Search API
To help control Chikungunya and Dengue infections reported among school-age children, the Ministry of Education in the Democratic Socialist Republic of Sri Lanka recently launched an innovative mosquito reduction program.
On June 12, 2025, Minister of Education Dr. Susil Premajayantha announced that a special mosquito alleviation program would be implemented before the new school term begins
According to the instructions, school principals could face legal action if mosquito breeding sites are found on school premises during inspections.
Each school must develop and implement its programme to eliminate mosquito breeding sites while continuing educational activities without disruption.
The ministry has sent notices to principals of all public and government-approved private schools, heads of religious schools, deans of national colleges of education, and heads of other educational institutions and provincial offices to inform them about the new measures.
The minister shared these insights during a sensitization program held at the Narahenpita District Secretariat, which was attended by all the principals of schools in the Colombo zone, police station officers, and representatives from the Colombo Municipal Council.
Like countries in the western Indian Ocean, Sri Lanka is also experiencing Chikungunya outbreaks in 2025.
Since late 2005, Chikungunya has caused major outbreaks in Southeast Asian countries.
In 2004, the Chikungunya virus East/Central/South Africa genotype spread from Africa to the Indian Ocean islands, resulting in a large epidemic in Southeast Asia. A retrospective review of cases in Southeast Asia found neurological manifestations or shock in 20% of hospitalized children.
As of June 15, 2025, Chikungunya can be prevented with either a U.S. Food and Drug Administration-approved vaccine, which is recommended for international travelers visiting endemic areas.
The latest data published by the UK Health Security Agency (UKHSA) shows an increase in travel-associated enteric fever cases, specifically typhoid and paratyphoid fever, serious but preventable illnesses caused by Salmonella bacteria.
In 2024, there were 702 reported cases in England, Wales, and Northern Ireland, representing an 8% rise from the 645 cases recorded in 2023.
This number marks the highest annual total of cases ever.
In the UK, most cases of enteric fever are contracted abroad, particularly in regions with poor hygiene and sanitation. Previous surveillance has indicated a troubling increase in antibiotic-resistant typhoid strains in Pakistan, which diminishes the effectiveness of commonly used antibiotics and complicates treatment.
As of June 14, 2025, a free typhoid vaccination is available in the UK for some travelers; however, there is currently no vaccine for paratyphoid fever.
The UK's Travel Health Pro website says, 'Ideally, travellers should consult their GP, practice nurse, pharmacist, or travel clinic at least 4 to 6 weeks before their trip for individual advice, travel vaccines, if relevant for their destination.
In the United States, the CDC estimates that 5,700 of cases of typhoid fever occur in the United States each year. Almost all cases occur among international travelers returning from South Asia, primarily Bangladesh, India, and Pakistan.
The CDC says vaccination can help prevent typhoid fever. Two typhoid vaccines are available in the United States: an oral vaccine and an injectable vaccine.
Bavarian Nordic's Vivotif® (Ty21a) is the only oral typhoid vaccine licensed by the U.S. Food and Drug Administration.
Find out if you need a typhoid vaccine at www.cdc.gov/travel. Or visit a local travel vaccine clinic or pharmacy.
