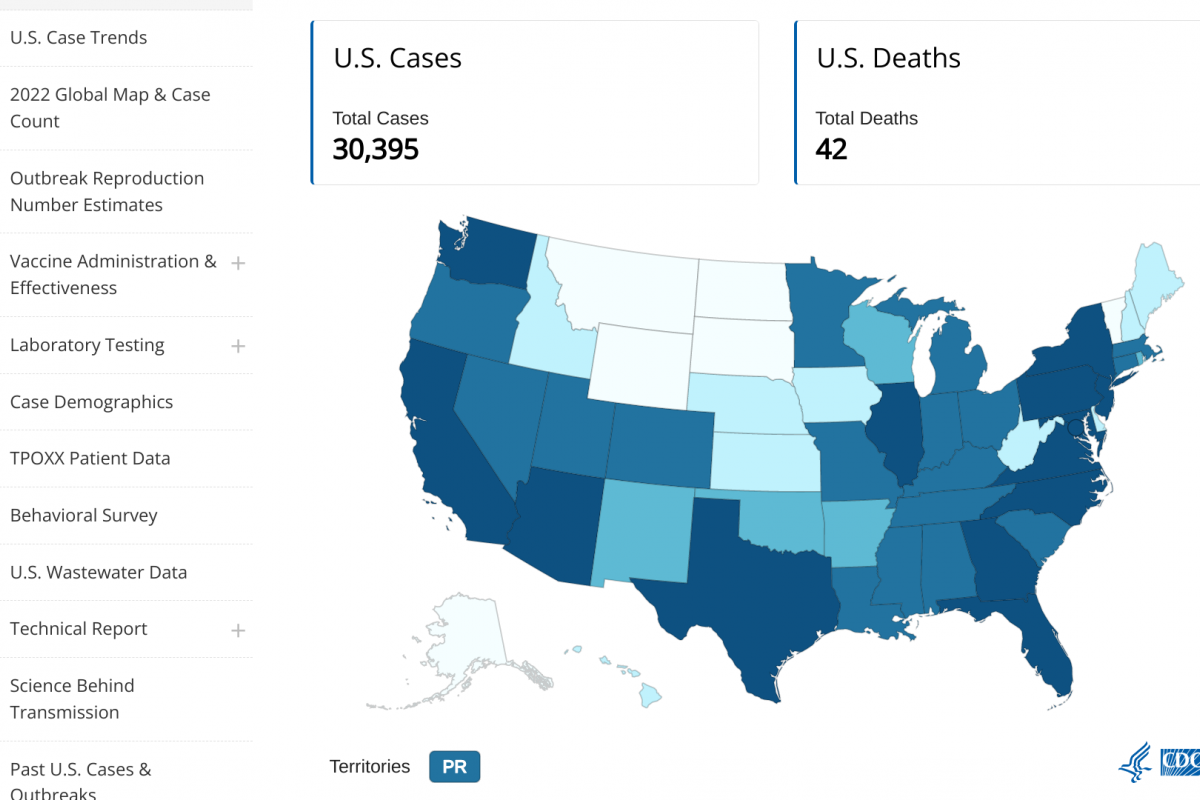
GSK plc today presented preliminary positive results from the phase III trial evaluating the immunological vaccine effectiveness and safety of its MenABCWY combination vaccine candidate.
On Mau 12, 2023, the vaccine candidate demonstrated non-inferiority in primary endpoints for five Neisseria meningitidis serogroups (A, B, C, W, and Y) compared to two doses of Bexsero® (meningococcal group B vaccine) and one dose of Menveo (meningococcal group A, C, W-135, and Y conjugate vaccine) in 10–25-year-olds.
In addition, the vaccine candidate was generally well tolerated, with a safety profile consistent with Bexsero and Menveo.
In a separate confirmatory arm of this phase III trial, the MenABCWY vaccine candidate showed immunological effectiveness against a panel of 110 diverse meningococcal serogroup B (MenB) invasive strains, which account for 95% of strains circulating in the US.
Professor Terry Nolan, principal investigator for the phase III trial and Head of the Vaccine and Immunisation Research Group at the Peter Doherty Institute for Infection and Immunity at the University of Melbourne and Murdoch Children's Research Institute, said in a press release, "Meningococcal vaccination can help save lives, and these results are significant in moving one step closer to protection against five meningococcal serogroups with a single vaccine."
"The potential for a simplified immunization schedule could improve accessibility for the target population susceptible to meningococcal disease."
Five Neisseria meningitidis serogroups (A, B, C, W, and Y) account for nearly all invasive meningococcal disease (IMD) cases worldwide.
Meningitis B is the most common serogroup in the US, accounting for more than half of meningococcal disease cases among 16–20-year-olds.
Currently, immunization coverage rates for Men B are estimated at approximately 31% of adolescents in the US.
GSK stated it is working closely with regulatory agencies to review the complete phase III data set, including the supplemental Biologics License Application for Bexsero, to confirm full licensure under the Accelerated Approval pathway. Detailed results will be submitted for publication in a peer-reviewed scientific journal later in 2023.
Precision Vaccinations publishes meningococcal vaccine news.