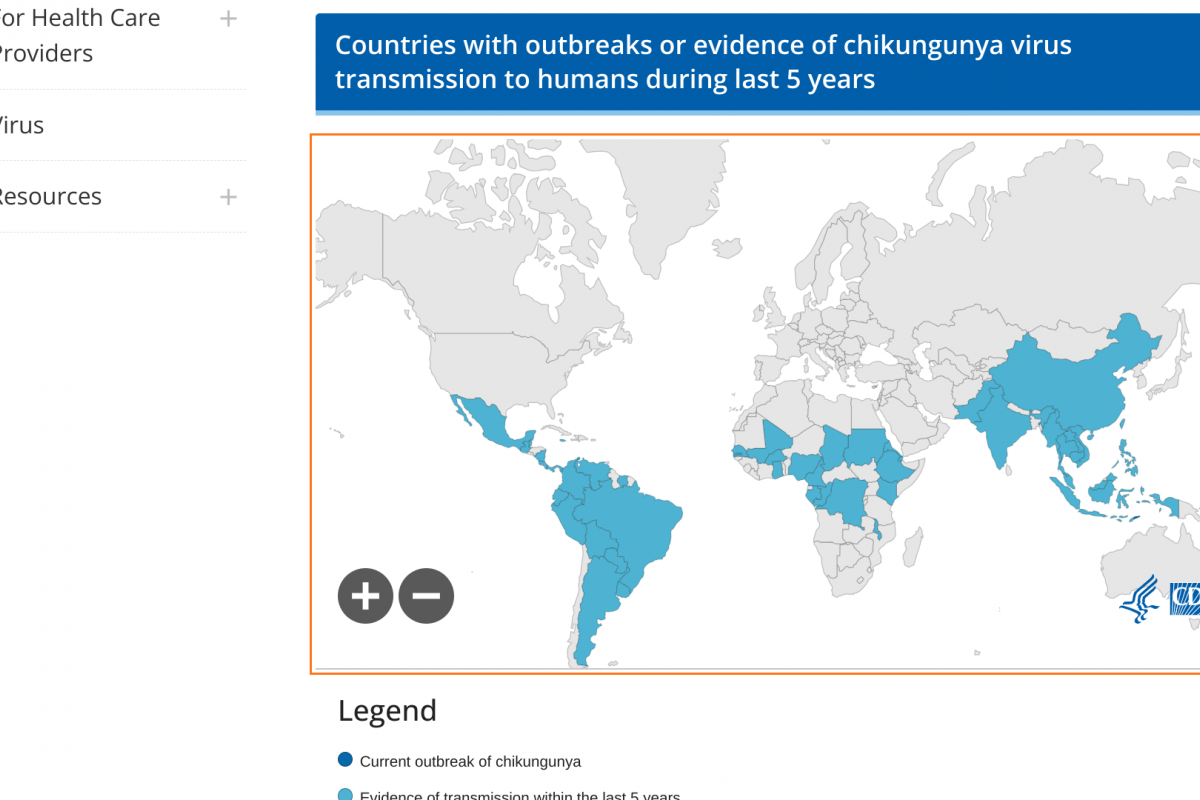
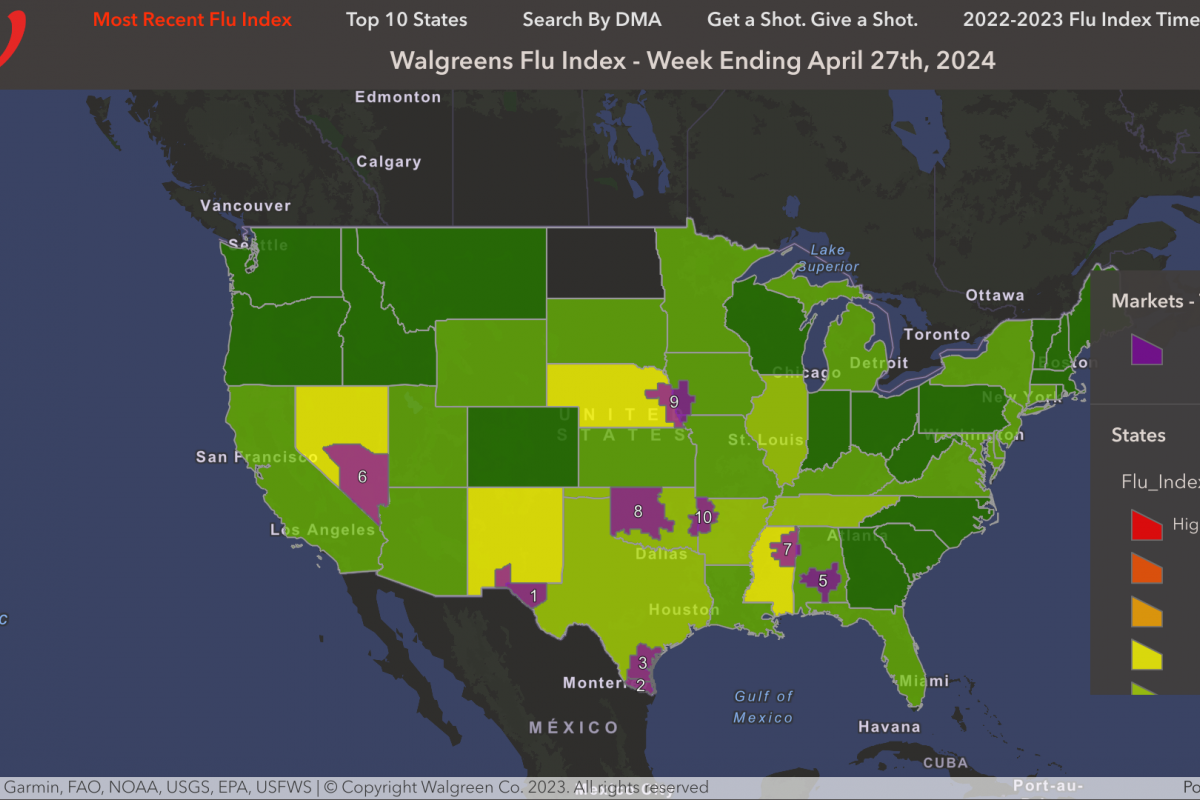
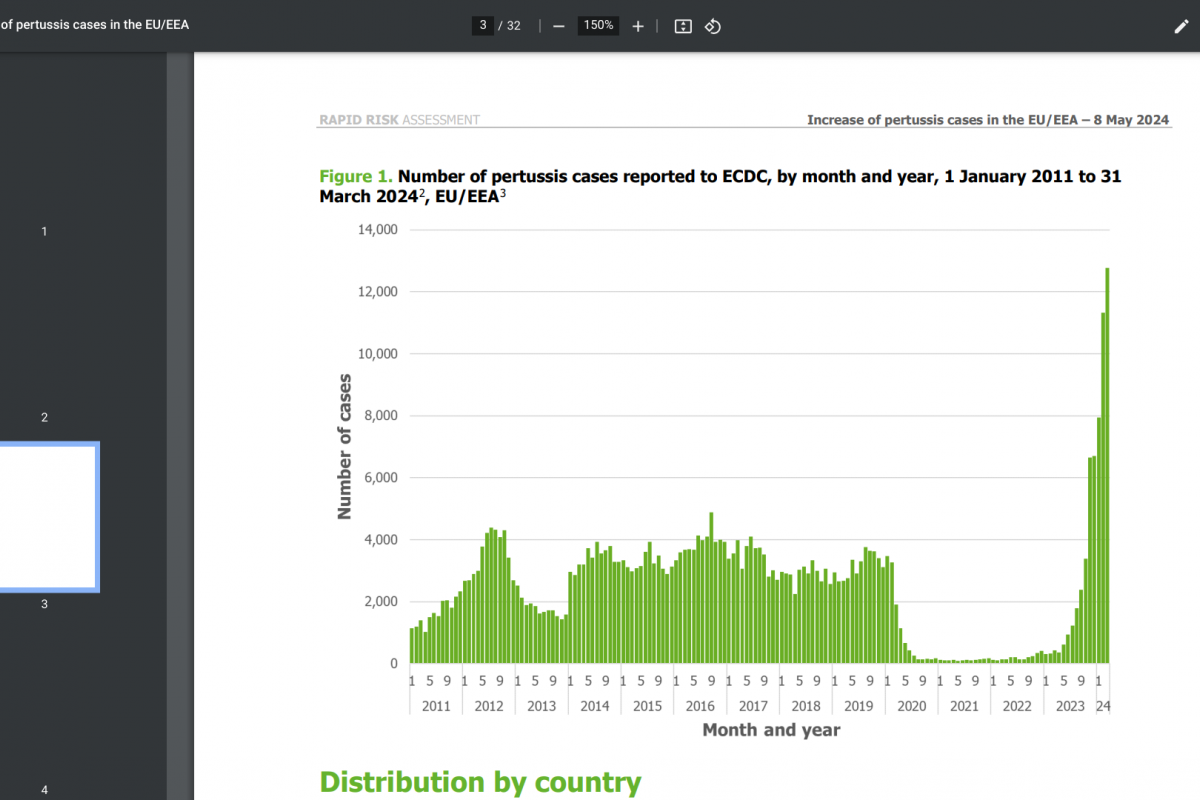
Although several countries have adopted a single-dose human papillomavirus (HPV) vaccination strategy, many other countries, such as the United States, continue to include multiple doses in their vaccination programs.
As of May 2024, six vaccines are authorized globally to protect males and females against cancers caused by HPV.
According to an article published by the Lancet Infectious Diseases on May 8, 2024, there are ethical reasons to transition to a single-dose strategy.
These scientists discuss how a single-dose HPV vaccination strategy advances equity in three dimensions: vaccine equity, health equity, and gender equity.
Adopting a single-dose strategy eases pressure on vaccine supply, lowers program costs, and is easier to distribute.
This change facilitates vaccine procurement and implementation programs (contributing to vaccine equity) and reaching hard-to-reach people or populations (contributing to health equity).
A lower number of cases of HPV-related diseases that stem from greater vaccine distribution reduces the burden on women, who are at a higher risk of HPV-related disease or who act as caregivers, which prevents them from accessing opportunities that contribute to their empowerment (contributing to gender equity).
Thus, these scientists wrote that pursuing the single-dose HPV vaccination program strategy is ethically desirable.
In April 2022, WHO's Strategic Advisory Group of Experts on Immunization concluded that a single-dose HPV vaccine delivers virus protection comparable to 2-dose schedules.
In the U.S., the Centers for Disease Control and Prevention (CDC) HPV vaccination has been recommended for women since 2006 and for men since 2011. Current CDC HPV vaccination schedules were updated in 2023.