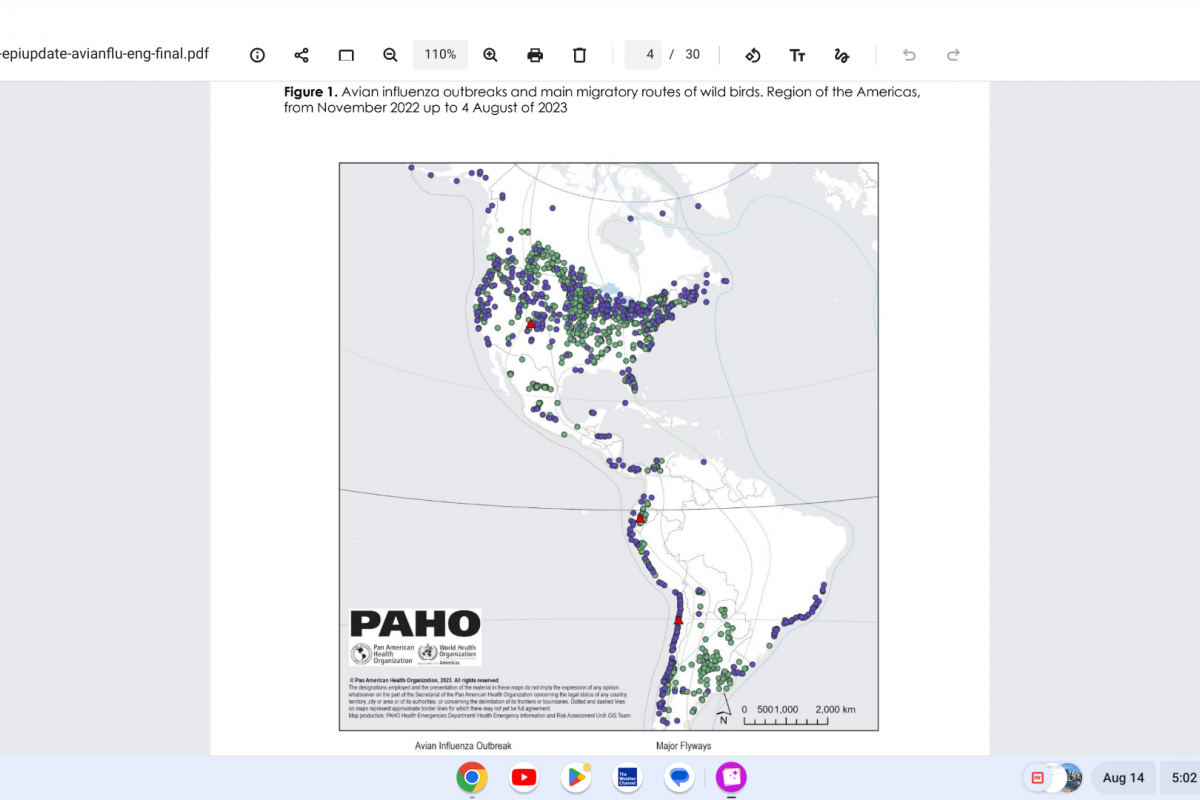
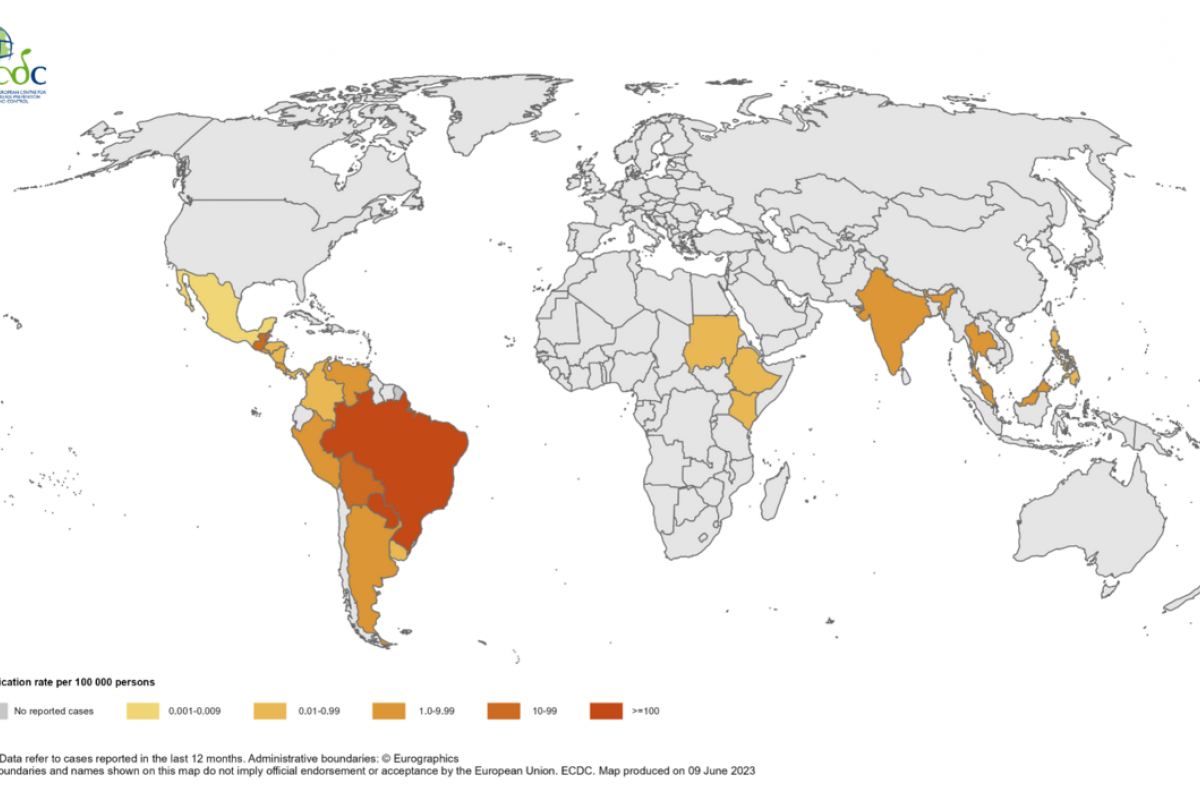
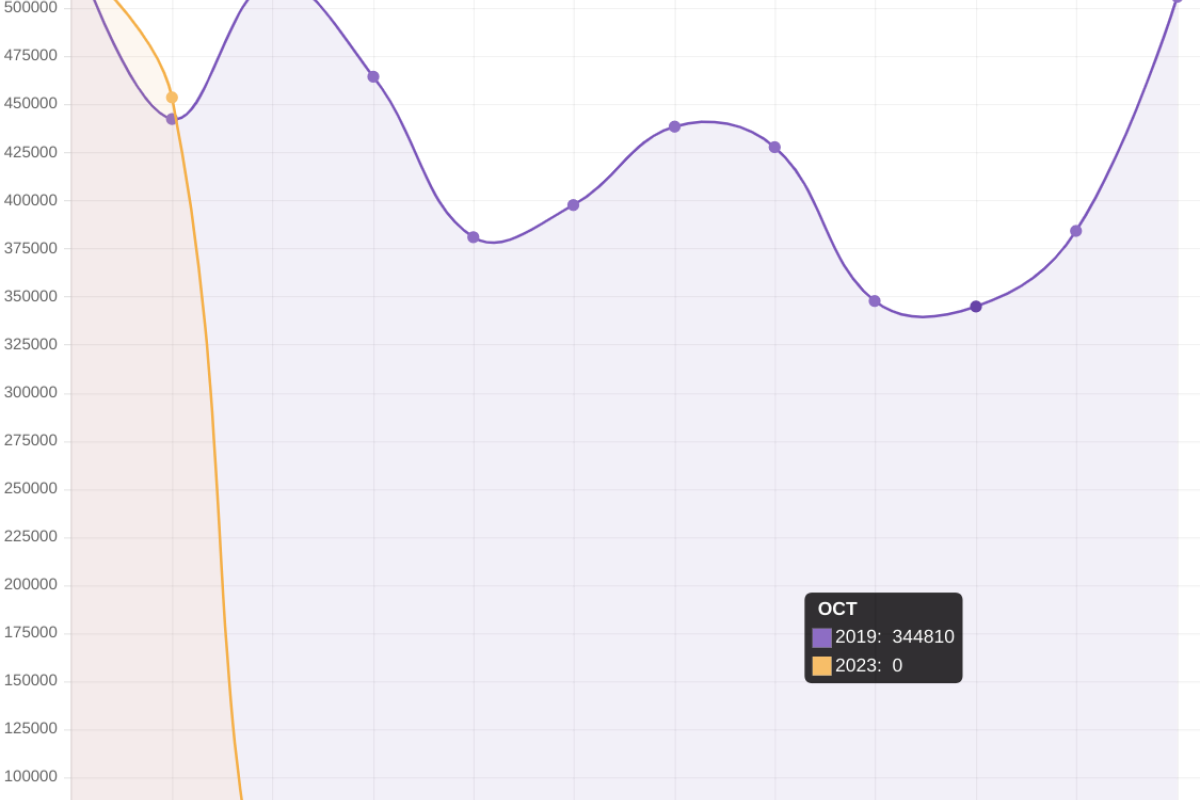
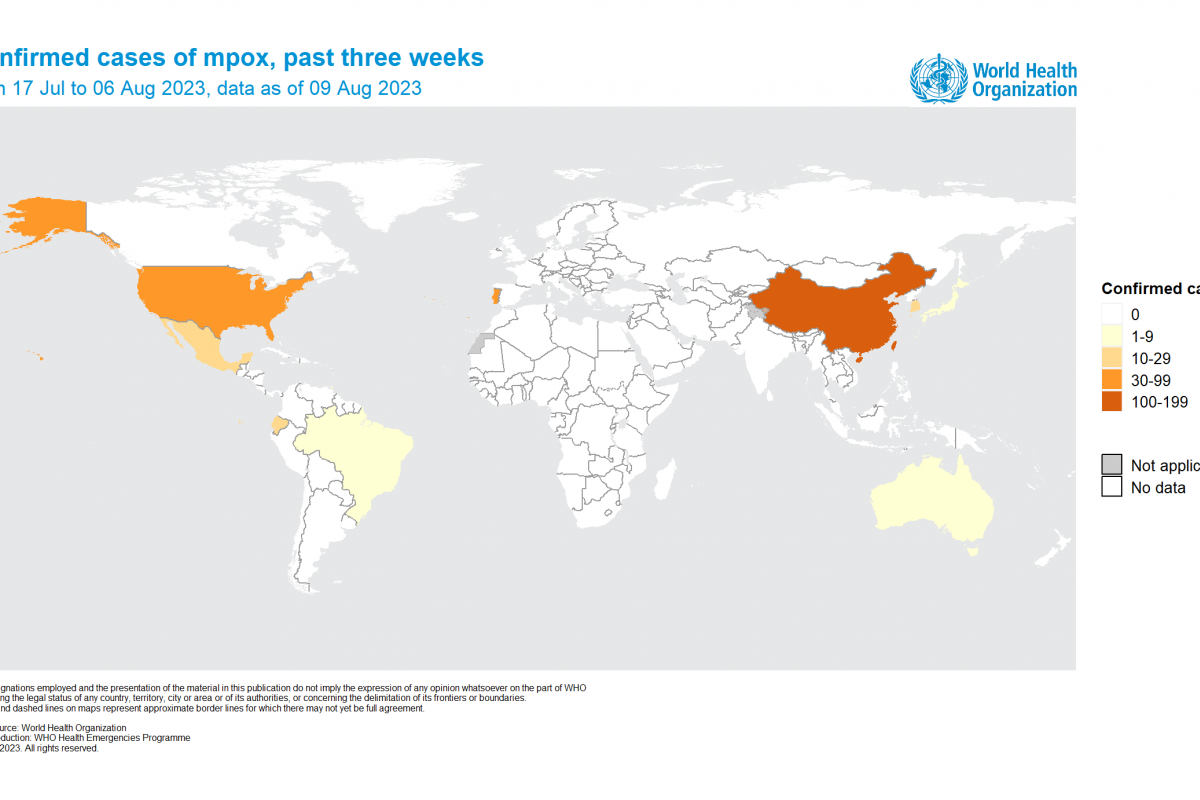
Vaxxinity, Inc. today announced The Lancet's eBioMedicine published results of Phase 2a clinical trial stating that UB-311 "was safe and well-tolerated," with early clinical data demonstrating a trend for slowing cognitive decline in mild Alzheimer's disease (AD).
In this 78-week, randomized, double-blind, placebo-controlled, parallel-group, multicenter Phase 2a, Vaxxinity-funded study, UB-311 was reported to elicit a robust, rapid, and titrated antibody response to Aβ.
And UB-311 was generally well-tolerated, with no cases of ARIA-E and limited cases of asymptomatic ARIA-H.
"This publication supports the innovative work that we and our collaborators are conducting to advance UB-311 for the potential treatment, and even prevention, of Alzheimer's disease," said Mei Mei Hu, CEO of Vaxxinity, in a press release on August 10, 2023.
