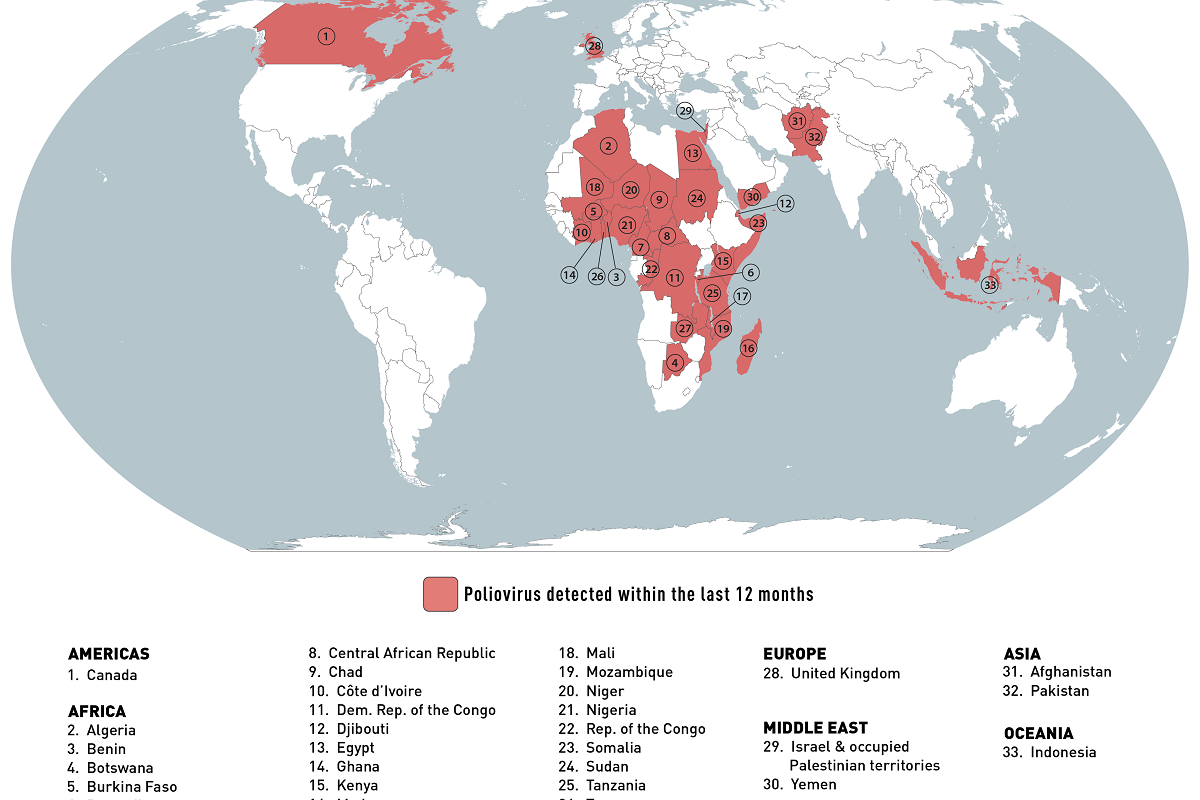
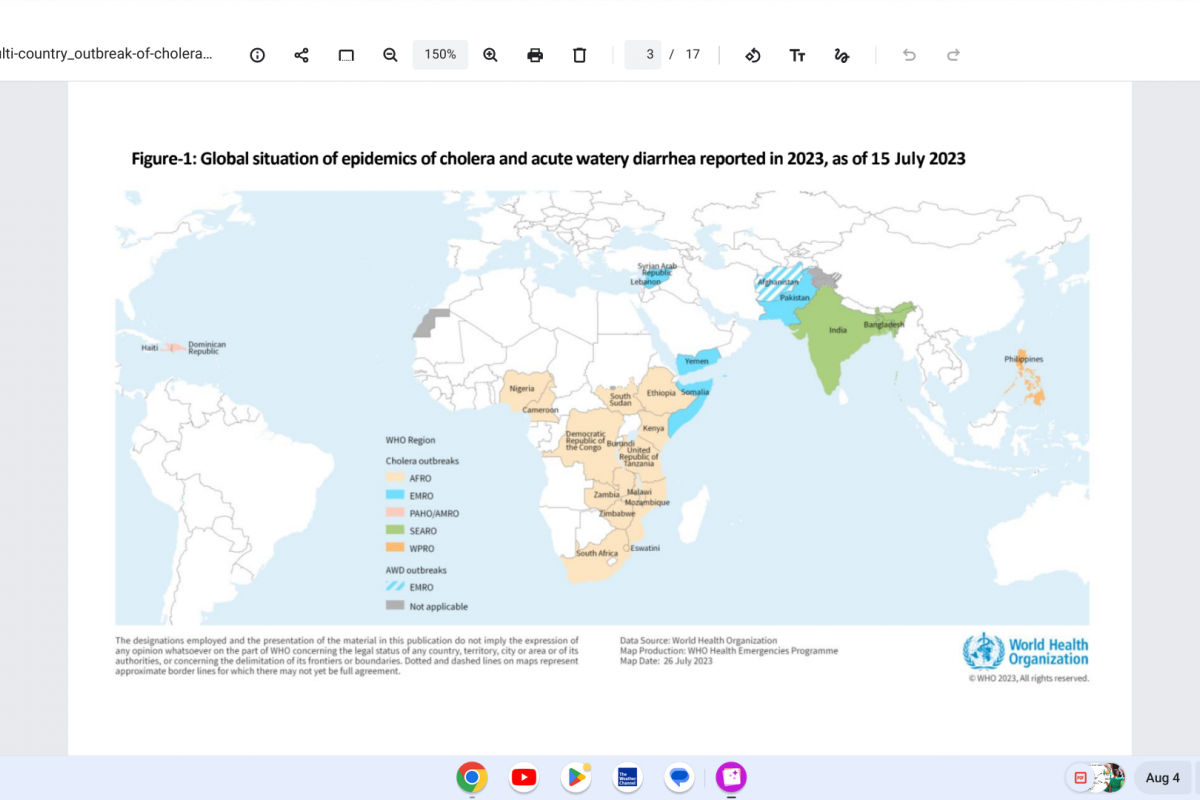
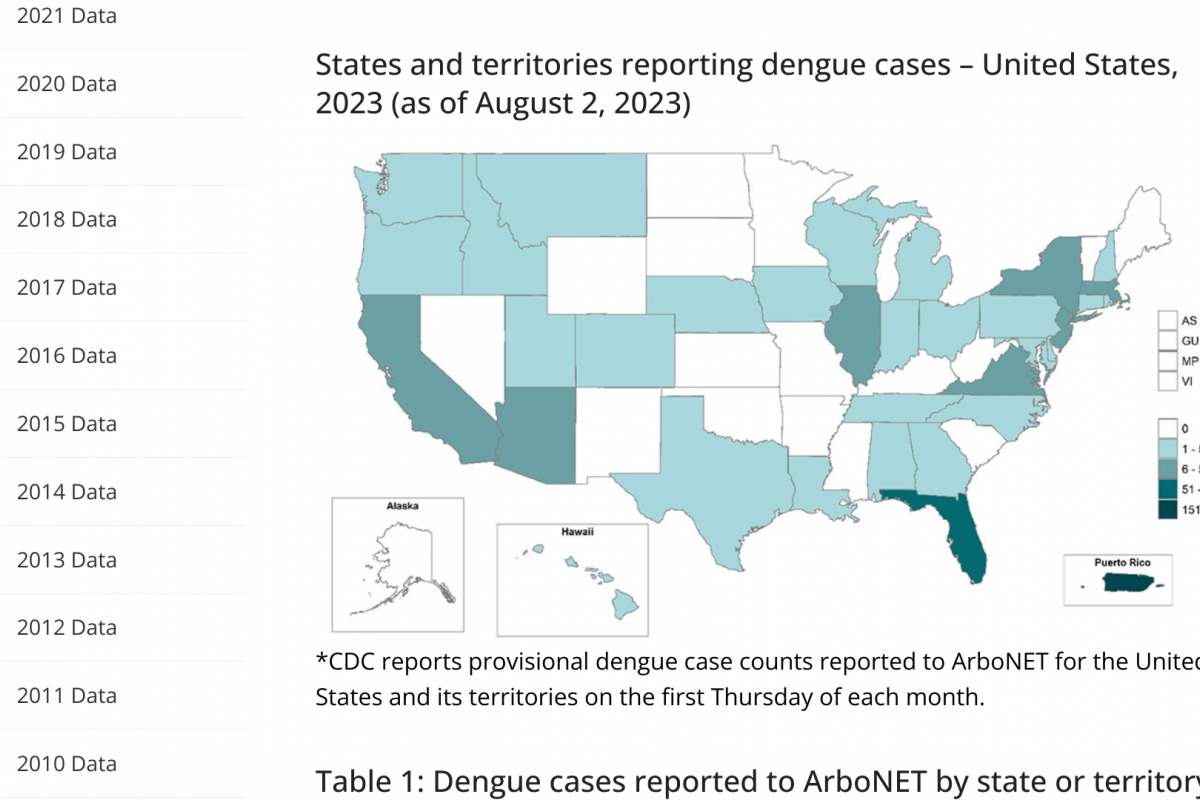
Every year, rare human infections with zoonotic influenza viruses usually spread in birds and pigs. The U.S. Centers for Disease Control and Prevention (CDC) recently reported the first two human infections with swine flu viruses in 2023.
These infections were caused by two different types of flu viruses that generally spread among pigs, and they occurred in two people who attended various agricultural fairs in Michigan and had exposure to pigs.
These influenza A(H1N2)v virus infections were thoroughly investigated to ensure that such viruses are not spreading in people and to limit further exposure of people to infected animals if infected animals are identified.
As of August 4, 2023, the CDC recommends people take precautions to prevent the spread of swine influenza viruses to people and has guidance for people exhibiting pigs at fairs, people attending fairs, and fair organizers.
Since 2005, over 500 swine influenza infections have been identified in the U.S.
Unlike, avian influenza (Bird Flu), which has recently spread among birds, mammals, and humans, there are no approved swine flu vaccines.
Furthermore, the CDC says the annual 'flu shot' does not prevent swine influenza infections in people.