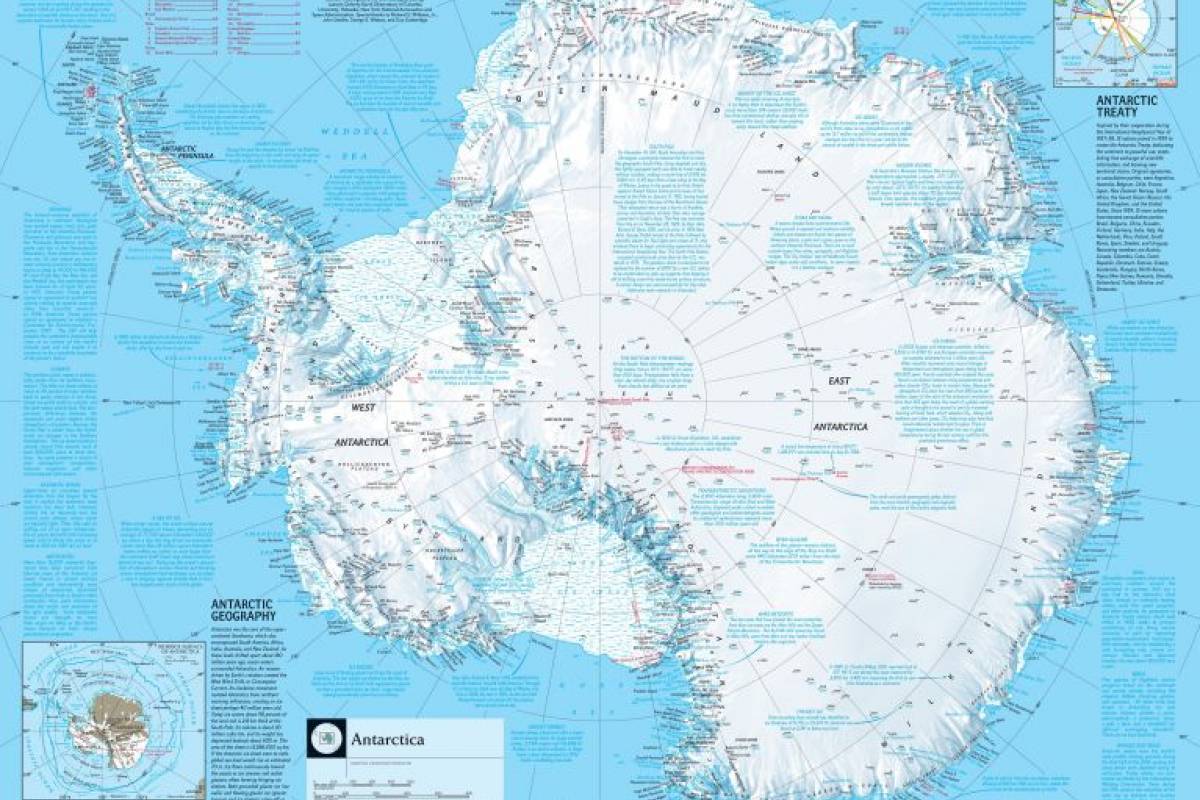
The U.S. Department of State recently updated it's Level 2: Exercise Increased Caution for when visiting the southernmost continent of Antarctica.
As of January 19, 2023, the State Department announced travelers should be aware of environmental hazards posed by extreme and unpredictable weather and limited emergency services.
For travelers to Antarctica, severe low temperatures and high winds are the primary health hazards.
Temperature patterns vary widely because Antarctica is covered in continuous darkness during the winter and constant sunlight during the summer.
The sun's effects in Antarctica can damage the eyes and skin, and protective measures should be taken.
