Search API
The World Health Organization (WHO) recently released its 62nd situation report on the multi-country mpox outbreak, emphasizing the ongoing circulation of all clades of the monkeypox virus (MPXV).
In 2025, there were 52,845 reported cases of mpox and 215 related fatalities across 98 countries, with about 44,000 instances originating in Africa.
The WHO urges continued vigilance, enhanced surveillance, and rapid response to contain outbreaks, warning that reduced activities could lead to underreporting.
In a related announcement on January 22, 2026, H.E. Dr. Jean Kaseya, the Director General of the Africa Centres for Disease Control and Prevention (Africa CDC), announced that the agency has lifted its declaration of a Public Health Emergency of Continental Security concerning mpox.
He highlighted improvements in country capacity, effective international partnerships, a sustained decline in reported suspected and confirmed cases, and a reduction in case fatality rates.
The Africa CDC stated that this change "signals a transition from emergency response to a sustained, country-led pathway toward the elimination" of mpox.
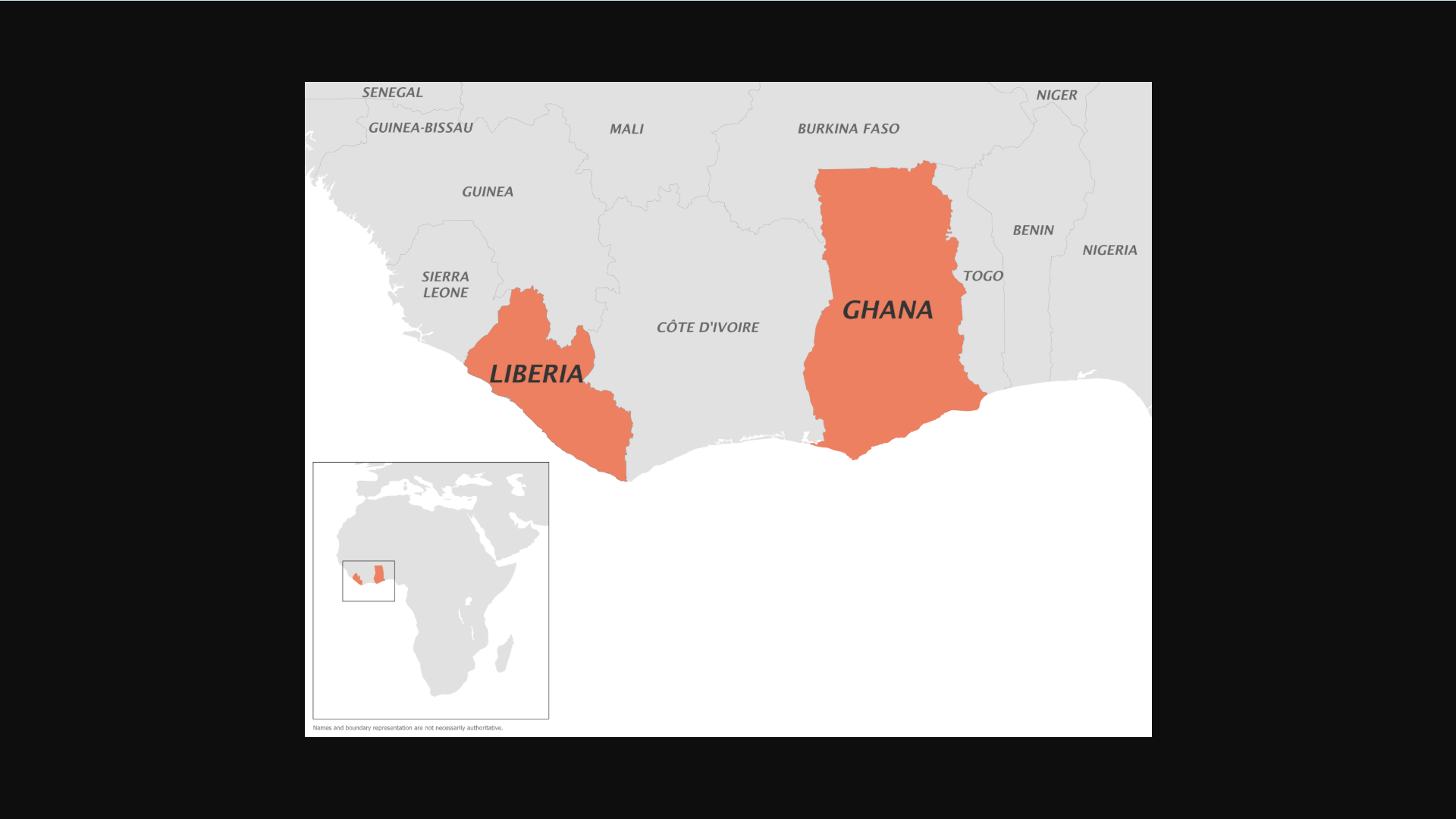
Separately, the U.S. CDC issued a Level 2 Travel Health Notice on January 26, 2026, confirming there are outbreaks of clade II monkeypox in Ghana and Liberia, where monkeypox is endemic. Unlike the ongoing global clade II mpox outbreak that began in 2022, these outbreaks have affected males and females approximately equally.
