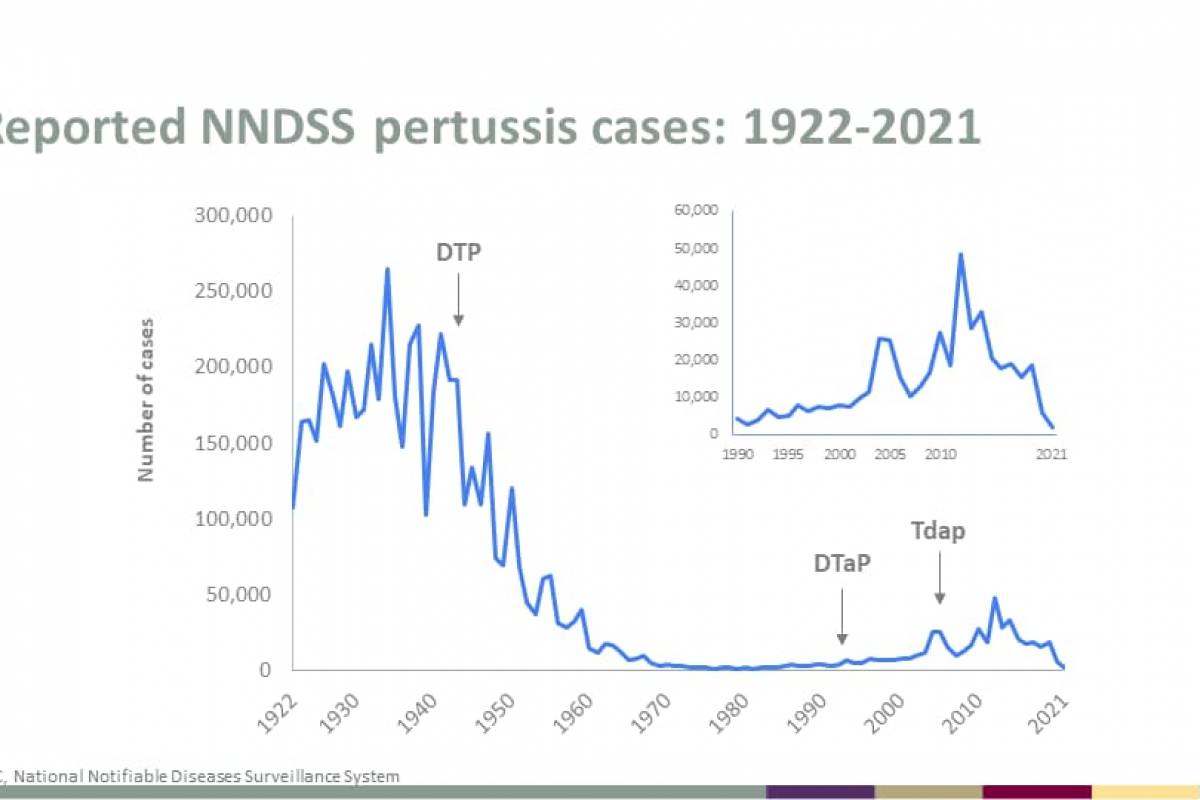
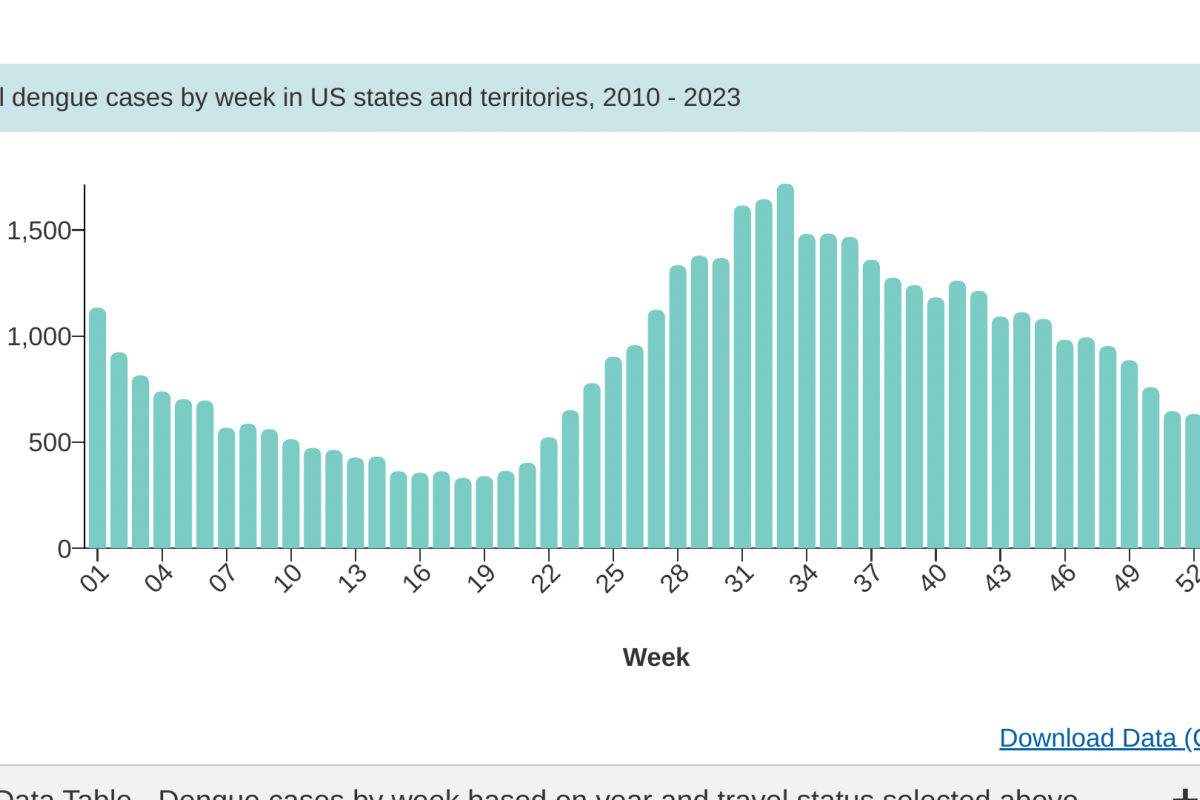
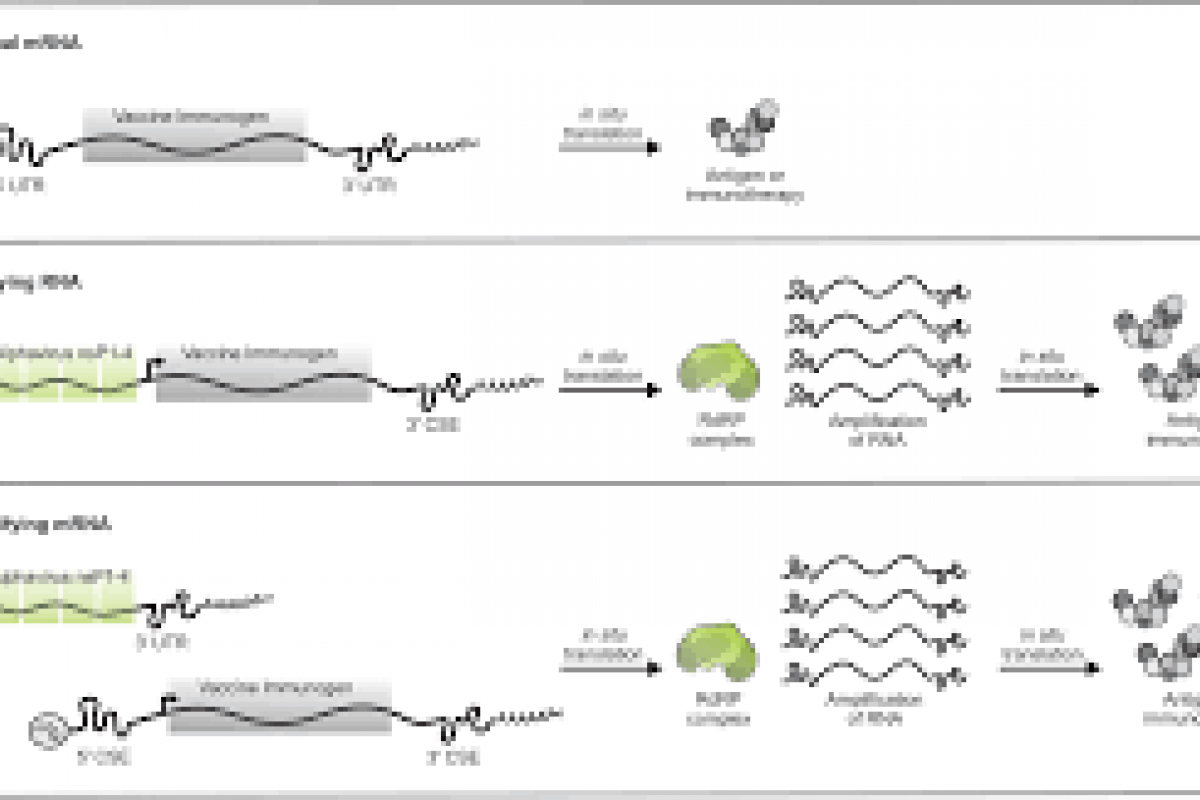
The U.S. Centers for Disease Control and Prevention (CDC) recently reported that three additional measles cases were confirmed in 2024.