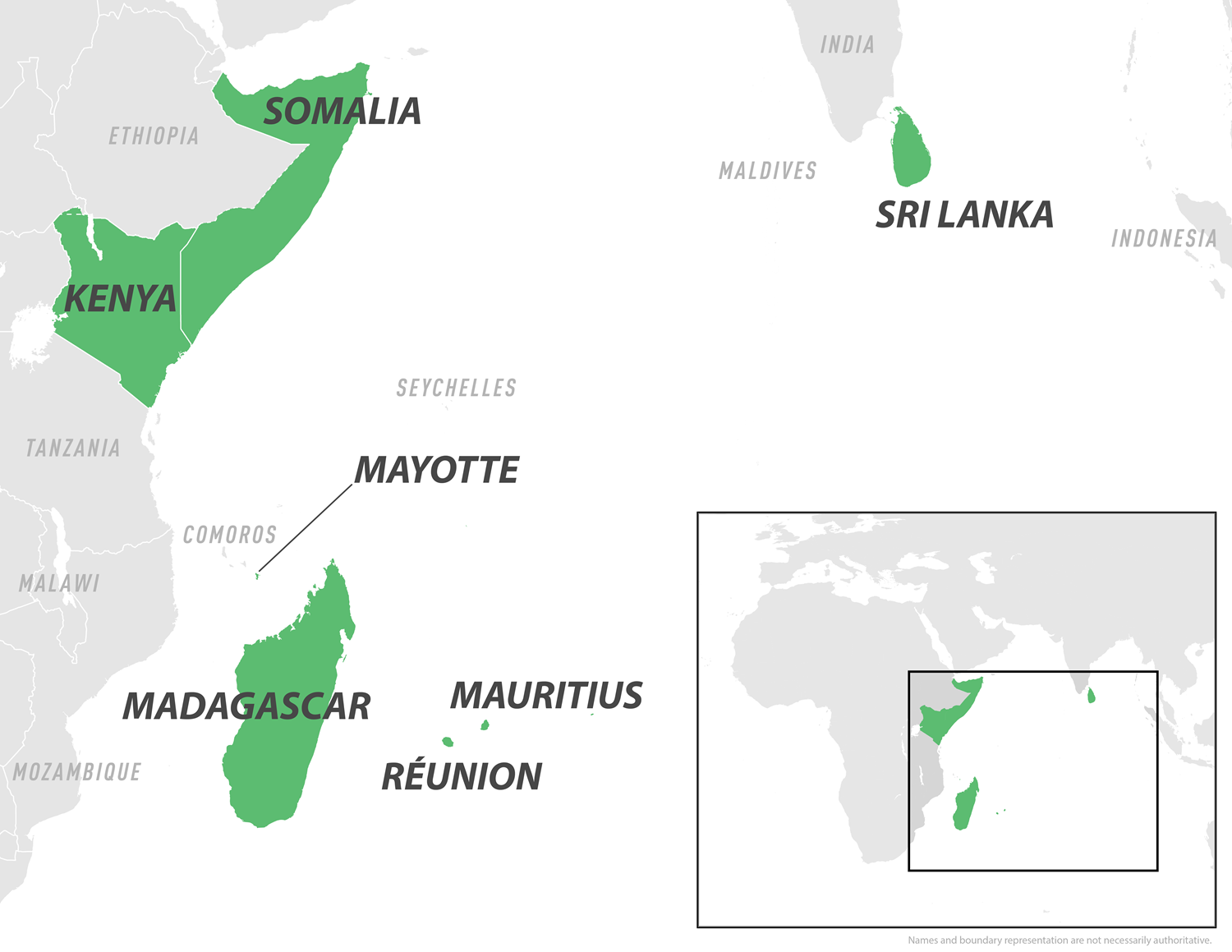
With all eyes focused on the ongoing Chikungunya outbreak in the Indian Ocean, particularly in France's Departments of Mayotte and Réunion, attention has turned to the first indigenous case in metropolitan France in 2025.
According to a press release from Public Health France, this case was reported on June 11, 2025, in the commune of La Crau (Var), located in southern France.
In 2017, two local cases of Chikungunya were confirmed in the Var Department.
During 2024, one local case was reported in Île-de-France (Paris).
An indigenous (local) case is when a person contracted the disease on national territory and did not travel to a contaminated area in the 15 days preceding the onset of symptoms.
As a reminder, the Chikungunya virus, similar to Dengue and Zika viruses, is transmitted from person to person through the bite of a mosquito (tiger mosquito). Dengue and Zika cases have also been confirmed in France.
As of June 14, 2025, the U.S. Centers for Disease Control and Prevention (CDC) maintains a Level 2 Travel Health Advisory for the Indian Ocean region, which includes Réunion, Mayotte, Mauritius, Somalia, Sri Lanka, and the Maldives to the northeast.
