Search API
While the Zika virus (ZIKV) has faded mainly from global headlines since the major outbreaks of 2015-2016, low-level transmission of this mosquito-borne and sexually transmitted virus continues in parts of Mexico, according to recent data from health authorities.
As of January 11, 2026, the Pan American Health Organization (PAHO) and Mexico's National Epidemiological Surveillance System report that Mexico confirmed 30 autochthonous (locally acquired) Zika cases in 2024, with only four additional cases reported in 2025.
The three Mexican states with the highest number of ZIKV-positive cases during that outbreak were Veracruz, Nuevo León, and Yucatán. These areas, particularly in coastal and lowland regions, remain at-risk for millions of visitors each year due to the presence of Aedes mosquitoes.
A study published in October 2025 analyzed data from the 2016-2018 outbreak period in Mexico, reviewing 13,259 suspected ZIKV cases tested by RT-qPCR from August 2016 to January 2018. Of these cases, 10.7% (1,419) tested positive for Zika virus disease (ZVD).
Notably, 25% (3,313) of the ZIKV-positive cases were in pregnant women, highlighting the ongoing risk to maternal and fetal health during periods of higher virus circulation.
The PAHO and the U.S. Centers for Disease Control and Prevention (CDC) say ZIKV infection during pregnancy can lead to serious complications, such as congenital Zika syndrome, which includes microcephaly and other congenital disabilities.
The CDC classifies Mexico as having current or past Zika transmission and ZVD, which indicates a potential risk.
However, as of January 11, 2026, no CDC Travel Health Notice for Zika is in effect for Mexico. The CDC has shifted away from blanket recommendations against travel for pregnant women to Mexico but continues to advise caution.
These health agencies advise travelers, especially pregnant women or those planning pregnancy, to take strict measures to prevent mosquito bites when visiting at-risk areas such as Mexico.
Although there is currently no Zika vaccine available, clinical research continues.
Recent data from PAHO dashboards indicate that sporadic Zika confirmations have been reported in Brazil, Bolivia, Colombia, Costa Rica, El Salvador, Guatemala, and Peru.

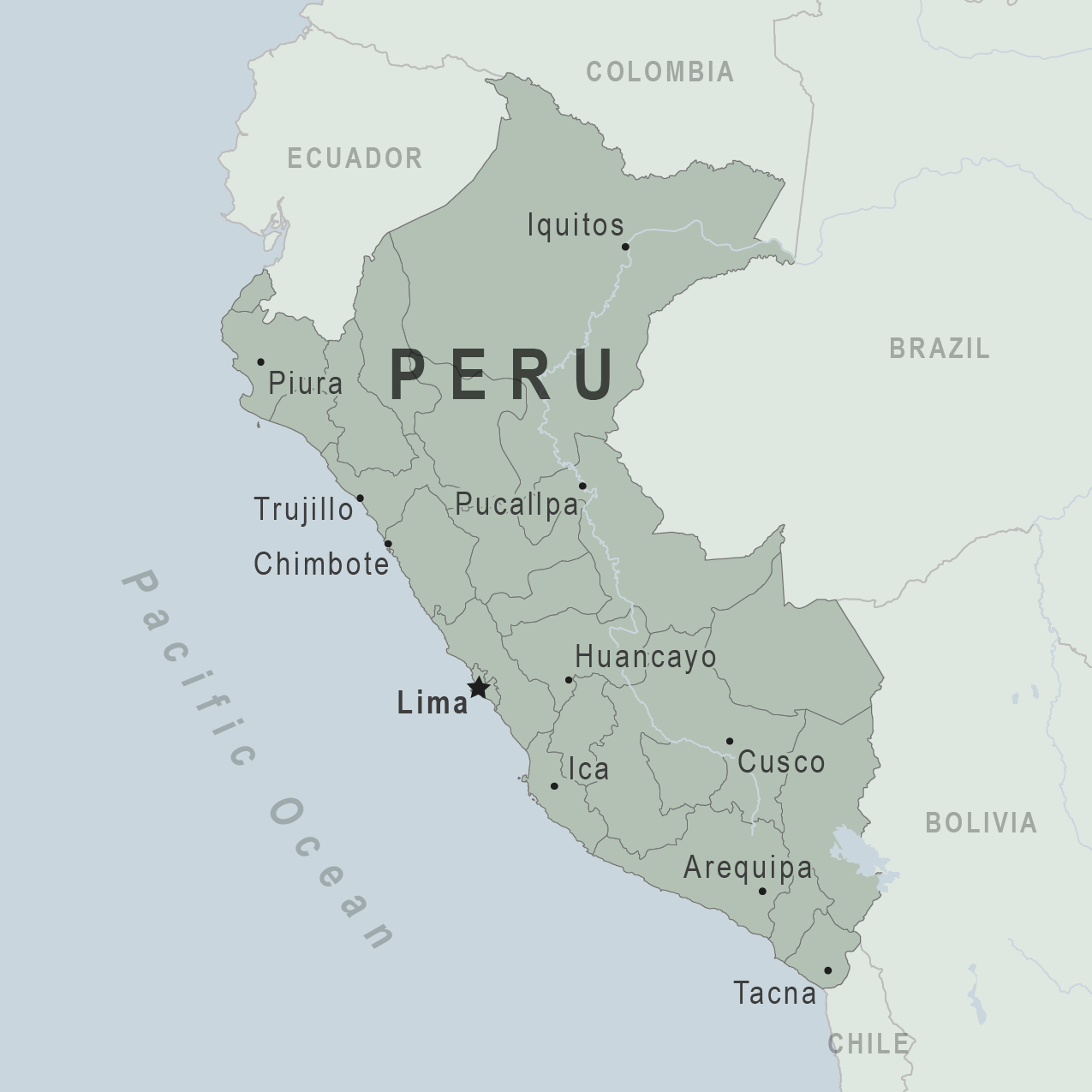
