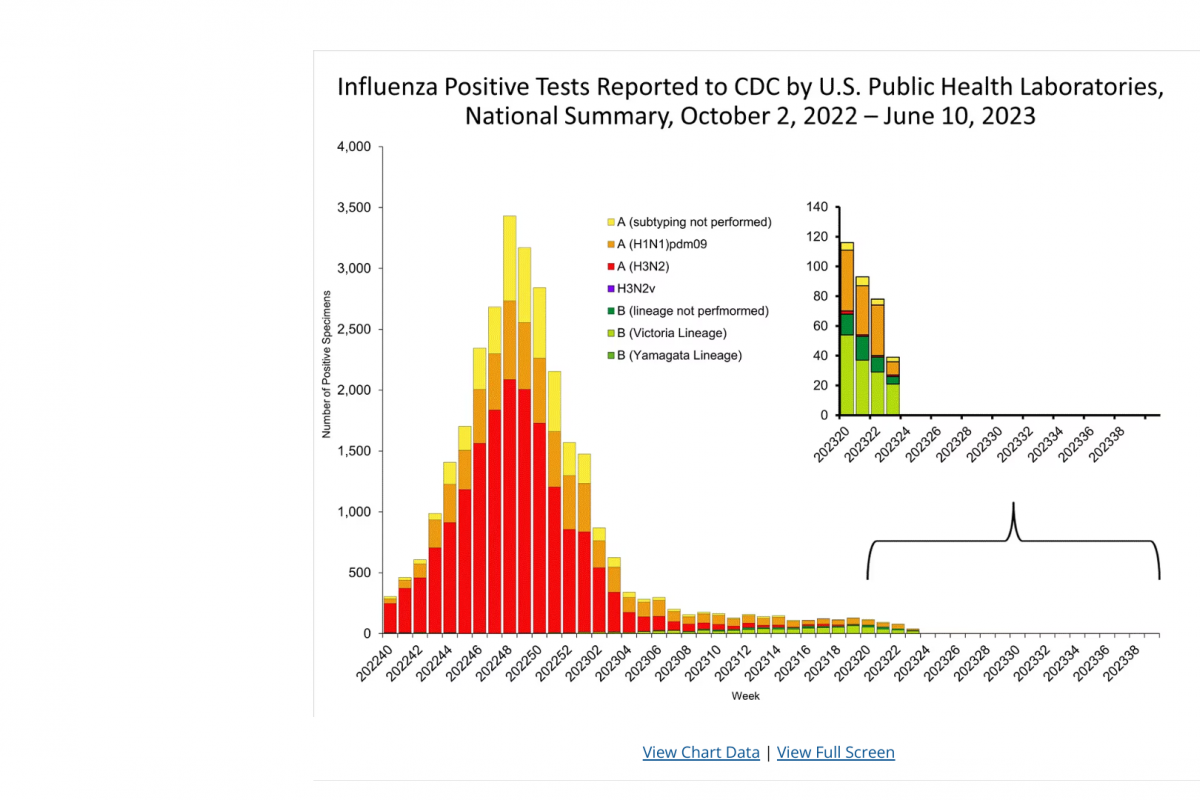
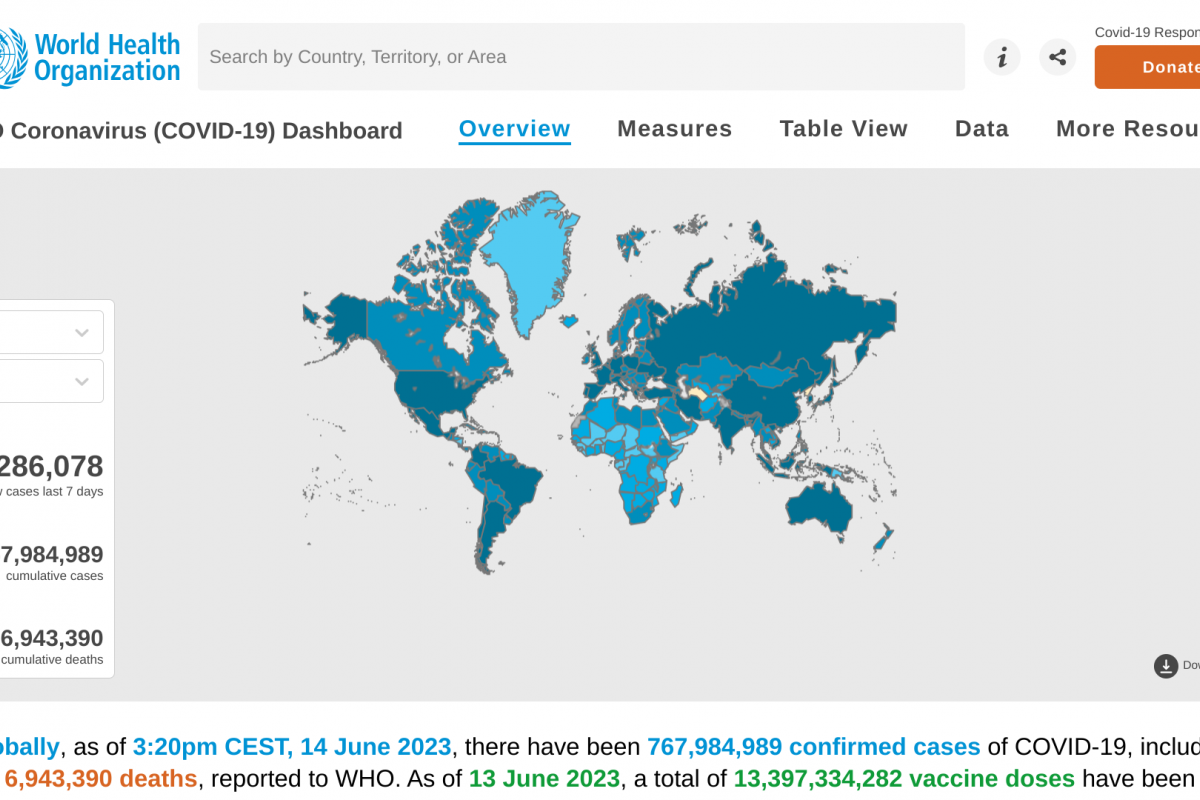
Brazil recently notified the World Health Organization (WHO) of a fatal laboratory-confirmed human case of infection with a swine-origin influenza A(H1N1) variant (v) virus in the state of Paraná.
The patient was a 42-year-old woman with underlying medical conditions who lived near a swine (pig) farm.
She developed a fever, headache, sore throat, and abdominal pain on May 1, 2023, and was hospitalized with a severe acute respiratory infection.
On May 4, the patient was admitted to the Intensive Care Unit, and she passed away on May 5, 2023.
Ongoing investigations reported that the patient did not have any direct contact with pigs, however, two of her close contacts worked at the swine farm.
The two contacts did not develop respiratory disease and tested negative for influenza.
To date, no human-to-human transmission associated with this case has been identified in Brazil.
This is the first human infection caused by an influenza A(H1N1)v virus reported in 2023 in Brazil and the third human infection reported in the state of Parana (2021 and 2022).
WHO assesses the risk of international disease spread through humans and/or community-level spread among humans posed by this event as low. The risk level will be amended if warranted by the investigations currently being conducted by national authorities.
As of June 20, 2023, there is no swine vaccine for Influenza A(H1N1)v infection currently licensed for use in humans.
And seasonal flu shots are generally not expected to protect people from influenza viruses that normally circulate in pigs.