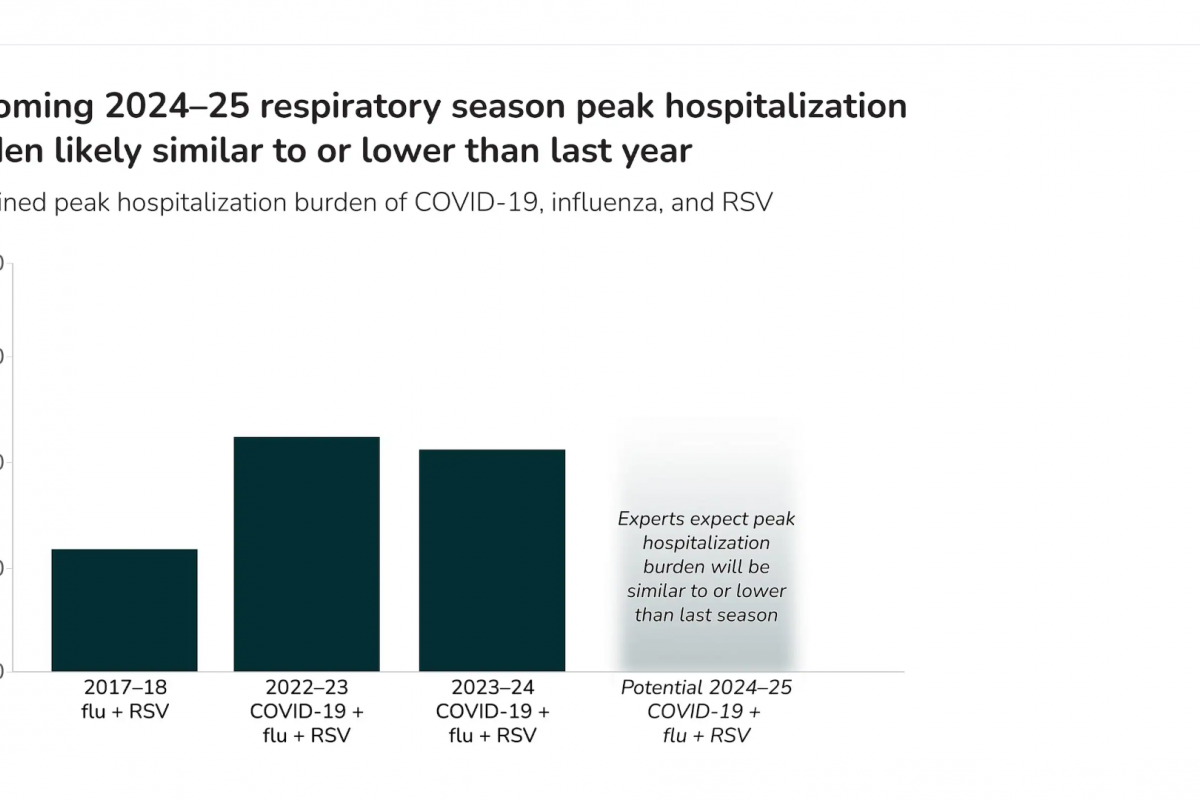
As the 2024-2025 Respiratory Syncytial Virus (RSV) season expands in the United States, one approved vaccine shared some positive news.
GSK plc today announced new data from a phase 3 clinical trial evaluating the efficacy and safety of a single dose of AREXVY RSV vaccine against lower respiratory tract disease (LRTD) over three full RSV seasons.
Published on October 8, 2024, a single dose of AREXVY's cumulative efficacy over three full RSV seasons was clinically meaningful at 62.9% against RSV-LRTD and 67.4% against severe RSV-LRTD compared to placebo.
In the third season, the vaccine’s efficacy was 48% against RSV-LRTD.
These results include efficacy against different RSV subtypes in older adults (70-79) and those with certain underlying medical conditions.
In a press release, GSK's chief scientific officer, Tony Wood, said, “We are excited by these new data, which show that a single dose of AREXVY could help protect millions of older adults at risk of RSV disease over three seasons to benefit public health."
"This is the only RSV vaccine with efficacy and safety data available through three full seasons."
As of October 2024, the U.S. FDA had approved three RSV vaccines and one monoclonal antibody therapy.