Two approved Chikungunya vaccines are commercially available at travel clinics and pharmacies in the United States for the prevention of the disease. The CDC recommends vaccination for international travelers visiting areas with an outbreak.
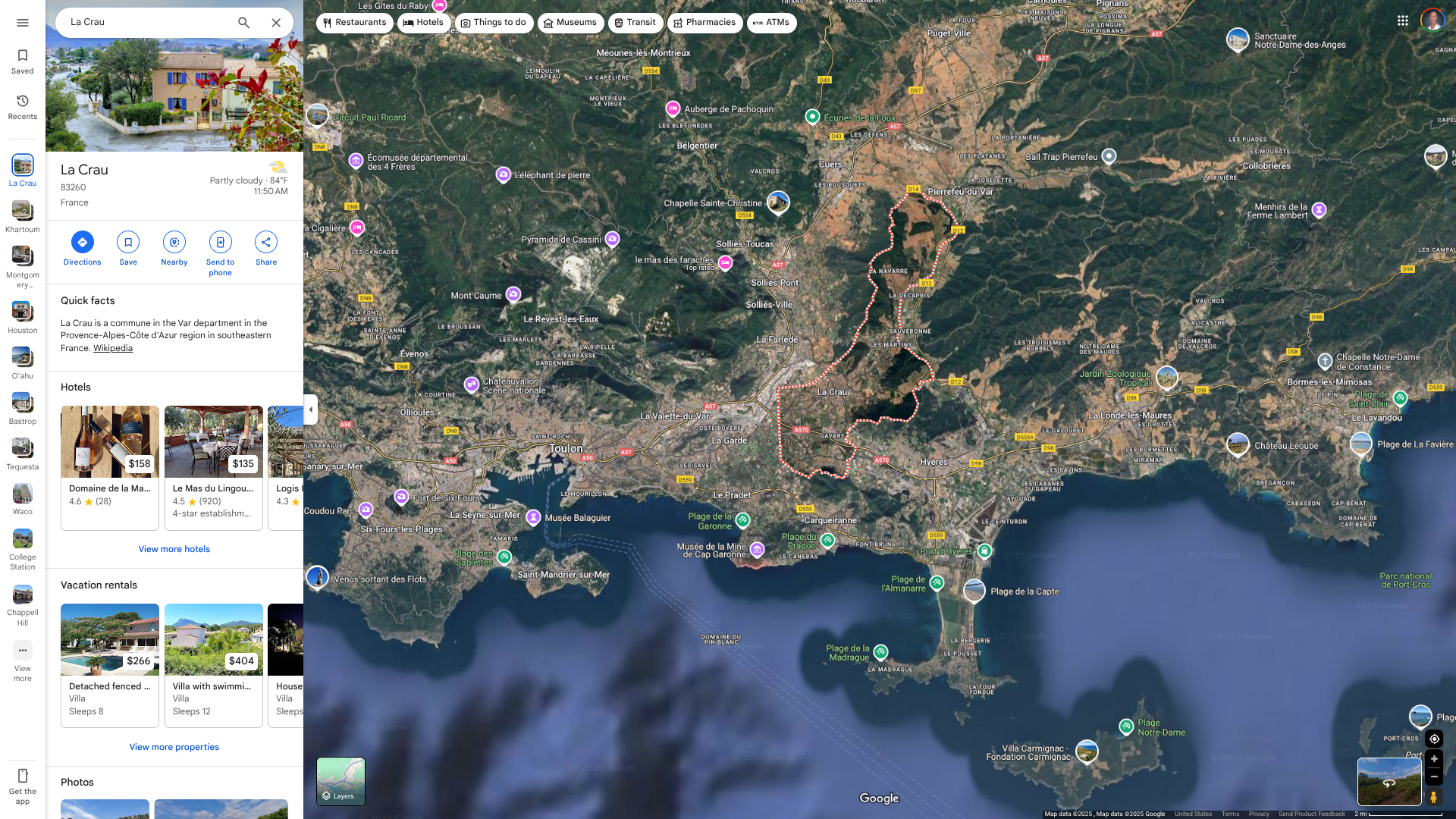
The UK Health Security Agency (UKHSA) has recently published its monthly update on measles cases in England, which shows that outbreaks are continuing, with 109 cases confirmed in April and 86 so far in May 2025.
