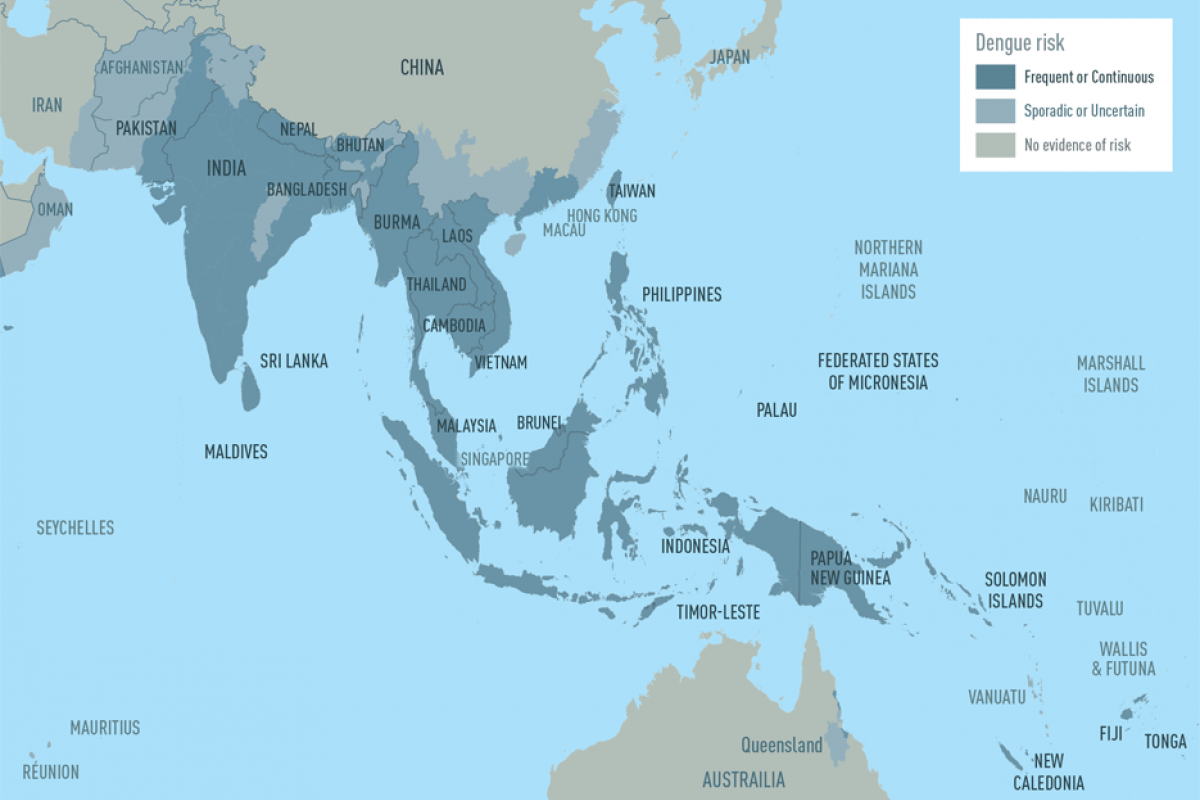
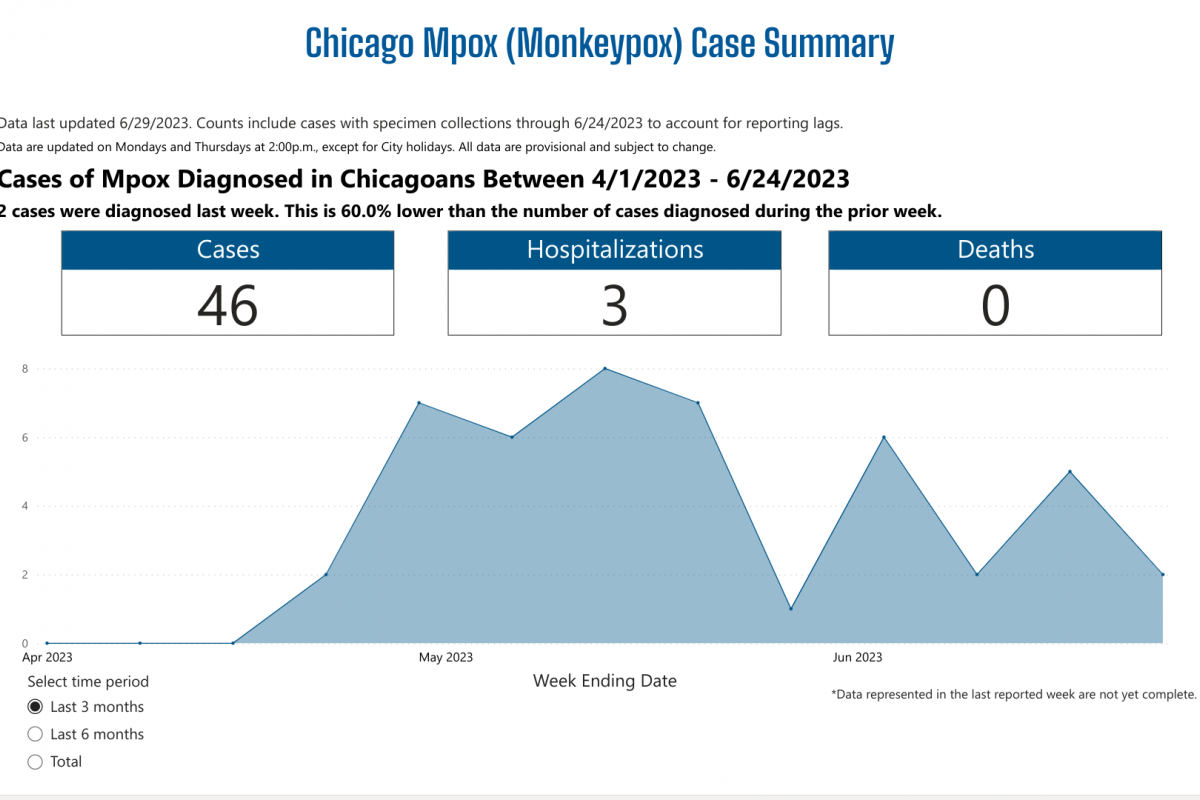
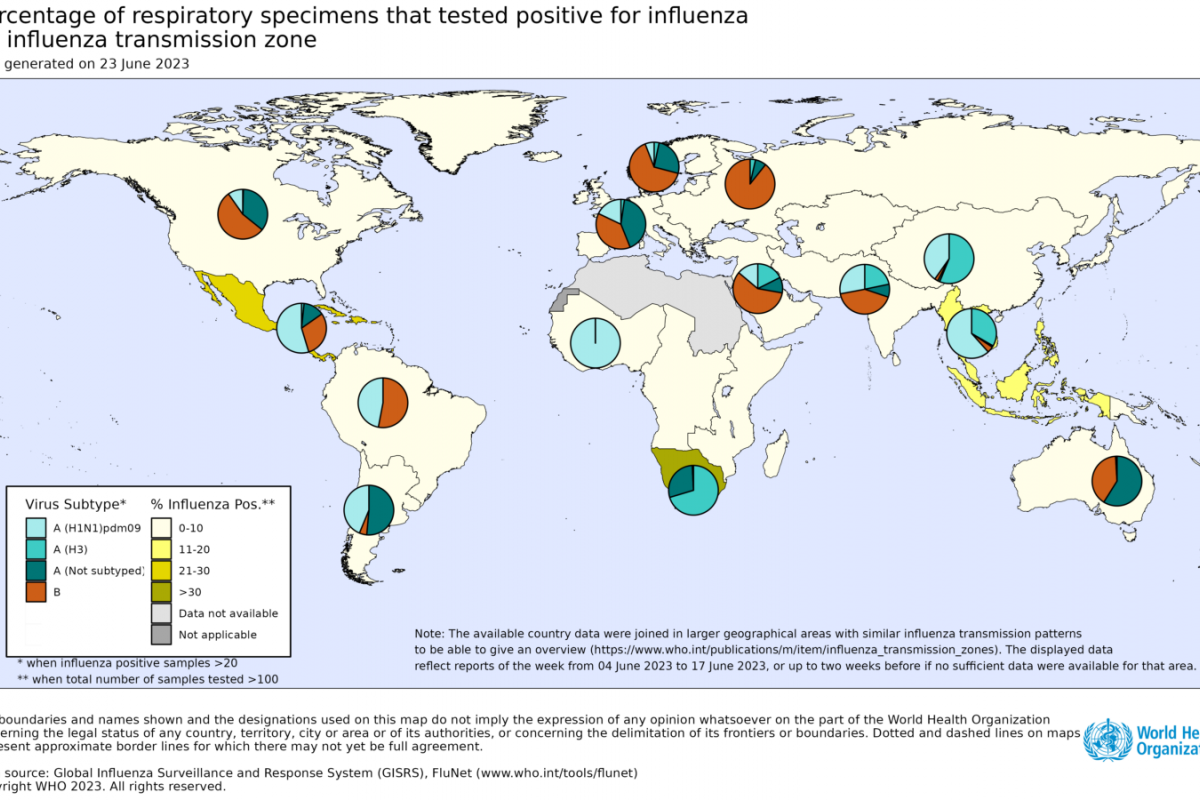
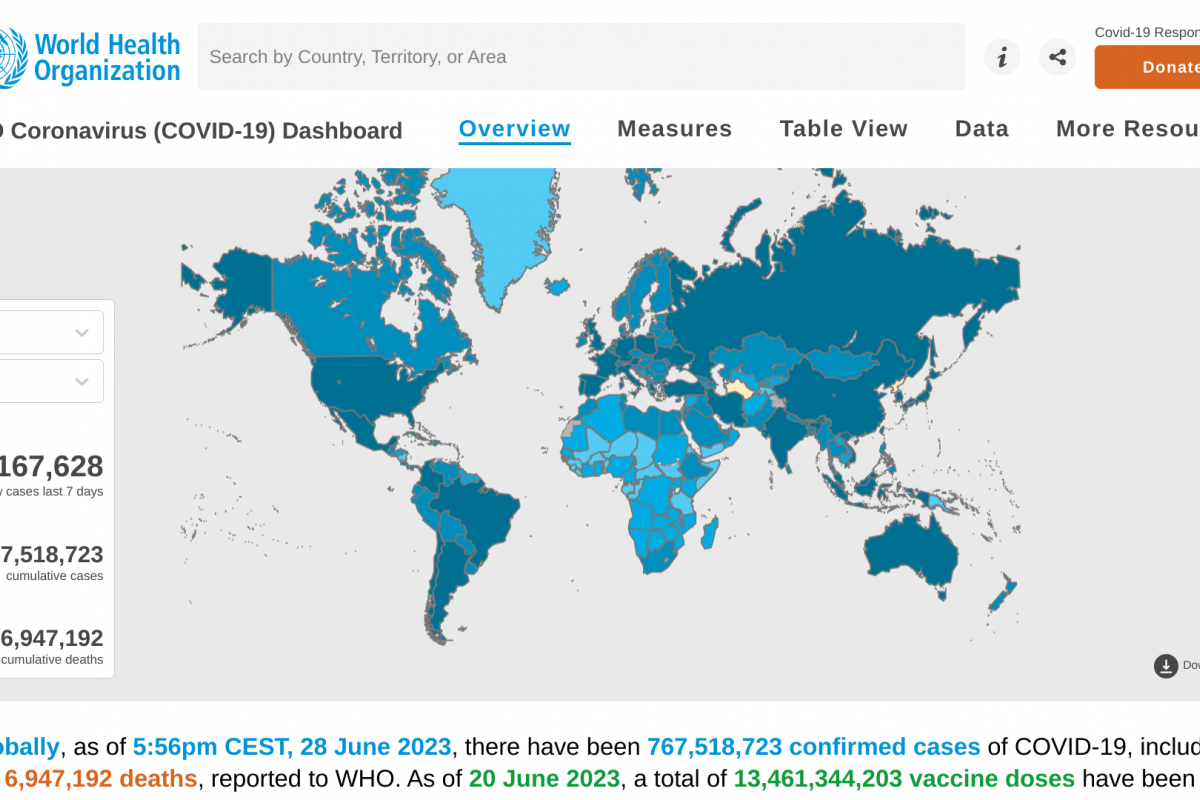
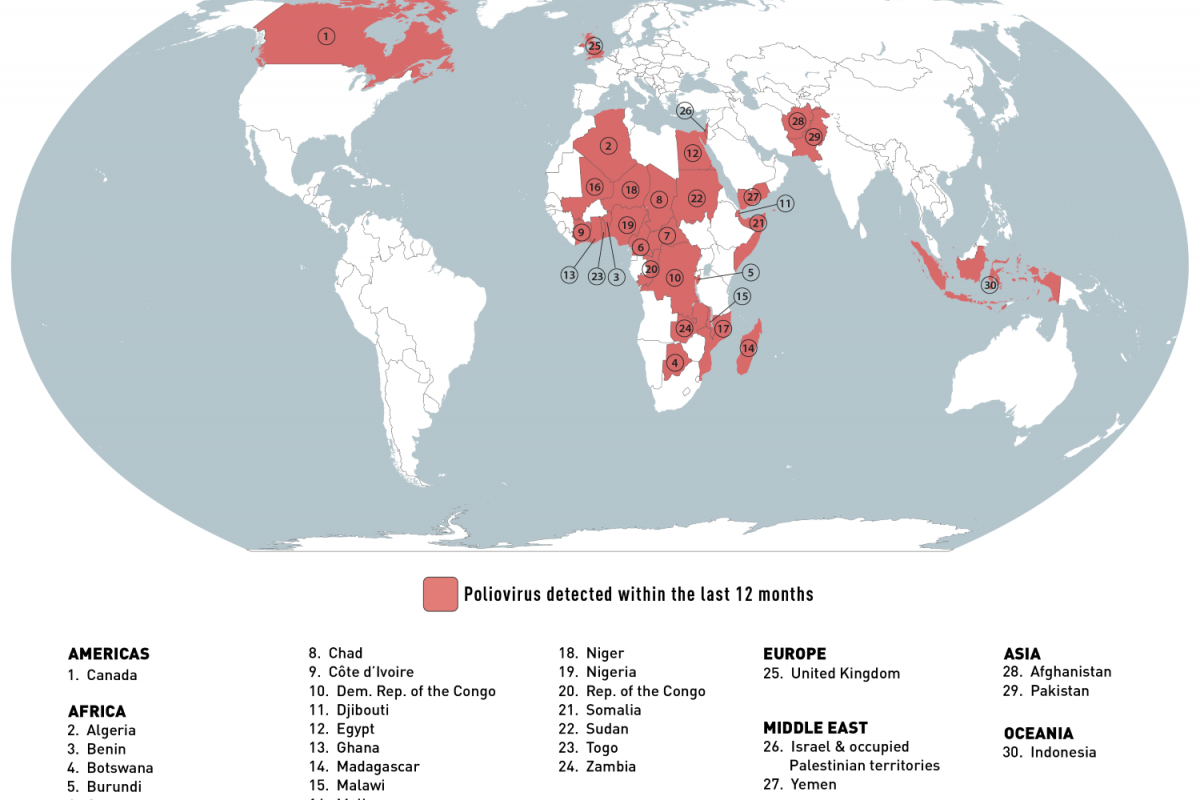
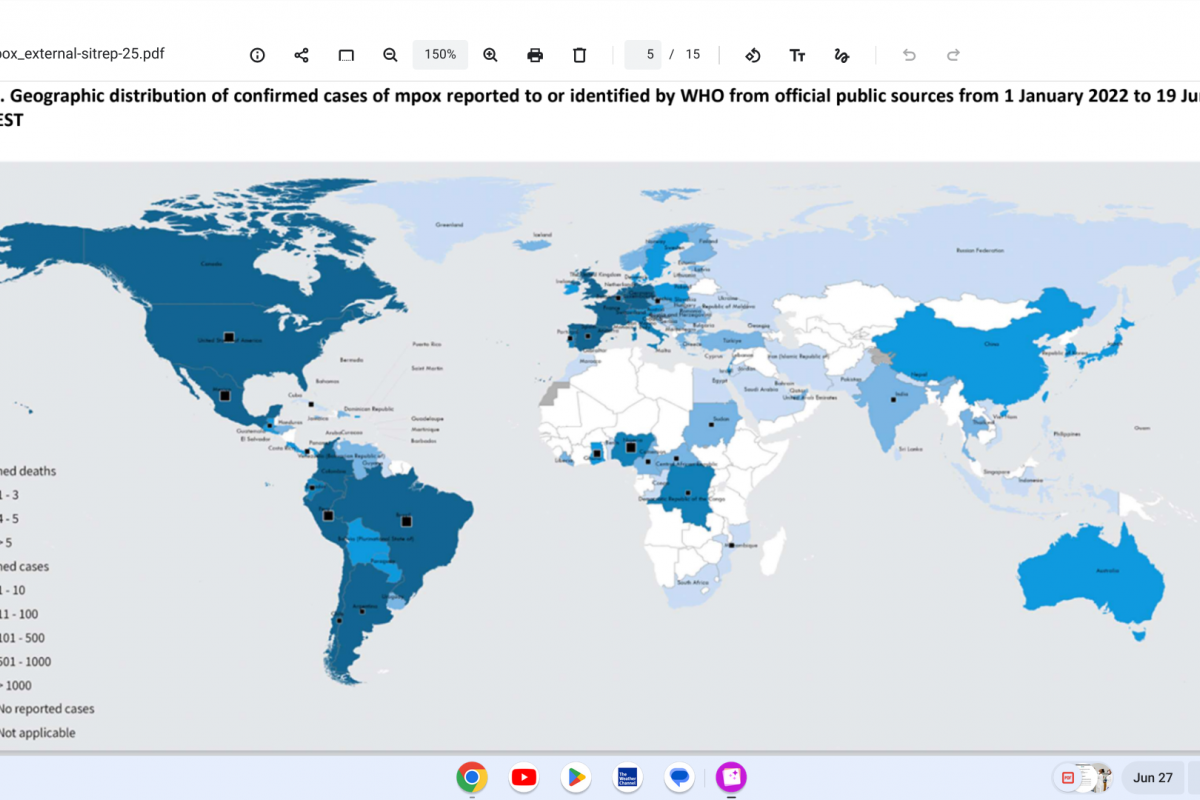
As the new flu season approaches this fall, influenza viruses from last season continue negatively impacting people.
On June 30, 2023, the U.S. Centers for Disease Control and Prevention (CDC) published the Weekly U.S. Influenza Surveillance Report confirmed respiratory illness, often called influenza-like illness, severely impacts people.
According to National Center for Health Statistics Mortality Surveillance data on June 29, 2023, 6.2% of the deaths during week #25 were due to pneumonia, influenza, and/or COVID-19 (PIC).
Among the 1,342 PIC deaths reported for this week, 240 had COVID-19 listed as an underlying or contributing cause of death on the death certificate, and eight listed as influenza.
The majority of deaths were related to pneumonia.
Furthermore, one additional influenza-associated pediatric death occurring during the 2022-2023 season was reported to CDC during week 25.
Throughout the 2022-2023 flu season, the total number of pediatric deaths totaled 160.
In the 2019-2020 flu season, 199 children died from influenza infections.
With the new flu season starting soon, the CDC recently announced good news regarding vaccines.
The U.S. CDC adopted the 2023-2024 recommendations on annual influenza vaccination for everyone six months and older on June 27, 2023.
Additionally, flu vaccinations in July and August are not recommended for most people, but there are several considerations for specific groups.