The Centers for Disease Control and Prevention (CDC) recently released a Morbidity and Mortality Weekly Report (MMWR) titled: Interim Clinical Treatment Considerations for Severe Manifestations of Mpox—United States, February 2023.
Published on March 3, 2023, this MMWR provides updated clinical treatment considerations about using therapeutic countermeasures to treat severe mpox cases.
Until data gaps are filled through randomized controlled studies and other carefully controlled research studies, this MMWR represents the best available information about human mpox treatment.
Previously, the CDC updated the U.S. National Mpox Vaccination Strategy on February 6, 2023.
The CDC continues to recommend people who have been exposed to the Mpox virus and people who may be more likely to contract mpox should be vaccinated.
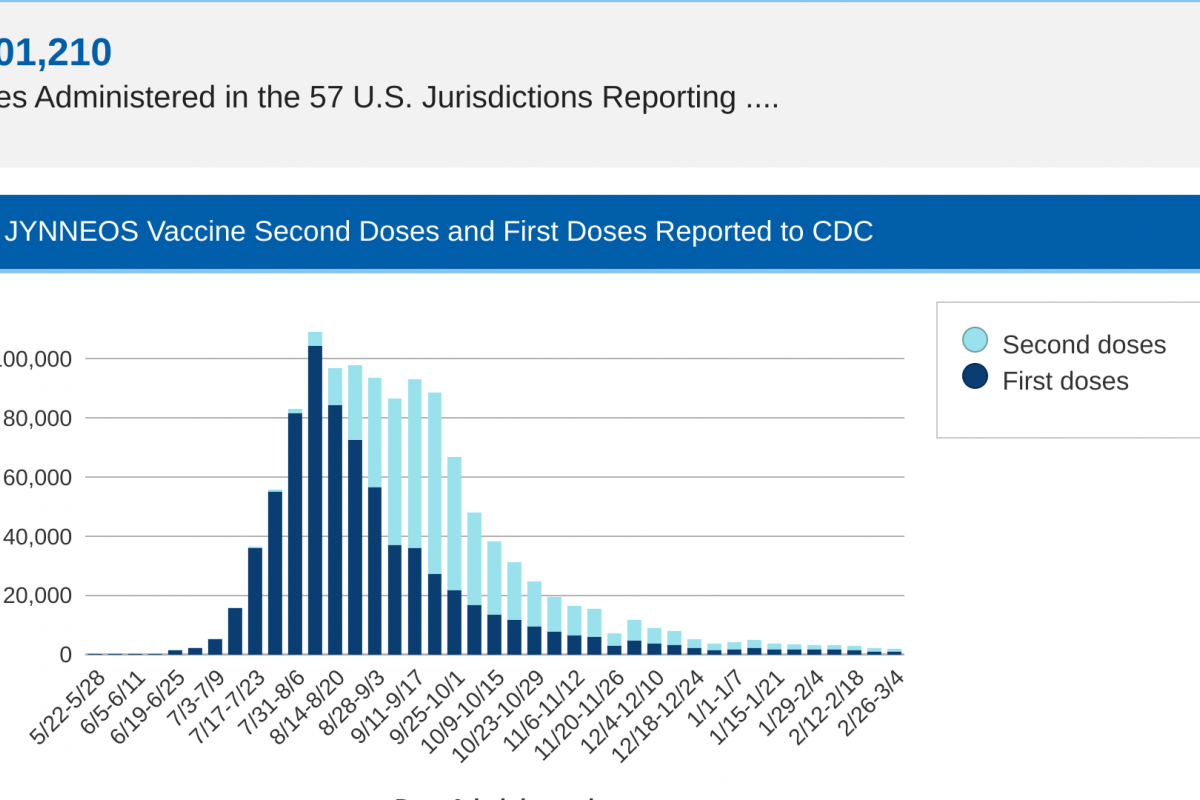
The JYNNEOS® vaccine is available at certain clinics and pharmacies in the U.S.
Mpox is a disease caused by infection with the Monkeypox virus, an Orthopoxvirus in the same genus as the Variola virus, which causes smallpox.
In 2022, a global outbreak involving mpox clade IIb was detected sourced from the Canary Islands in early May. Since then, 1,201,210 JYNNEOS doses have been administered in the 57 U.S. Jurisdictions reported data.