PharmaJet® today announced their support for the World Health Organization’s (WHO) polio eradication campaigns in Pakistan.
Starting in May 2023, PharmaJet is providing an additional 5 million syringes to the WHO for a supplemental immunization activity campaign.
Chris Cappello, President and CEO, PharmaJet, commented in a press release on May 23, 2023, “We are pleased to continue our collaboration with the WHO for over five years with a common goal of global eradication of polio."
"Now, with over 12 million needle-free syringes provided across multiple polio vaccination campaigns, we have confidence that our Precision Delivery Systems are safe, effective, and can be rapidly deployed in response to emergencies anywhere on the globe."
The WHO previously introduced needle-free intradermal delivery of inactivated polio vaccine (ID-IPV) in Pakistan and Somalia using the PharmaJet Tropis ID PDS to immunize millions of children.
Published results from Pakistan and recent survey results from Somalia showed that the Tropis PDS is an effective and preferred solution for polio immunization campaigns that can help increase campaign coverage by over 18%.
An ongoing study evaluating Tropis use for Routine Immunization Administration of the IPV vaccine has been previously reported.
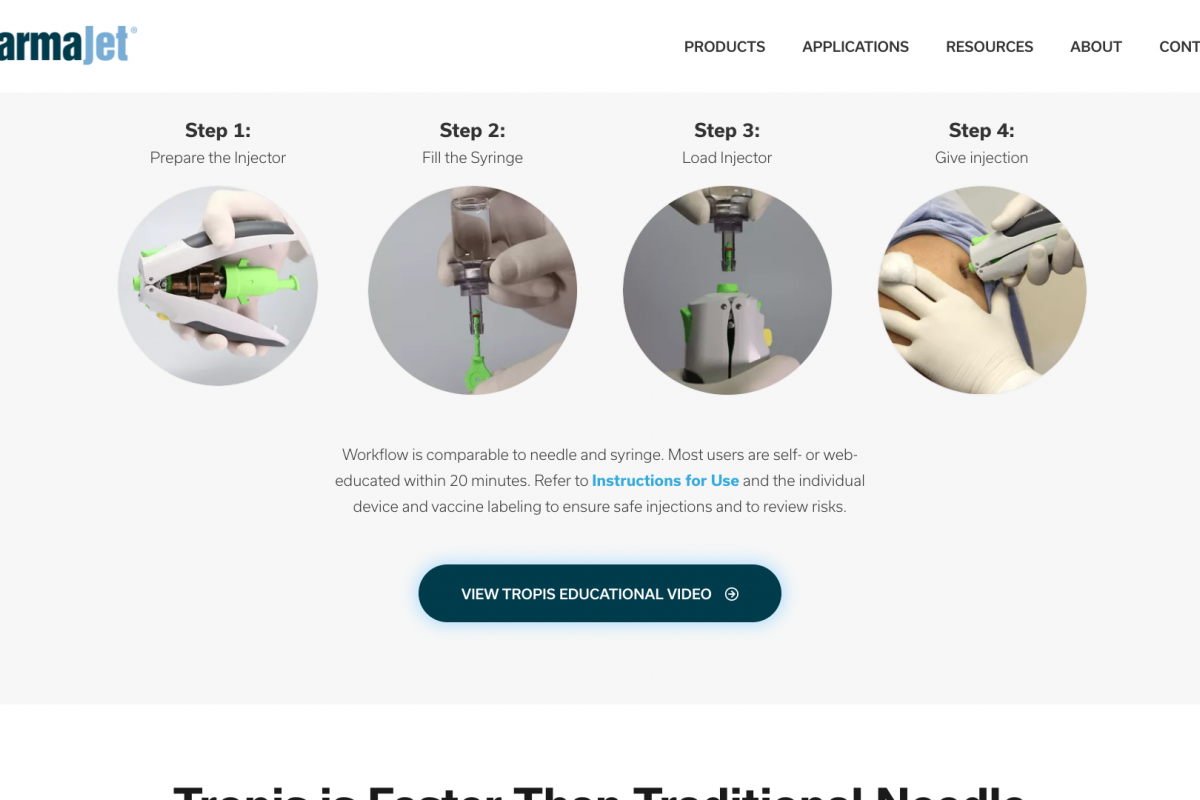
The PharmaJet Tropis Precision Delivery System™ was selected based on its proven cost savings, ease of training, and improved immunization coverage benefits.