The U.S. State Department will soon begin correctly spelling the Republic of Turkey as "Türkiye" in formal settings.
According to the U.S. Department of State spokesperson Ned Price, on January 5, 2023, the name change was approved by the U.S. Board on Geographic Names following a request from the Turkish embassy.
In June 2021, President Recep Tayyip Erdogan requested the name change by launching a global rebranding campaign. He asked the rest of the world to embrace his country's original name, not the anglicized version.
"Türkiye is the best representation and expression of the Turkish people's culture, civilization, and values," President Erdogan commented.
Turkish people have called their country Türkiye since 1923, at the end of the Ottoman Empire and when the Turkish Republic was formed.
Data sources indicate Türkiye recorded 16 million tourists in 2020, ranking 15th worldwide.
Türkiye's cosmopolitan capital Ankara is positioned in the central Anatolia region.
While Istanbul, formerly known as Constantinople, is Turkey's largest city and is the gateway between Europe and Asia across the Bosphorus Strait.
While the U.S. Centers for Disease Control and Prevention does not have an active travel notice, its website says to check the vaccines and medicines list and visit your doctor or pharmacist at least a month before visiting Türkiye to get what is needed for your trip.
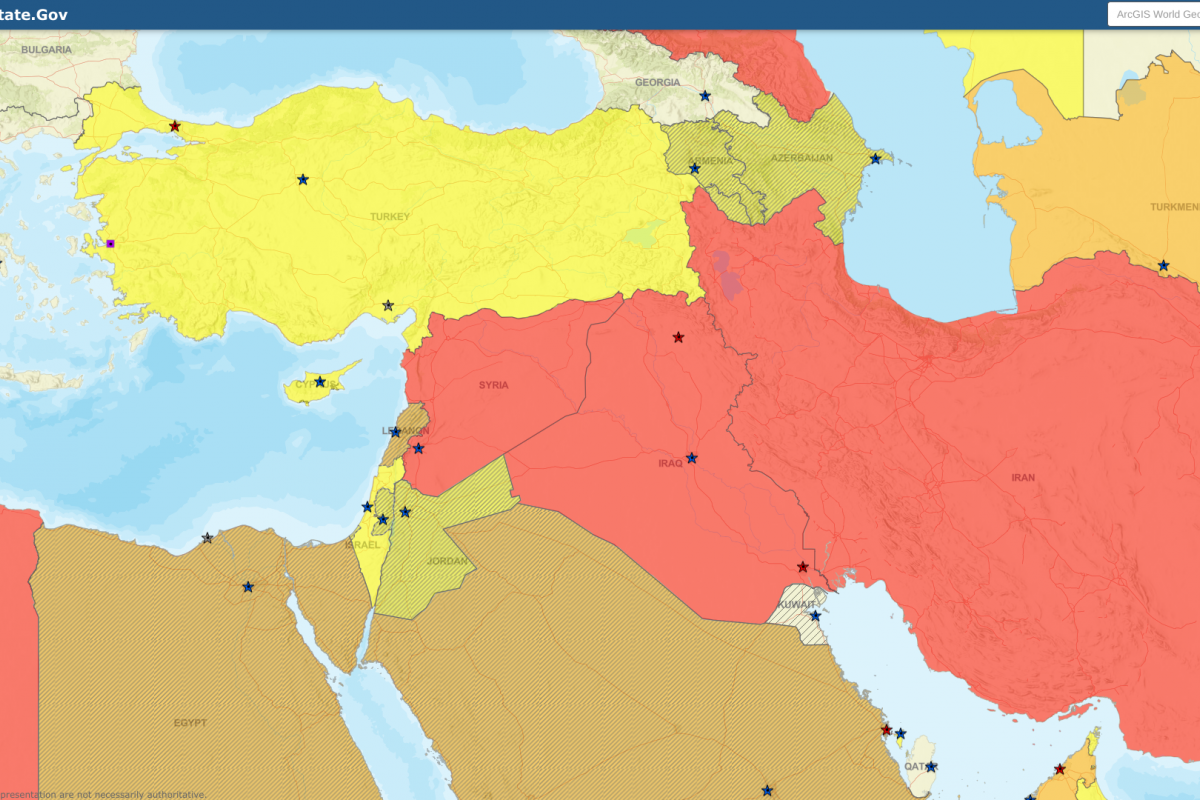
And the Department of Stated suggests visitors exercise increased caution when traveling to Türkiye as some areas have increased risk.
The U.S. Embassy in Ankara is located at 1480 Sok No:1 Cukurambar Mah Cankaya 06530, Ankara.