Peru's Ministry of Health (MINSA) today announced that it is urging both domestic and international tourists to prioritize yellow fever prevention when planning trips to the country's rural and jungle areas, which are known to be endemic for the mosquito-transmitted disease.
Given the ongoing risk of transmission, MINSA stated in a press release on January 10, 2026, that the most effective protection against yellow fever is vaccination.
During the first half of 2025, Peru reported 35 confirmed human cases of yellow fever, including 12 deaths (case fatality rate of approximately 34%).
The regions with the highest risk include Amazonas, San Martín, Junín, Ayacucho, Cusco (rural areas only), Loreto, Madre de Dios, Huánuco, Pasco, Puno, and Ucayali. Urban areas, such as the city of Cusco, Machu Picchu, and the Inca Trail, are not included in these warnings.
In Peru, MINSA recommends that travelers receive the vaccine at least 10 days before visiting at-risk areas.
The yellow fever vaccine (Stamaril, YF-Vax) is free, safe, and provides lifelong protection with just a single dose. It is recommended for individuals aged 15 months to 59 years.
In 2025, between 3 and 5 million people visited Peru, with over 1 million traveling to Machu Picchu.
While Peru does not require proof of yellow fever vaccination for entry in all cases, visitors should consult a healthcare provider to confirm their eligibility for immunization.
Additionally, travelers may need to present an International Certificate of Vaccination or Prophylaxis, commonly referred to as the "yellow card," as proof of vaccination at some entry points.
When departing from the United States for Peru, the Centers for Disease Control and Prevention recommends the YF-Vax vaccine for all travelers ≥9 months old traveling to areas below 7,550 ft elevation in at-risk regions. This vaccine is commercially offered at travel clinics in 2026.
The Pan American Health Organization/World Health Organization (PAHO/WHO) issued an alert today regarding the possibility of earlier or more intense respiratory virus activity during the 2025-26 season compared to previous years.
As of January 9, 2026, PAHO/WHO recommends that Member States remain vigilant to adjust their health service preparedness and organizational plans in anticipation of possible concurrent outbreaks of seasonal influenza and respiratory syncytial virus.
