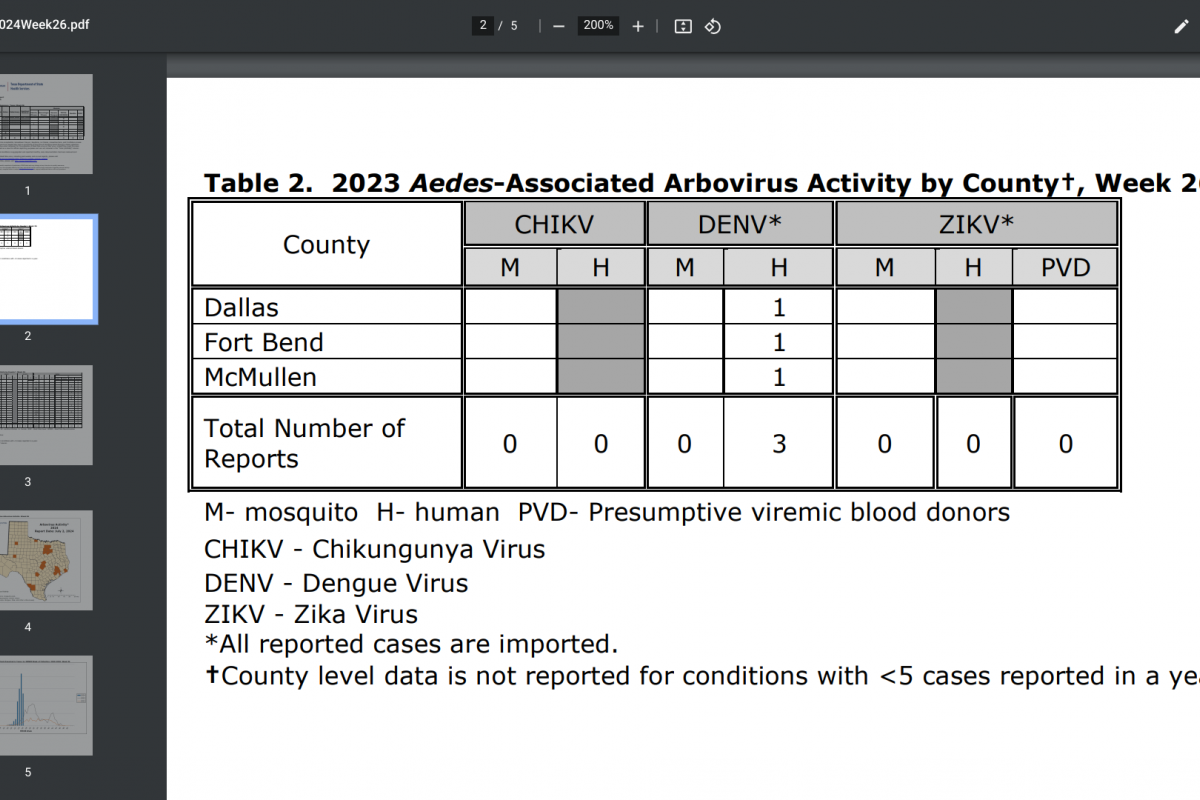
CSL Seqirus today announced that it has commenced shipping its differentiated portfolio of influenza vaccines. This year, the company's influenza vaccines are being produced as trivalent influenza vaccine formulations, in compliance with the U.S. Food and Drug Administration's directive in 2024 to remove the B/Yamagata strain.
For the 2024-2025 influenza vaccine portfolio, CSL Seqirus is the leading manufacturer offering a differentiated influenza vaccine option approved for use in people six months and older.
For example, FLUCELVAX® is the first and only cell-based influenza vaccine indicated for use in people six months and older.
"Influenza continues to pose a significant threat, as evidenced by recent flu seasons," said Dr. Gregg Sylvester, Chief Health Officer, CSL Seqirus, in a press release on July 9, 2024.
"As we begin distributing influenza vaccines to healthcare providers throughout the U.S., it is imperative that we work to maintain high vaccination rates this season to help reduce the burden of influenza-related illnesses and the risk of severe outcomes."
CSL Seqirus is part of CSL, a global leader in the protection of public health and one of the largest influenza vaccine providers in the world.