The CDC says that if you are sick and could have mpox, follow isolation and infection control measures at home and during travel.
The CDC recommends mpox vaccination (JYNNEOS®, MVA-BN®) for travelers visiting Ghana and Liberia in 2026. For those recommended to receive vaccination, get two doses at least 28 days apart.
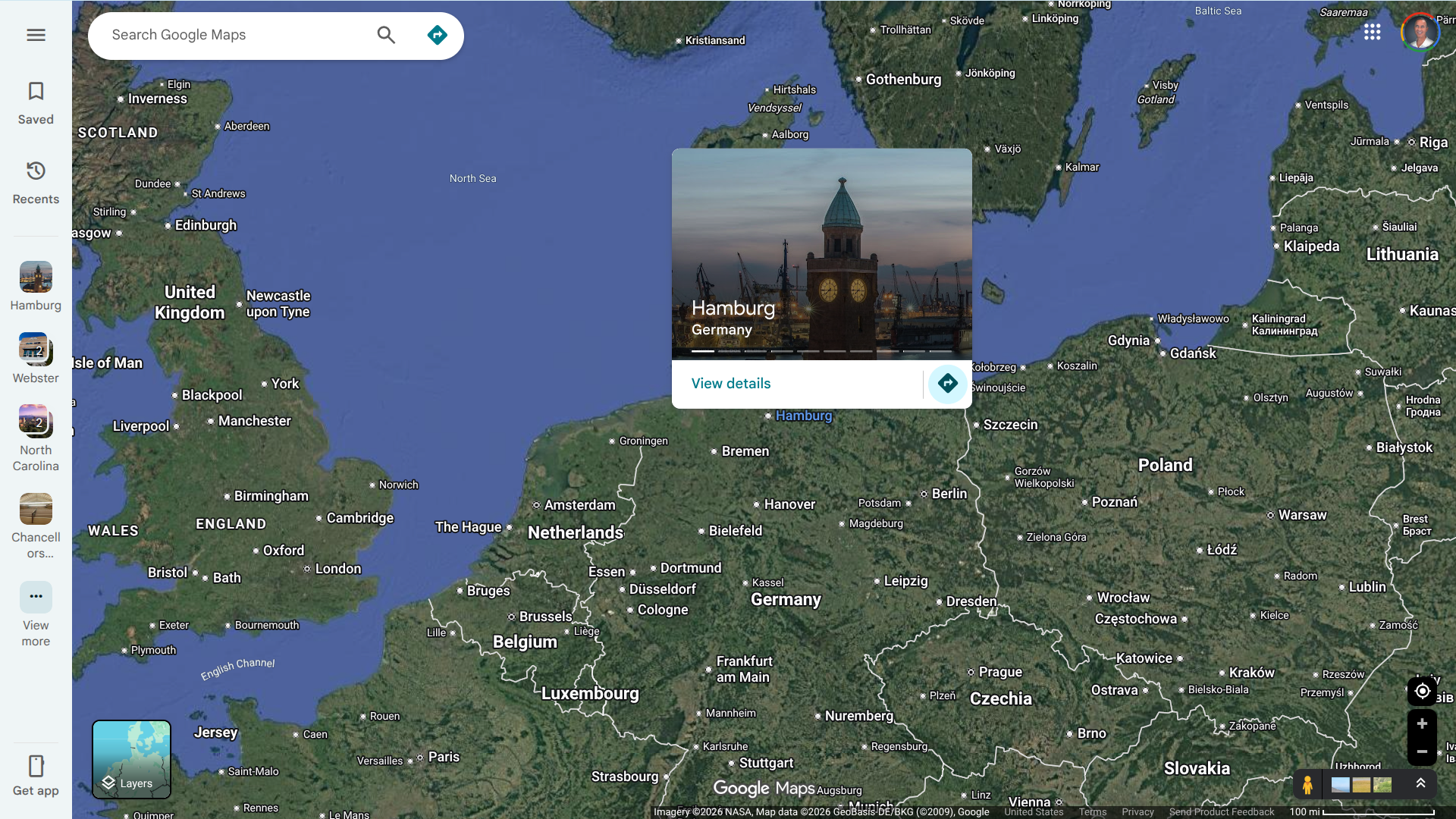
German health authorities have confirmed the detection of two additional environmental samples positive for wild poliovirus type 1 (WPV1) in Hamburg, a city in northern Germany.
The wastewater samples were collected from the same sewage site in October 2025.
As of January 21, 2026, this marks the first detection of WPV1 in Germany and the first such finding in Europe since 2010.
Significantly, no human cases of paralytic polio have been associated with these findings. Furthermore, there is no evidence of local community transmission or circulation of the virus within Germany.
Genetic analysis indicates that the isolates from these samples in Hamburg are closely linked, suggesting they originate from the same introduction of the WPV1 virus.
The virus strain found in these samples is genetically related to a WPV1 strain previously identified in an environmental sample from Kandahar, Afghanistan, in late August 2025.
The detections appear to be confined to wastewater, likely resulting from virus excretion by one or more individuals who were in the area around the time of sampling—possibly travelers or visitors from regions where WPV1 remains endemic, such as parts of Afghanistan or Pakistan.
Except for those countries, the WHO South-East Asia Region has maintained its polio-free status in 2026.
These detections in Germany were reported as part of the routine environmental surveillance in collaboration with the Global Polio Eradication Initiative (GPEI). The GPEI stresses the need for continued efforts to achieve the complete eradication of poliovirus worldwide.