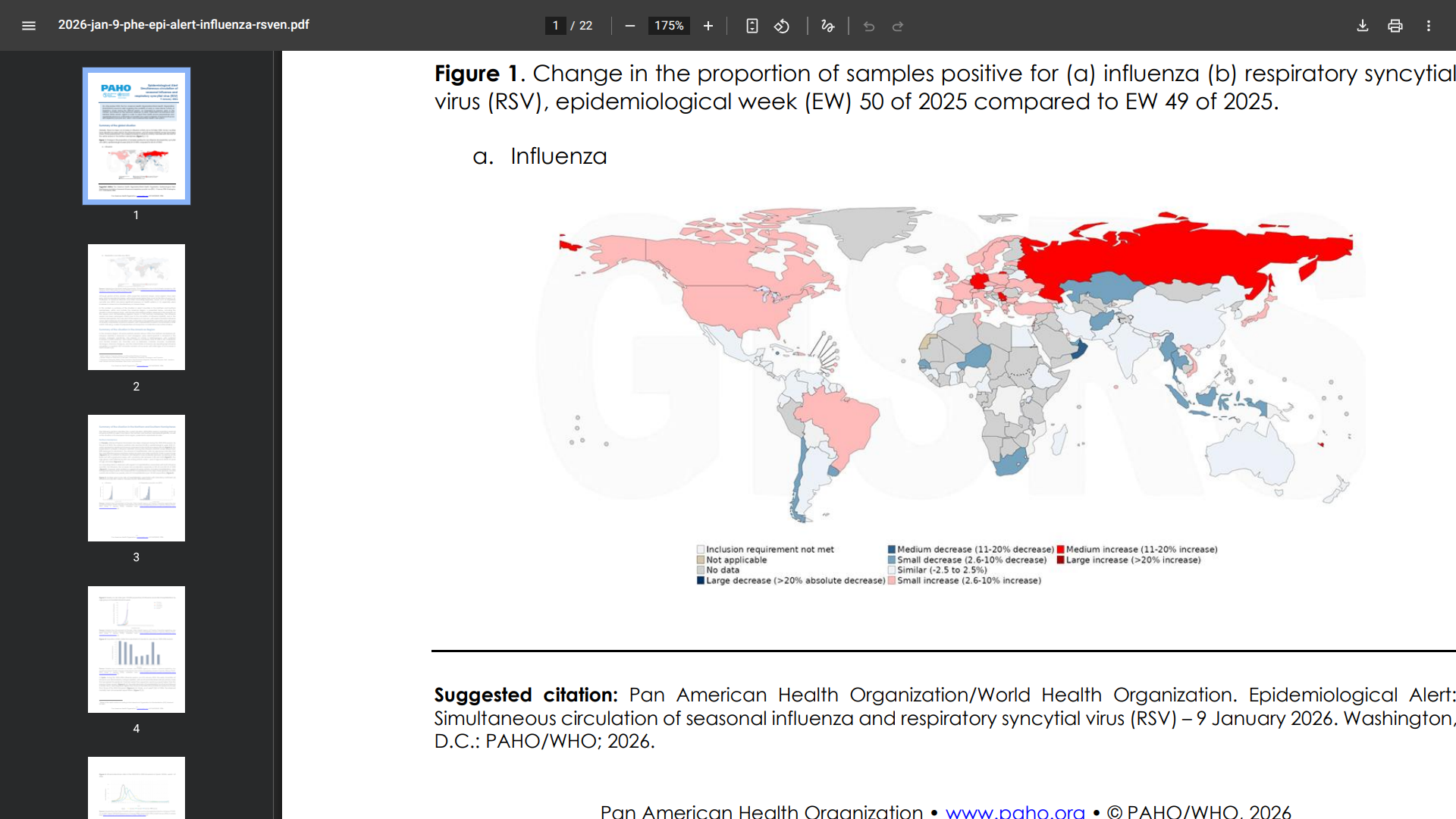
Globally, influenza activity has increased since late 2025.
Some countries are reporting an early onset of the influenza season, with influenza A(H3N2) viruses being the most predominant, while A(H1N1)pdm09 is the primary strain in the Andean subregion.
In the Americas Region, the influenza positivity rate remains above 10% in the Northern Hemisphere. The intensity of activity varies, with positivity levels approaching 20% in the Caribbean and Central America.
Countries such as Barbados, Canada, Ecuador, Guatemala, Nicaragua, Panama, Paraguay, and the United States are reporting high levels of influenza circulation.
In contrast, RSV circulation remains low.
Regarding this season's flu shots' effectiveness, many are questioning whether the updated vaccines provide adequate protection against the disease.
A study from the University of Pennsylvania released on January 6, 2026, found that while subclade K viruses are antigenically advanced compared to the vaccine strain, the 2025-2026 vaccine still elicited robust antibody responses in many participants.
Furthermore, the PAHO/WHO recommends flu shots for international travelers before visiting outbreak areas.
Access to influenza vaccines remains robust in the USA, with most clinics and pharmacies offering services as of January 10, 2026.
Camel tours remain a bucket-list experience for international tourists visiting the Middle East and North Africa, with global participation in 2025 estimated in the hundreds of thousands.
Popular destinations such as Morocco's Sahara Desert, Egypt's Giza Plateau, and the Kingdom of Saudi Arabia (KSA) desert landscapes continue to draw visitors eager for camel rides in the desert, often without reported incidents.