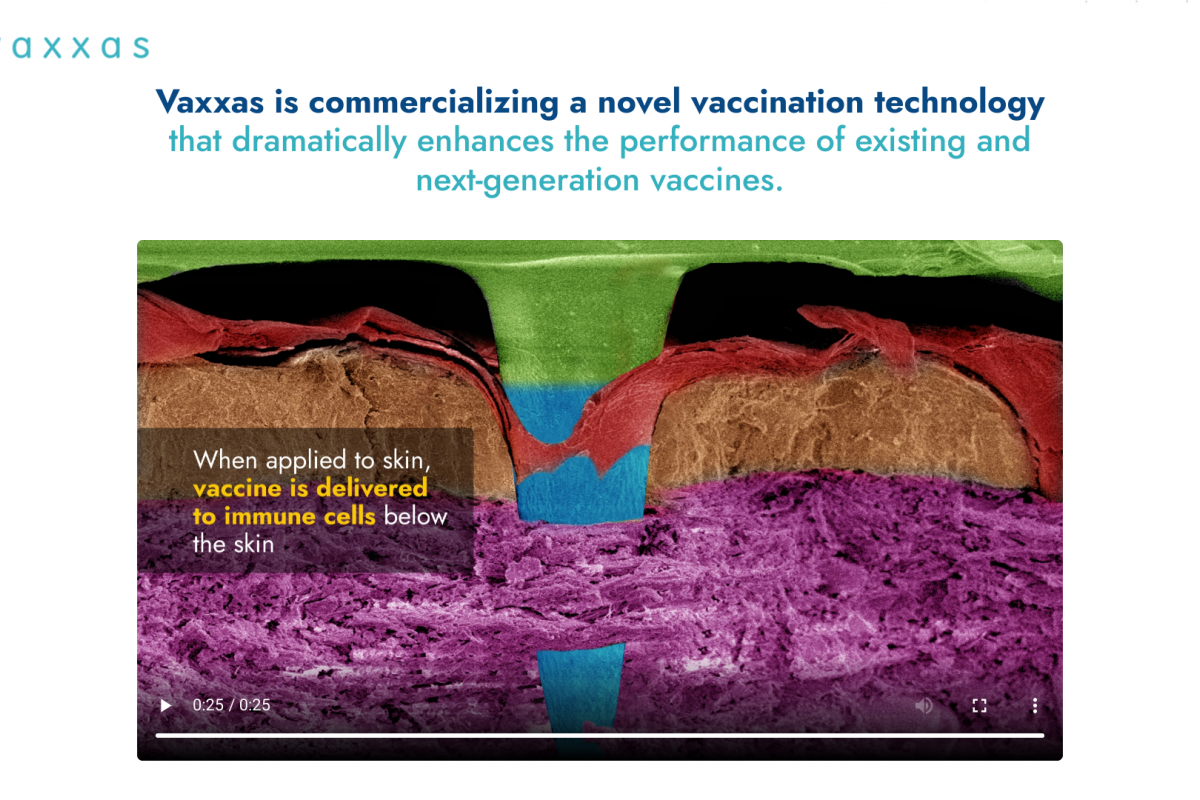
The U.S. Centers for Disease Control and Prevention (CDC) suggests various travel vaccinations, such as measles, before visiting Antarctica.
And if you travel to Antarctica, obtain comprehensive travel, medical, and medical evacuation insurance; see our webpage for more information on insurance providers for overseas coverage, says the State Department.
The U.S. government does not maintain an embassy or consulate in Antarctica.
If you need U.S. consular services when in Antarctica, contact the closest U.S Embassies/Consulates in Argentina, Australia, Chile, New Zealand, and South Africa.
From outside the U.S., call 1-202-501-4444 to speak with a U.S. representative.
Furthermore, government resources in the Antarctic Region are committed to the Antarctic Program (USAP).
Private expeditions should be self-sufficient and are encouraged to carry adequate insurance coverage against the risk of incurring financial charges or material losses while in the Antarctic.
The National Science Foundation, as manager of the USAP, reserves the right to seek, in accordance with international and domestic law, recovery of all direct and indirect costs of any such emergency search and rescue.
The Antarctic also includes island territories within the Antarctic Convergence, says the National Geographic Society.
Most of the islands and archipelagos of Lesser Antarctica are volcanic and heavily glaciated.
The islands of the Antarctic region are the South Orkney Islands, South Shetland Islands, South Georgia, and the South Sandwich Islands, all claimed by the United Kingdom.
Peter I Island and Bouvet Island, claimed by Norway.
Heard and McDonald islands, claimed by Australia.
And Scott Island and the Balleny Islands, claimed by New Zealand.