Currently, the U.S. government identifies 32 countries where poliovirus is a health risk, and advises international travelers to speak with a healthcare provider about immunization options, including a polio booster dose when visiting at-risk areas in 2026.
In 2026, as the world confronts emerging infectious diseases, the World Health Organization (WHO) remains at the forefront of developing vaccine candidates against the deadly Nipah virus (NiV).
The WHO has outlined a comprehensive pipeline of NiV vaccine candidates, which includes live-attenuated and replication-defective recombinant platforms based on various viruses, such as poxviruses, vesicular stomatitis virus (VSV), adenovirus, measles, rabies, and virus-like particles, as well as subunit vaccines.
As of January 27, 2026, no vaccine or treatment for NiV has received authorization from the U.S. Food and Drug Administration or the European Medicines Agency.
However, clinical trials are actively progressing, marking significant milestones in vaccine development.
In 2026, the UK and The Pirbright Institute are supporting the development of vaccines against henipaviruses, including the NiV.
One standout candidate is the ChAdOx1 NipahB vaccine, developed by the University of Oxford's Pandemic Sciences Institute. In December 2025, the university launched the world's first Phase II clinical trial in Bangladesh, a known hotspot for NiV outbreaks.
This trial is conducted in partnership with the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b), and is funded by the Coalition for Epidemic Preparedness Innovations (CEPI). The trial builds on successful Phase I results from 2024, where the vaccine demonstrated safety and immunogenicity in 51 participants.