However, health officials are urging travelers to exercise caution due to recent updates regarding the Middle East respiratory syndrome coronavirus (MERS-CoV).
On January 9, 2026, the European Centre for Disease Prevention and Control (ECDC) and the World Health Organization (WHO) reported new cases of MERS-CoV, highlighting the ongoing zoonotic risk posed by dromedary camels.
According to the ECDC's update, there have been a total of 19 MERS-CoV cases reported worldwide since the beginning of 2025, including four fatalities.
Notably, 17 of these cases, which also include the four deaths, originated in the KSA and were spread across regions such as Riyadh, Taif, and Najran.
The ECDC's situation overview indicates that since MERS-CoV was first identified in 2012, authorities have recorded approximately 2,635 to 2,647 laboratory-confirmed cases globally, with 959 to 964 deaths, resulting in a case fatality ratio of around 37%.
The vast majority of these cases (over 84%) have occurred in the KSA.
MERS-CoV is a zoonotic virus primarily transmitted through direct or indirect contact with infected dromedary camels or the consumption of raw or undercooked camel products, including milk. While human-to-human transmission mainly occurs in close-contact settings like hospitals, sustained community spread remains rare.
The ECDC assesses the overall global and regional risk as moderate and notes that the recent cases prompt no changes.
However, travelers—especially those with underlying conditions such as diabetes, chronic lung disease, or weakened immune systems—are advised to avoid close contact with camels, whether at farms, markets, or during tourist rides. It is also recommended to refrain from consuming raw camel milk, urine, or undercooked meat.
Local health authorities are closely monitoring the situation and, at this time, do not recommend any travel restrictions or special entry screening. Travelers planning desert experiences should consult updated health advisories before their departure, according to the ECDC.
The World Health Organization (WHO), in its latest fact sheet (updated December 11, 2025), states that "No vaccine or specific treatment is currently available" for MERS-CoV. However, several candidates are in development."
Multiple promising platforms have advanced to early clinical stages, including viral vector-based candidates like ChAdOx1 MERS and MVA-MERS-CoV.
However, challenges persist in MERS vaccine clinical trials, as low case numbers make large-scale Phase 3 efficacy trials difficult.

As the global effort to eradicate polio intensifies in 2026, needle-free technology is becoming increasingly popular in the Middle East and surrounding regions.
In a notable advancement for polio eradication in one of the world's last endemic countries, Afghanistan's polio program began using needle-free technology for the first time in late 2025.