Since January 2025, the UKHSA has reported 420 laboratory-confirmed cases of measles in England.
As of June 6, 2025, 39% (162 of 420) of these cases have been in London, 14% in the South West, and 14% in the East of England.
Last year, there were 2,911 laboratory-confirmed measles cases in England, the highest number of cases recorded annually since 2012. The 2024 outbreak was initially reported in Birmingham but was soon overtaken by a large outbreak in London.
Dr. Amanda Doyle, National Director for Primary Care and Community Services at NHS England, stated in a media release, "Too many babies and young children are still not protected against the diseases, which are contagious infections that spread very easily and can cause serious health problems."
"MMR jabs are provided free as part of the NHS routine immunisation programme – and I would encourage all parents to act on invites or check vaccination records if they think they may have missed their child's vaccination."
In England, the decline of the uptake of childhood vaccinations, including MMR, in the past decade (below the WHO 95% vaccination target) means thousands of children are left unprotected.
London has the lowest MMR uptake rates compared with other English regions (MMR2 uptake at 5 years is just 73.3% in London compared to the English average of 83.9%).
To warn visitors to England of this health risk, the U.S. CDC included numerous areas of the United Kingdom in its May 28, 2025, Level 1, Global Travel Advisory.
The CDC recommends prospective visitors to England speak with a travel vaccine expert one month before departure to ensure maximum protection can be achieved.
The Florida Department of Health (FDH) published its Arbovirus Surveillance update #23, disclosing that mosquito-borne diseases, such as Dengue, continue to be detected in international travelers and Floridians.
On June 7, 2025, FDH reported 88 cases of Dengue fever among individuals who had traveled internationally, with 40 cases from Cuba.