Professor Dame Sarah Gilbert, a Vaccinology professor at the University of Oxford and developer of the vaccine, highlighted the significance of these advancements in a press release: "This new trial in Bangladesh marks an important step forward in our work to develop a vaccine against the Nipah virus, a deadly health threat that currently lacks an approved vaccine or treatment."
Additionally, the Serum Institute of India is collaborating with Oxford and CEPI to manufacture up to 100,000 investigational doses of ChAdOx1 NipahB, creating the world's largest NiV vaccine reserve for potential emergency use.
While clinical trial challenges remain, such as evaluating efficacy without large outbreaks, these trials represent a critical push toward countering NiV.
As CEPI's investments and international collaborations, such as Hendra@30, the global community moves closer to developing vaccines to prevent future outbreaks caused by this bat-borne virus.
The Ministry of Health of the Federal Democratic Republic of Ethiopia today officially declared the end of the country's first-ever outbreak of Marburg Virus Disease (MVD), a severe and often fatal hemorrhagic fever.
This declaration was made on January 26, 2026, after two consecutive incubation periods—totaling 42 days—without any new confirmed cases.
This timeline aligns with the World Health Organization (WHO) DON592 recommendations for declaring the end of an MVD outbreak.
The outbreak was first confirmed by the WHO on November 14, 2025, in the South Ethiopia Region, particularly in Jinka.
As of January 2026, there had been a cumulative total of 19 cases: 14 laboratory-confirmed cases (including nine deaths and five recoveries) and five probable cases, all of which were fatal.
This Horn of Africa outbreak marks the 20th reported case of MVD globally. Previous outbreaks have occurred in several African countries, including Angola, the Democratic Republic of the Congo, Equatorial Guinea, Ghana, Guinea, Kenya, Rwanda, South Africa, Uganda, and most recently, Tanzania.
MVD is caused by the Marburg virus, which is transmitted to humans from fruit bats and through human-to-human contact via bodily fluids. Since it was first observed in 1967 during an outbreak in West Germany, the case fatality rate can exceed 80% in some outbreaks.
Currently, there are no licensed vaccines or specific treatments for the Marburg virus, but research is ongoing. The Marburg Virus Vaccine Consortium, established in response to earlier outbreaks in Guinea and Ghana, is advancing vaccine development.
And in early January 2026, the Oxford Vaccine Group and the Pandemic Sciences Institute confirmed they are collaborating with the Institute for Drug Discovery at Leipzig University and Moderna, with support of up to $26.7 million from the Coalition for Epidemic Preparedness Innovations and the European Union's Horizon Europe program to develope multivalent vaccines that provide broad protection against multiple filoviruses, including the Ebola virus (Zaire ebolavirus), Sudan virus, Bundibugyo virus, and MVD.
Vax-Before-Travel will continue monitoring developments related to emerging infectious diseases.
