The U.S. Department of State recently announced revised travel advisories for two counties located in Central America.
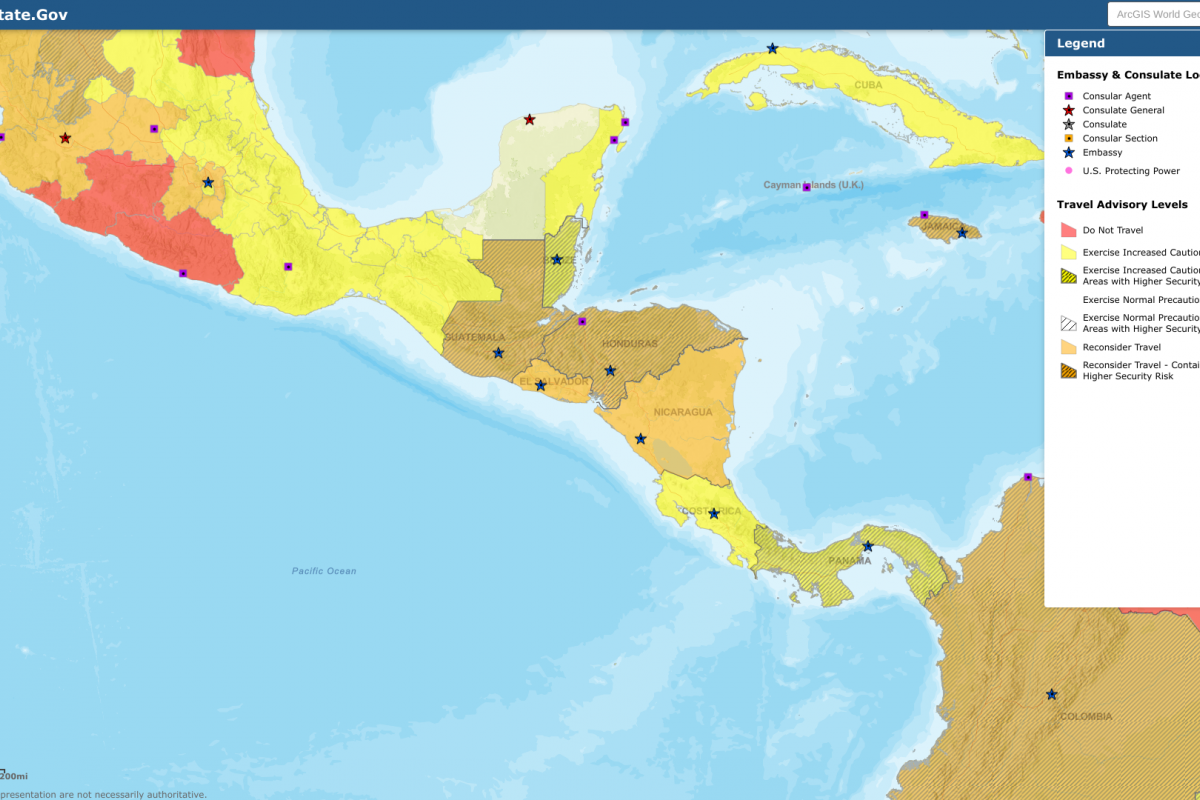
On March 1, 2023, the State Department reissued its Level 3: Reconsider Travel advisory for the Republic of Honduras.
This high-level advisory highlights civil unrest in areas such as the Gracias a Dios Department.
Separately, the State Department reissued its Level 3: Reconsider Travel to the Republic of Guatemala.
This Gualemalia alert identifies says do not travel to:
- San Marcos Department (except the city of San Marcos).
- Huehuetenango Department (except the city of Huehuetenango).
- Zone 18 and the city of Villa Nueva in Guatemala City.
Within both advisories, travelers to these countries are encouraged to enroll in the State Department's Smart Traveler Enrollment Program to receive digital alerts in an emergency.
Previously, in support of the region's development, the United States Agency for International Development announced on February 15, 2023, it had awarded $42.5 million in new humanitarian funding for the people of El Salvador, Guatemala, and Honduras.
This brings USAID's total humanitarian funding for these three countries to nearly $261 million since April 2021.
Additional travel advisories have been recently issued for Costa Rica and El Salvador.
From a health perspective, the U.S. Centers for Disease Control and Prevention suggests various travel vaccinations before visiting Central America.
These vaccines include yellow fever, measles, and influenza.
Travel vaccination services are offered at certified health clinics and pharmacies in the U.S.
Updated with links on March 6, 2023.