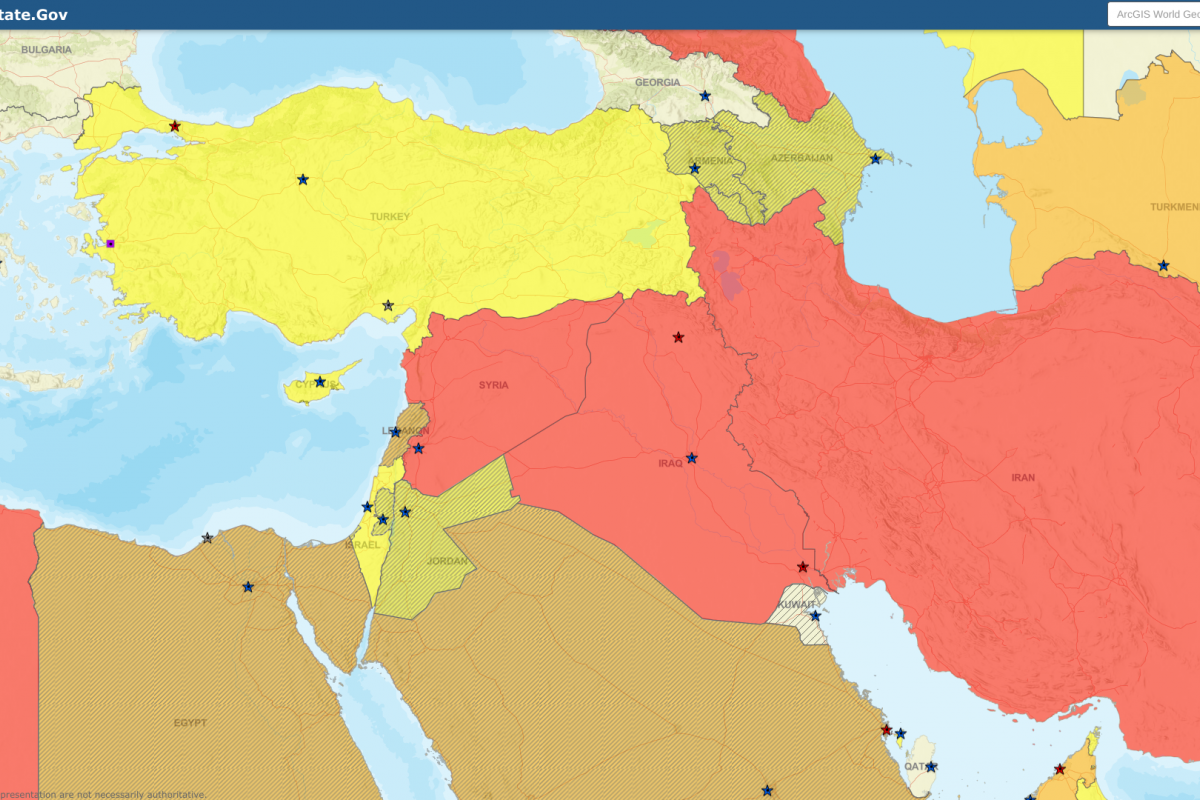
The U.S. Department of State reissued its Level 4: Do Not Travel advisory for the Republic of Iraq. As of January 3, 2023, this travel alert indicates significant civil unrest and Mission Iraq’s limited capacity to support U.S. citizens.
Furthermore, U.S. citizens in Iraq face high risks to their safety and security.
Due to risks to civil aviation operating in the Baghdad Flight Information Region, the U.S. Federal Aviation Administration (FAA) has extended its Special Federal Aviation Regulation for an additional two years, prohibiting certain flights at altitudes below 32,000 feet.
Because of security concerns, U.S. government personnel in Baghdad are instructed not to use Baghdad International Airport.
U.S. citizens should consult the FAA Prohibitions, Restrictions, and Notices website for more information.
For local assistance, the U.S. Embassy in Baghdad is located at Al-Kindi Street, International Zone, Baghdad, Iraq.
From a health perspective, the U.S. CDC publishes travel vaccine recommendations at this link.