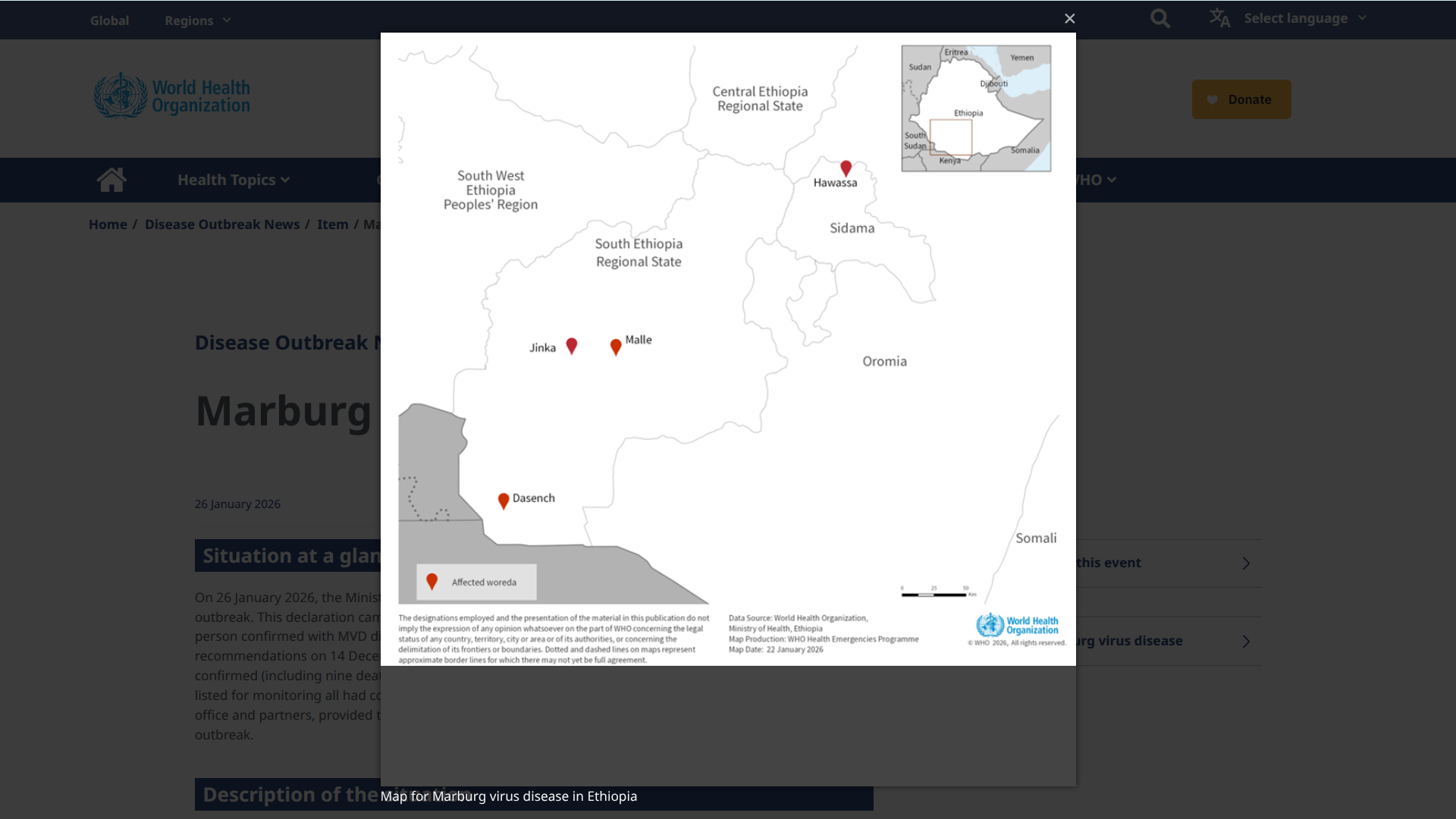
The U.S. Centers for Disease Control and Prevention (CDC) recently announced that seasonal influenza activity is continuing to decline nationwide; however, trends differ among various age groups and regions.
As of January 25, 2026, the CDC's FluView report for week #3 noted a 4.4% decrease in visits to healthcare providers for respiratory illnesses compared to the previous week.
Tragically, the CDC also reported twelve additional influenza-associated pediatric deaths last week, bringing the total for the 2025-2026 season to 44 reported deaths related to influenza in children. Among the children who were eligible for influenza vaccination and had known vaccination status, approximately 90% of the reported pediatric deaths this season occurred in those who were not fully vaccinated against influenza.
However, the CDC's in-season severity assessment framework classified this flu season as moderate across all ages.
To visualize this CDC flu information, the Walgreens Respiratory Illness Index updated its national graphic that highlights which states are reporting flu activity at retail pharmacies.
According to Walgreens, the current leaders include Louisiana, Michigan, Nevada, and Oklahoma.
Since the flu season is expected to last a few more months, pharmacies and clinics continue to offer various flu shots throughout the U.S. Additionally, the FluMist vaccine can be administered at home in 2026.