During a multi-phase fractional inactivated polio vaccine (fIPV) campaign in Afghanistan's eastern region, the initiative targeted over 1.2 million children across 50 districts in four provinces.
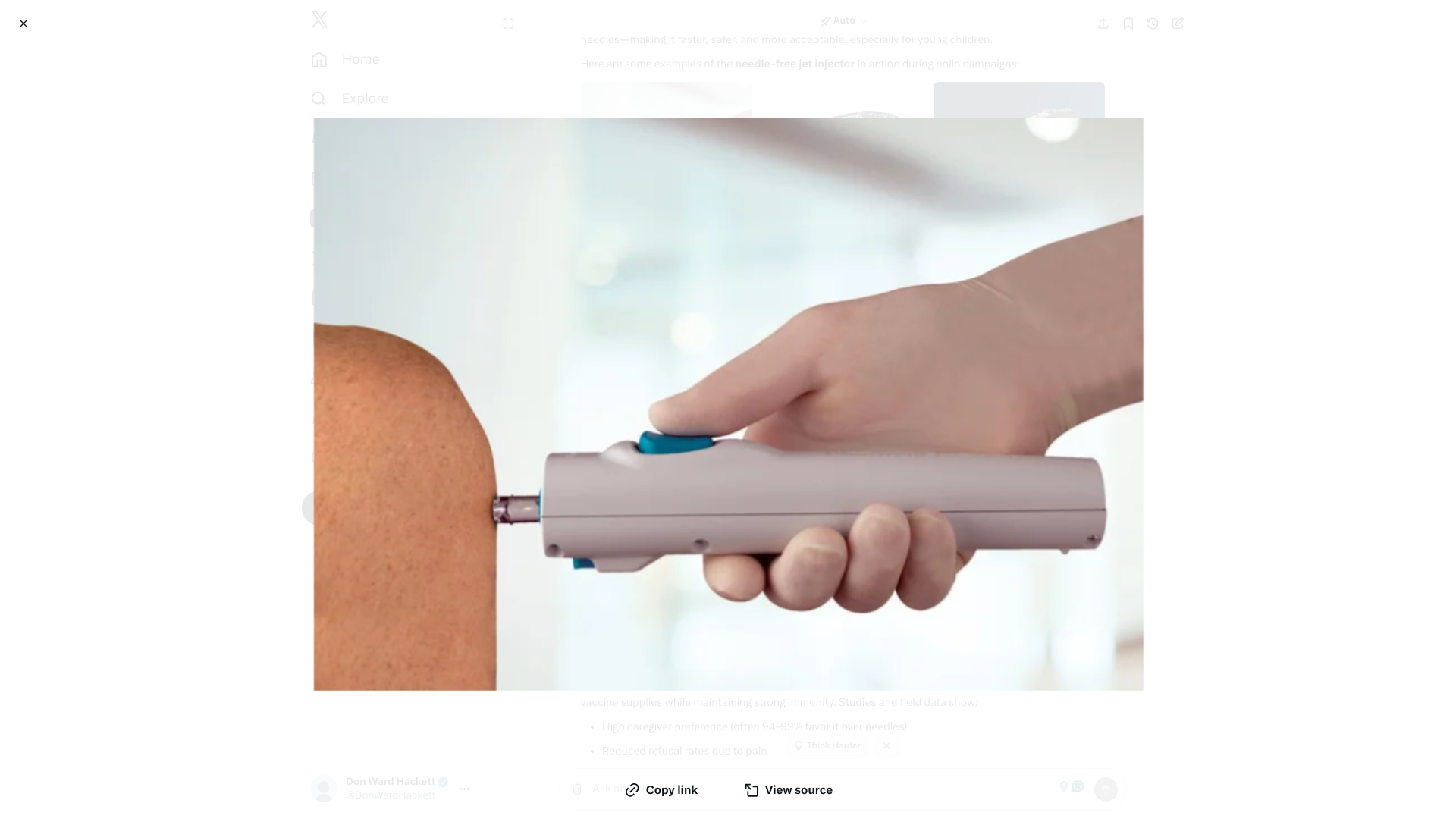
The new intradermal jet injectors—also known as needle-free devices—were combined with the oral polio vaccine (OPV) to enhance immunity against wild poliovirus type 1 (WPV1).
This innovative approach provided children with both a fractional IPV dose via jet injector and the oral vaccine, offering dual layers of protection against polio.
The PharmaJet Tropis® ID system, a WHO-prequalified needle-free injector, delivered tiny, precise streams of the fractional-dose IPV through the skin in a split second. This painless method eliminated the need for needles, reducing fear among children and caregivers while improving acceptance in remote and high-risk communities.
Speaking about the campaign to the Polio Eradication Initiative on January 1, 2026, polio vaccinator Mohammad Ibrahim expressed his excitement in a press release: "It is much easier than needle injections. I'm confident to go into the field and vaccinate children."
Vaccinator Zahir Islam noted that when communities learned the device was needle-free and painless, they became eager to participate. Community and religious leaders endorsed the method as safer and faster, helping to rebuild trust in vaccination efforts. This campaign not only provided essential protection but also strengthened community engagement in a region critical to global polio eradication.
As of late 2025, Afghanistan had reported around 9 WPV1 cases, confirmed in provinces like Uruzgan, Helmand, Badghis, Nuristan, Paktika, and Farah.
This new, large-scale introduction builds on the successful use of the Tropis system in countries like Egypt.
In November 2025, PharmaJet announced the incorporation of Tropis into immunization programs, supporting Egypt's "1000 Golden Days" initiative.
These polio eradication programs are essential, as a WHO Committee unanimously concluded in 2025, the risk of the international spread of poliovirus continues to constitute a Public Health Emergency of International Concern.
In the long term, the manufacturing of Tropis and other needle-free product development initiatives may expand the benefits of vaccination against other infectious diseases and enhance pandemic preparedness.
With over 40 million people living with HIV and more than 1 million new infections annually, the need for an effective preventive vaccine remains urgent.
On December 15, 2025, the first doses of experimental HIV vaccine antigens were administered at the Perinatal HIV Research Unit in Soweto, South Africa, marking the launch of the IAVI G004 Phase 1 clinical trial.
