Mpox Vaccine Receives Recommendation for Routine Use in Adults at Risk

Bavarian Nordic A/S today announced the U.S. Centers for Disease Control and Prevention’s (CDC) Advisory Committee on Immunization Practices (ACIP) voted in favor of recommending the routine use of JYNNEOS® (MVA-BN®), the Company’s U.S. FDA-approved mpox / smallpox vaccine.
Specifically, the ACIP voted to recommend that individuals 18 years and older with certain risk factors receive the two-dose JYNNEOS regimen, a live, nonreplicating vaccine.
Previously, the ACIP had recommended JYNNEOS for individuals at risk during a mpox outbreak.
This represents the second national recommendation for Bavarian Nordic’s mpox vaccine in adult risk groups following a similar endorsement by the Standing Committee for Vaccination in Germany in 2022.
More recently, the European AIDS Clinical Society also recommended using the vaccine for adults infected with HIV or on pre-exposure prophylaxis treatment, which may support additional national recommendations for future vaccine use.
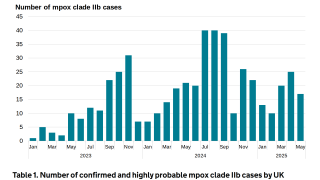
“Since the outbreak of mpox last year (May 2022), Bavarian Nordic has supplied millions of doses of our vaccine to more than 70 countries worldwide,” said Paul Chaplin, President and CEO of Bavarian Nordic, in a press release on October 25, 2023.
The CDC estimates that 2 million U.S. individuals are eligible for a vaccination against mpox under this recommendation.
To date, approximately 23% of this group has received the recommended two doses of JYNNEOS, leaving a significant number of people vulnerable to infection with mpox.
The ACIP reported in October 2023 JYNNEOS Vaccine Effectiveness against mpox ranges from 36% to 75% for 1-dose vaccination and 66% to 89% for 2-dose vaccination.
Pending approval of the updated recommendations, Bavarian Nordic is targeting a commercial launch of JYNNEOS in the U.S. in the first half of 2024.
Our Trust Standards: Medical Advisory Committee