Bavarian Nordic A/S today announced an agreement with Eurofarma, granting the Brazilian pharmaceutical company exclusive rights to sell and distribute Bavarian Nordic's chikungunya vaccine, CHIKV VLP (VIMKUNYA®), in Brazil.
Additionally, under the terms of the agreement announced on January 23, 2026, Eurofarma will have the right of first refusal for any future opportunities to register and commercialize the virus-like particle (VLP) vaccine in the rest of Latin America.
According to the press release, pending discussions with the Brazilian Health Regulatory Agency (Anvisa), a regulatory submission is anticipated in the first half of 2026, which could support a potential launch of the vaccine in Brazil in the latter half of 2027.
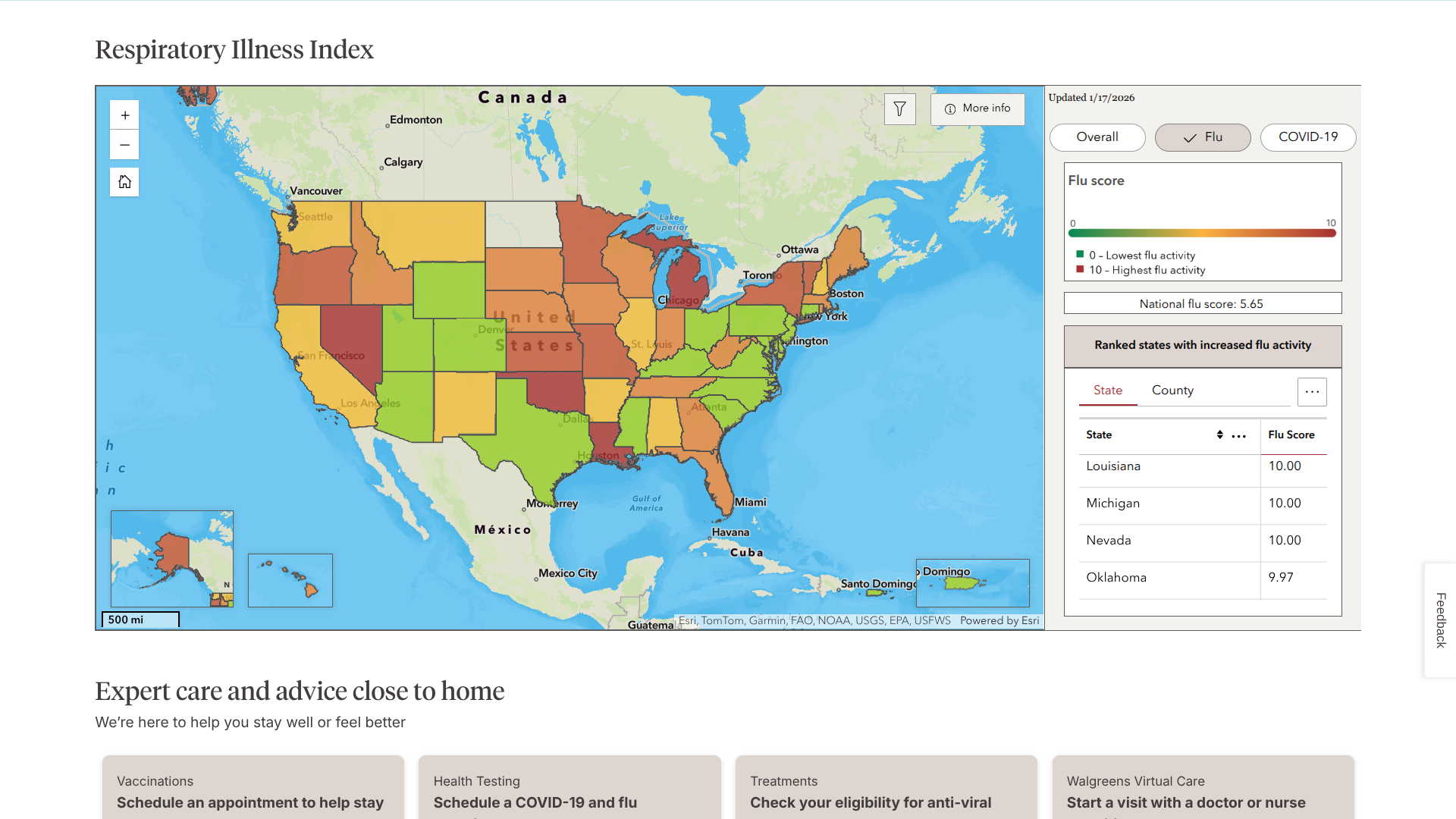
Chikungunya is a mosquito-borne viral disease caused by the chikungunya virus. Over the past two decades, chikungunya has been reported in over 110 countries. Since 2013, the virus has been detected in the Americas Region.