This trial advances the IAVI/Scripps Research strategy to train the immune system to produce broadly neutralizing antibodies (bnAbs) capable of protecting against diverse HIV strains. IAVI G004 builds on positive proof-of-concept data from earlier studies (G001, G002, and G003). It will evaluate three mRNA-based immunogens developed by IAVI and Scripps Research: eOD-GT8 60mer, Core-g28v2 60mer, and N332-GT5 gp151, delivered using Moderna's mRNA platform.
"It is very exciting to see the launch of the IAVI G004 clinical trial," said Mark Feinberg, M.D., Ph.D., President and CEO of IAVI, in a press release on January 6, 2026.
"We believe this is the most promising path toward an effective HIV vaccine."
Funding for the trial comes from the Bill & Melinda Gates Foundation, with materials manufactured by Moderna. The government of the Netherlands has provided additional support.
Currently, no HIV vaccine of any kind has been approved by the U.S. Food and Drug Administration (FDA), but scientists are actively pursuing new approaches. Therapeutic HIV vaccines are only available through a clinical trial.
The FDA says exploring therapeutic HIV vaccines to achieve HIV remission or a functional cure is essential, keeping viral load suppressed without the need for antiretroviral therapy. Ongoing HIV and AIDS clinical trials include studies of new HIV medicines, vaccines to prevent or treat HIV, and medicines to treat infections related to HIV and AIDS.
In 2025, mainland Europe saw a notably high number of locally acquired chikungunya virus cases, primarily in France and Italy. Chikungunya outbreaks in 2025 highlight the growing health risk of mosquito-borne diseases to residents and visitors in Europe.
This represented a significant increase compared to previous years, driven by the presence of the Aedes albopictus mosquito vector and favorable summer conditions.
According to the European Centre for Disease Prevention and Control (ECDC) seasonal surveillance report as of December 31, 2025, a total of 788 cases were reported in France and 384 cases in Italy, all locally acquired.
As of January 8, 2026, no new cases have been reported, and all clusters in both countries are now closed. The ECDC has concluded that until weather conditions change, there is minimal risk until the warmer spring or summer months.
As of early 2026, two chikungunya vaccines have been authorized for use in the European Union: IXCHIQ, a live-attenuated single-dose vaccine approved in June 2024, and VIMKUNYA, a non-live virus-like particle vaccine approved in February 2025. Both vaccines protect against the virus, with VIMKUNYA serving as an alternative for those who may not be suitable candidates for live vaccines.
Vaccine availability varies by member state, and vaccination is generally recommended for travelers to endemic areas in France and Italy.
The ECDC says these vaccines represent a crucial tool in preventing future outbreaks amid the expanding presence of mosquito vectors in Europe.
In the United States, chikungunya vaccines are commercailly offered at travel clinics such as Passport Health.
With nine reported pediatric fatalities related to influenza this season, many are questioning whether the updated vaccines provide adequate disease protection.
A new preprint study from researchers at the University of Pennsylvania (UPenn) offers encouraging insights into the ongoing 2025-2026 influenza season, which has been dominated by a drifted variant of the H3N2 virus known as subclade K.
The study, posted on medRxiv on January 6, 2026, examined blood samples from vaccinated individuals to assess how well the current seasonal flu vaccine triggers antibodies against this emerging strain.
Subclade K, characterized by several key substitutions in the hemagglutinin (HA) protein—including mutations like K2N, S144N, N158D, and others, which emerged late in 2025- was not included in the Northern Hemisphere vaccine formulation.
These UPenn researchers found that while subclade K viruses are antigenically advanced compared to the vaccine strain, the 2025-2026 vaccine still elicited robust antibody responses in many participants.
Before vaccination, only about 11% of individuals had neutralizing antibodies against subclade K.
After receiving the flu shot, this figure rose to around 39%.
Lead researcher Scott Hensley, a professor of microbiology at the UPenn, described the results as "unexpected good news."
The findings suggest that the vaccine is likely to provide meaningful protection against infections caused by subclade K, particularly against severe outcomes such as hospitalization.
This news comes amid a challenging flu season, with subclade K driving the majority of cases across the U.S. and globally.
U.s. Centers for Disease Control and Prevention (CDC) data for Week 52 indicate H3N2 accounts for nearly all influenza activity so far, with subclade K comprising over 90% of characterized H3N2 viruses.
Vaccine experts at the CDC emphasize that vaccination remains the best tool available, even against mismatched strains. Furthermore, there is an ample supply of flu shots offered at clinics and pharmacies in 2026
This preprint has not yet undergone peer review, but preliminary data align with observations from public health agencies monitoring the season.