Valneva SE announced today that the European Commission (EC) has granted marketing authorization in Europe for the IXCHIQ® vaccine, which is used to prevent diseases caused by the chikungunya virus in adults.
IXCHIQ® is the world’s only licensed chikungunya vaccine.
The EC decision marks the third approval the Company has received for IXCHIQ® following approval from the U.S. FDA in late 2023 and Health Canada last month.
On July 1, 2024, Valneva stated it expects to deliver the first vaccine doses in Europe in the fourth quarter of 2024.
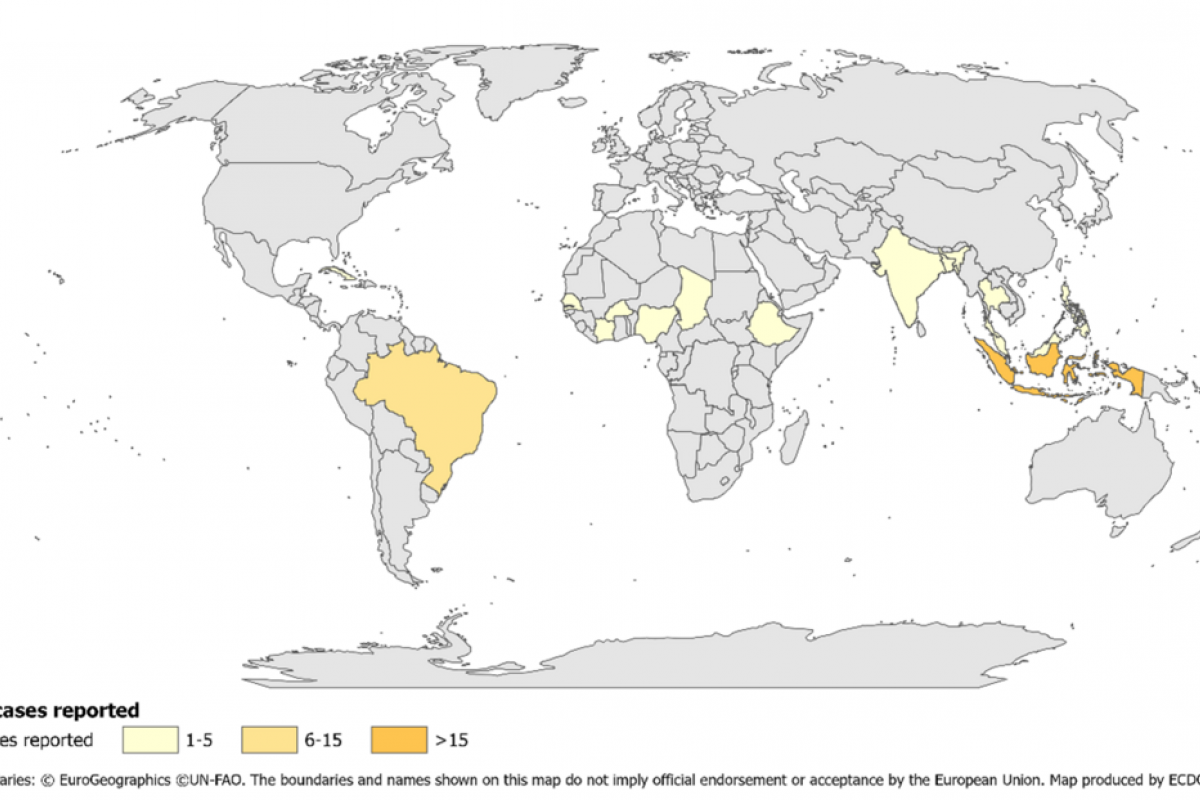
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, commented in a press release, “The EC approval marks a crucial milestone toward making this vaccine available to as many European citizens as possible .... It is critical to provide a vaccine solution not only to European travelers going to endemic chikungunya areas, such as South America or Africa.
Valneva has also submitted a Marketing Authorization Application to the UK Medicines and Healthcare products Regulatory Agency and the Brazilian Health Regulatory Agency, with potential approval in 2024.