As of May 23, 2024, the CDC confirmed a total of 142 measles cases were reported by 21 jurisdictions: Arizona, California, Florida, Georgia, Illinois, Indiana, Louisiana, Maryland, Michigan, Minnesota, Missouri, New Jersey, New York City, New York State, Ohio, Pennsylvania, Vermont, Virginia, Washington, West Virginia, and Wisconsin.
Recently, the city of Philadelphia reported another measles case after managing an outbreak earlier in 2024.
About 44% of these cases are in unvaccinated children under five years of age, with 63% hospitalized.
Ten measles outbreaks (3+) were reported in 2024, compared to four outbreaks in 2023.
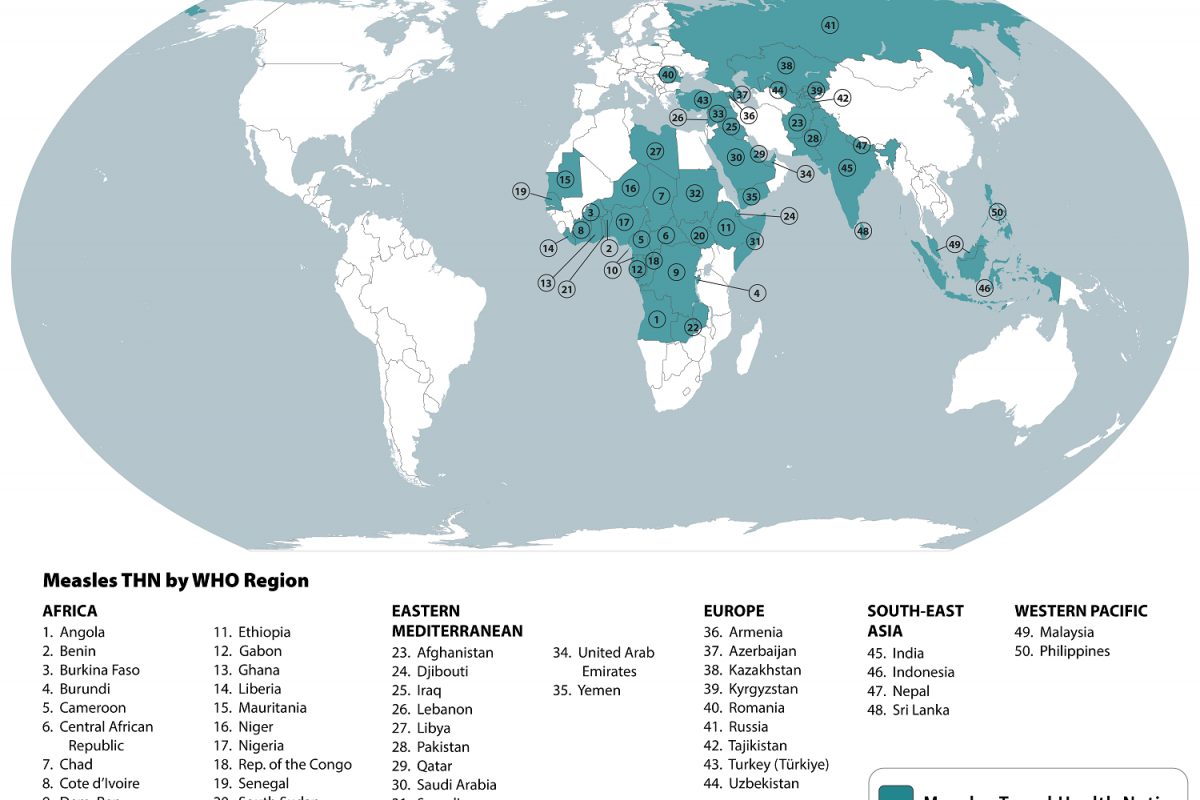
Globally, about 50 countries have reported measles outbreaks in the past year.
The CDC says it is uncommon for someone fully vaccinated to develop measles. In 2024, the number of breakthrough infections (approximately 5%) was consistent with previous years' reports.
Two doses of MMR vaccine are 97% effective at preventing measles, and one dose is 93% effective. Measles vaccines are available at most clinics and pharmacies in the U.S.