Brazil remains one of the most heavily affected countries, having reported a significant share of global cases and hospitalizations in recent years.
In 2025, Brazil reported over 250,000 chikungunya cases and 120 related fatalities.
Paul Chaplin, President & CEO of Bavarian Nordic, commented, "Brazil continues to bear a significant burden from chikungunya, accounting for a substantial portion of global reported cases and related deaths."
In the United States, both travel-related and locally acquired chikungunya have been confirmed in Florida in early 2026. During 2025, numerious cases were related to travelers from Cuba.
CHIKV VLP is a single-dose, prefilled, adjuvanted VLP recombinant protein vaccine designed for the active immunization and prevention of disease caused by the chikungunya virus.
The vaccine contains no viral genetic material, making it non-infectious and unable to cause disease, thereby providing a favorable safety profile suitable for a broad range of individuals.
VIMKUNYA® was approved by the U.S. Food and Drug Administration in February 2025, by the European Commission in February 2025, and by the United Kingdom in May 2025, initially for use in travelers and at-risk populations aged 12 years and older.
This is the second chikungunya vaccine targeting Brazil.
In April 2025, Anvisa granted marketing authorization to its single-dose vaccine IXCHIQ®.
As of January 24, 2026, VIMKUNYA® is commercailly offered at travel clinics throughout the U.S.
For more information, visit www.bavarian-nordic.com.
The Jamaica Tourist Board recently welcomed the U.S. Department of State's update to its Travel Advisory for Jamaica, which was downgraded to Level 2: Exercise Increased Caution, effective January 17, 2026.
This positive adjustment returns Jamaica to the advisory level it held before Hurricane Melissa, a Category 5 Caribbean storm in 2025, reflecting significant progress in the island's recovery and a continued commitment to visitor safety.
Recovery efforts have restored major airports, cleared roads, and reopened the majority of hotels and resorts, with tourism operations now vibrant across key destinations like Montego Bay, Negril, and Ocho Rios.
Edmund Bartlett, Minister of Tourism, stated, "The U.S. Department of State's decision to lower the advisory to Level 2 is a strong endorsement of Jamaica's swift and effective recovery."
"Our people, tourism partners, and government have worked tirelessly to ensure that visitors can once again enjoy the warmth, culture, and beauty that make Jamaica one of the world's premier destinations."
"We are open, welcoming, and ready for you."
For example, Jamaica's annual Reggae Month celebrations are held throughout the island each February.
The updated State Department advisory continues to highlight risks related to crime, health, and natural disasters, with no changes to the underlying risk indicators. It also notes that some high-risk regions remain designated as Level 4 ("Do Not Travel") due to crime concerns.
Additionally, Basic and specialized medical care may not be available in many parts of Jamaica. This includes slower emergency service response times and less availability of care for illness or injury.
Furthermore, the U.S. CDC recommends various routine and travel vaccinations before visiting Jamaica in 206. It also says there may be an increased risk of waterborne (including leptospirosis), vector-borne, and fungal diseases.
When planning a trip to Jamaica in 2026, the State Department advises enrolling in the Smart Traveler Enrollment Program to receive digital updates and alerts from the U.S. embassy or consulate.
For more information, visit jamaica.com or contact the Jamaica Tourist Board.