"Imagine expanding the addressable patient population of beta-amyloid immunotherapies by multiple orders of magnitude, potentially over 1,000x, and delivering life-changing medicine at a fraction of the cost. That is our vision for UB-311 and the potential power of active immunotherapies."
AD is the most common form of dementia, is a progressive neurodegenerative disorder that slowly destroys memory and cognitive skills and eventually the ability to carry out simple tasks.
UB-311 is a synthetic, peptide-based active immunotherapy that targets toxic beta-amyloid (Aβ) oligomers and fibrils and oligomers.
Two passive immunotherapies – monoclonal antibodies targeting Aβ – have recently been authorized by the U.S. FDA, validating Aβ as a target for disease-modifying immunotherapies of AD.
However, these passive immunotherapies have been associated with amyloid-related imaging abnormalities (ARIA), which can present as vasogenic edema or sulcal effusion (ARIA-E) or as hemosiderin deposits such as micro hemorrhages and superficial siderosis (ARIA-H).
Although the trial was not powered to make conclusions about efficacy, secondary efficacy outcomes on cognitive, functional, behavioral, and global assessments such as ADAS-Cog, MMSE, ADCS-ADL, and CDR-SB were evaluated.
Trends of slowing disease progression were observed across key cognitive and functional measures for UB-311-treated versus placebo-treated participants over 78 weeks of observation, including a 48% slowing of decline on CDR-SB in the UB-311 quarterly boosting group.
Furthermore, the U.S. FDA-licensed mAbs require IV infusions every two weeks and are priced at $26,500 annually, not including the cost of administering them or monitoring for ARIA.
In contrast, UB-311 has the potential to offer multiple competitive advantages, including lower rates of ARIA-E, improved convenience through less frequent dosing and ease of administration through intramuscular injection, and overall improved accessibility and cost-effectiveness for patients and health systems.
As of August 10, 2023, the U.S. FDA had not approved an Alzheimer's vaccine candidate.