Last year, Florida reported 1,016 travel-associated dengue cases, primarily among travelers from Brazil, Cuba (567), and Puerto Rico.
So far in 2025, one locally acquired dengue case (DEN-3) has been reported in Miami-Dade County. In 2024, 91 locally acquired dengue cases were reported from ten counties, with Miami-Dade leading the way with 50 cases.
Since the state of Florida has identified 14 mosquito species as vectors of infectious diseases that can transmit to humans, one country has recently taken action to protect its residents by reducing the population of mosquitoes and their habitats.
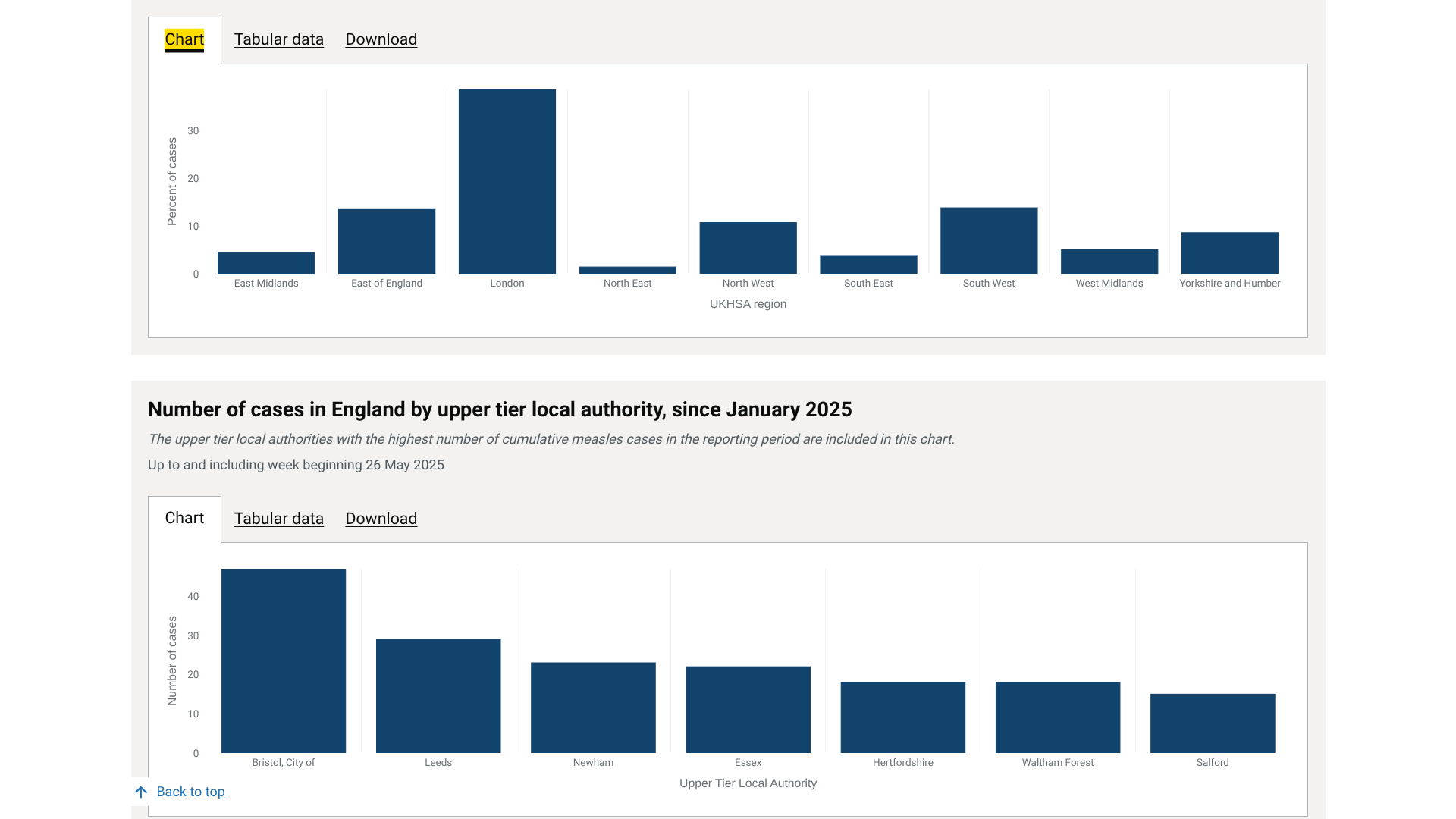
On May 1, 2025, Broward County's Mosquito Control Section announced it began spraying larvicide in areas of Hollywood, Miramar, Pembroke Park, and West Park as a proactive measure to kill the larvae of Aedes aegypti and Aedes albopictus mosquitoes, known to potentially spread diseases such as Dengue, yellow fever, and chikungunya.
According to the County, the larvicide, VectoBac WDG, is not harmful to humans, pets, bees, aquatic habitats, or environmentally sensitive areas. The active ingredient is Bacillus thuringiensis israelensis (Bti, strain AM6552), a naturally occurring, biodegradable bacterial mosquito larvicide.
While this area of Broward County is limited, and local cases of Dengue and malaria have been detected in other Florida counties, this effort is a good step in reducing the number of preventable diseases in 2025.
One of North America's favorite vacation destinations, located just 193 miles east of South Florida, accessible by boat or plane, has been informed by Canada to 'Exercise a high degree of caution.'
On June 3, 2025, Canada affirmed that the Commonwealth of The Bahamas is experiencing a high level of civil unrest, especially in Nassau and Freeport.
The Bahamas is an island country located in the Atlantic Ocean, welcoming approximately 11 million visitors in 2024, many of whom came from Canada and the United States.
