During March 2024, many college students will enjoy the warm and friendly South Florida beaches.
However, various diseases also impact those Florida communities.
For week #10, Florida Health's Mosquito-Borne Disease Surveillance reported new dengue and malaria cases.
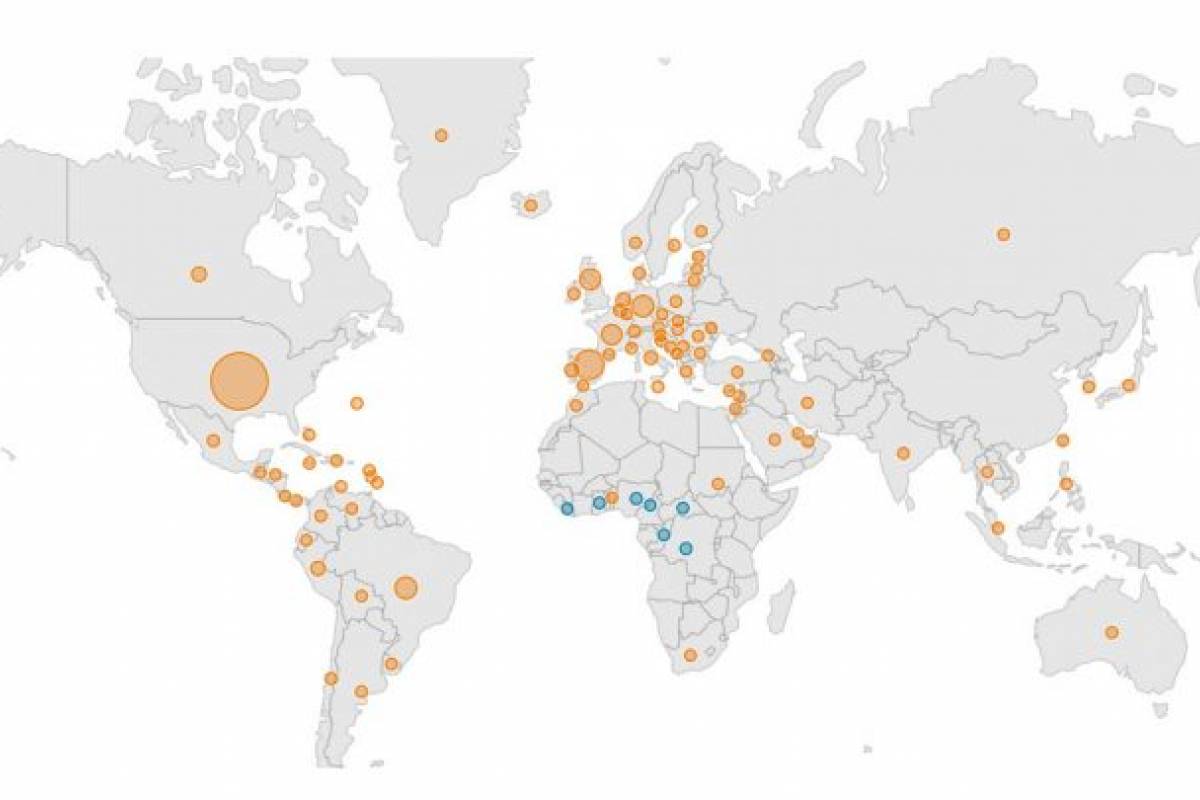
In 2024, 59 travel-associated dengue cases were reported, mainly by visitors from Cuba.
And four cases of locally acquired dengue have been reported from two counties this year. In 2023, positive samples from 186 humans were reported from five counties.
Also, in 2024, twelve cases of malaria have been reported in individuals with a travel history to a malaria-endemic area, such as Africa.
In 2023, seven locally acquired malaria cases were reported in the Sarasota area.
On the bright side, the agency also communicated that the recent measles outbreak in Broward County has been contained.
No additional measles cases have occurred at Manatee Bay Elementary since Friday, February 16, 2024; thus, the 21-day infection period has ended.
However, due to the contagiousness of measles and the rate of international travel, there is a possibility for more sporadic cases throughout Florida and the United States.
"It's important that public health leaders consider all available data and make decisions that promote health while respecting the rights of individuals," said Florida Surgeon General Dr. Joseph Ladapo in a press release on March 8, 2024.
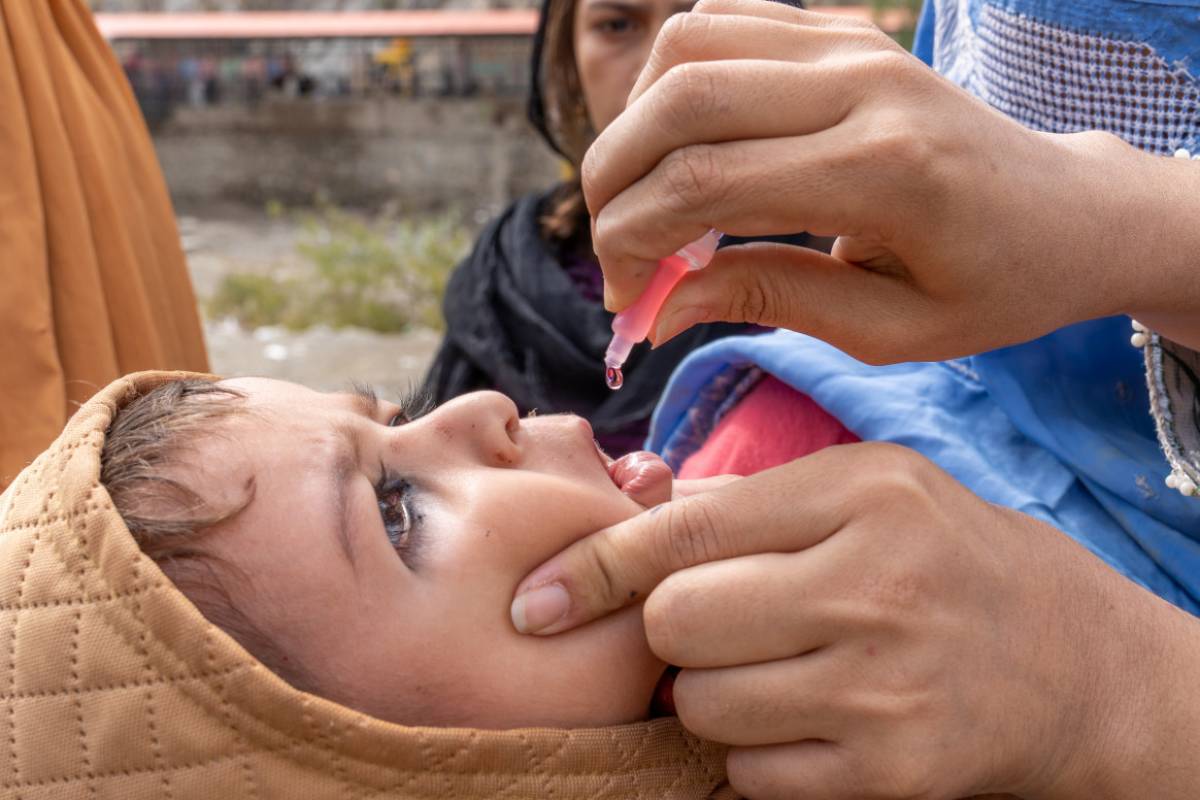
The State Surgeon General communicated that unprotected people should speak with their healthcare provider about receiving approved vaccines.
As of March 12, 2024, the state of Florida, the U.S. Centers for Disease Control and Prevention, Canada, and the United Kingdom have not issued any vaccination requirements for visitors to Florida.