RSV Monoclonal Antibody Therapy Found Very Effective

The peer-review JAMA Network published an Original Investigation focused on the most appropriate monoclonal antibody (mAb) for preventing respiratory syncytial virus (RSV) in children.
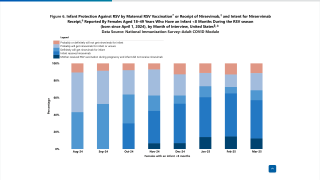
This analysis was essential since RSV is a leading cause of respiratory disease in children and the primary cause of hospitalization for viral respiratory infections.
Published on February 17, 2023, this systematic review and network meta-analysis comparing the efficacy and safety of 4 mAbs for preventing RSV infection in children found associations with the improvement of clinically meaningful outcomes and no significant associations with RSV-related adverse effects and mortality.
These findings suggest that motavizumab, nirsevimab, and palivizumab are associated with reduced rates of RSV infections and hospitalizations.
Similar results were observed in the rate of supplemental oxygen use.
While RSV vaccine candidates are completing late-stage clinical studies, mAbs targeting RSV prevention have been U.S. Food and Drug Administration (FDA) for years.
For example, the U.S. CDC CDCHAN-00479 stated on November 4, 2022, that eligible high-risk children should receive the mAbs palivizumab (Synagis) treatment according to AAP guidelines to prevent RSV-associated hospitalizations.
Recently, the Biologics License Application for nirsevimab (Beyfortus®) was accepted for review by the FDA on January 5, 2023. The FDA's Prescription Drug User Fee Act date for its decision is in the third quarter of 2023.
If approved, nirsevimab would become available in the U.S. for the 2023/2024 RSV season.
Nirsevimab (MEDI8897) was developed in partnership between AstraZeneca and Sanofi.
Our Trust Standards: Medical Advisory Committee