Amongst the various warnings, Canada states that the water sports rental industry in the Bahamas is poorly regulated, and reputable operators are limited. Boating accidents occur frequently, particularly on jet skis.
Canada offers the following advice to vacationers: Only rent water sports equipment from reputable, registered operators. Ensure that proper training is provided before using the equipment, and verify that beach and aquatic equipment is safe and in good condition.
The U.S. State Department has also issued a Level 2: Exercise Increased Caution travel advisory for The Bahamas.
Similar to Canada, the U.S. Embassy in Nassau issued a Security Alert on April 9, 2025, advising U.S. citizens of the risks associated with renting jet skis in The Bahamas.
The Embassy wrote, Nassau jet ski operators have raped and sexually assaulted two U.S. citizen women in 2025, and three rapes were reported in 2024.
Women victims reported that male jet ski operators picked them up from downtown Nassau and Paradise Island beaches. This includes the area from Junkanoo Beach to Saunders Beach and Cabbage Beach.
The victims said they were raped and assaulted on isolated islands near New Providence.
Furthermore, due to security risks, U.S. government employees working in The Bahamas are prohibited from renting and using jet skis on New Providence and the Paradise Islands.
From a health perspective, mosquito-transmitted disease reporting from The Bahamas appears lacking.
While the entire Region of the Americas, including the Caribbean countries, set records for Dengue virus infections in 2024, The Bahamas didn't report any cases. Nor has it reported any in 2025.
Which leads to questions about the number of Chikungunya, Malaria, and Zika infections vacationers experience following a Bahamas vacation.
However, the U.S. CDC did include The Bahamas in a recent measles outbreak alert.
Therefore, when scheduling a quick trip to enjoy the warm waters of the Caribbean in June 2025, vacationers should heed the advice from these government agencies and exercise caution.

As thousands of French vacationers plan their summer holiday to visit islands in the Indian Ocean, a mosquito-transmitted disease has become a significant health nuisance in 2025.
To notify international travelers, the U.S. Centers for Disease Control and Prevention (CDC) recently reissued a Level 2 Travel Advisory regarding chikungunya outbreaks in several countries in the region, including Kenya, Madagascar, Mauritius, Mayotte, Réunion, Somalia, and Sri Lanka.
Numerious cases of Chikungunya, both imported and local, have been detected in these vacation destinations.
As of June 10, 2025, the CDC writes, 'Most people infected with the chikungunya virus develop some symptoms. You can protect yourself from disease by preventing mosquito bites, which includes using insect repellent, wearing long-sleeved shirts and pants, and staying in places with air conditioning or that have screens on the windows and doors.
Additionally, vaccination is recommended for travelers who are visiting an area with a chikungunya outbreak. Two chikungunya vaccines are approved for use in the United States, which are commercially available at clinics and pharmacies.
The World Health Organization (WHO) today announced that the Republic of Sudan's Khartoum State is facing an infrastructure breakdown and severe constraints on access to healthcare.
Since May 2025, there has been a rapid increase in cholera cases in Khartoum State, with over 16,000 cumulative cases and 239 deaths reported.
Effective as of June 10, 2025, a 10-day reactive oral cholera vaccination (OCV) campaign was launched in five localities to protect more than 2.6 million people aged 1 year and above from cholera infection, interrupt transmission, and help contain the ongoing cholera outbreak.
WHO Representative in Sudan, Dr Shible Sahbani, commented in a press release, "The vaccines will help stop cholera in its tracks as we strengthen other response interventions."
Khartoum is the capital city of Sudan, with over 7 million residents, located adjacent to the Nile rivers.
The WHO's Global Cholera and Acute Watery Diarrhoea Dashboard was updated in June 2025, indicating that over 224,000 cholera cases and 2,800 related fatalities were reported from 26 countries this year.
Cholera is a vaccine-preventable disease caused by consuming food or water contaminated with the bacterium Vibrio cholerae. It poses a global threat to public health.
As of June 12, 2025, OCV remains in limited supply worldwide, except for the United States.