$3.5 Million Helps Fund Rift Valley Fever Vaccine Candidate
In a significant step toward enhancing global epidemic preparedness, the Serum Institute of India (SII), today announced it has partnered with the University of Oxford under the support of the Coalition for Epidemic Preparedness Innovations (CEPI) to establish an investigational reserve of the ChAdOx1 RVF vaccine candidate against Rift Valley fever (RVF).
This is an essential initiative as no RVF vaccine is currently available.
Confirmed on January 13, 2026, SII will produce up to 100,000 doses of the investigational ChAdOx1 RVF vaccine. An initial 10,000 doses are earmarked for a potential upcoming clinical trial to evaluate the candidate's safety and immunogenicity in outbreak-affected regions. The remaining doses will be maintained as a strategic investigational reserve, ready for rapid deployment in future outbreaks to generate critical clinical data and accelerate evidence for licensure.
ChAdOx1 RVF has completed Phase I trials. Results published from the UK and Uganda trials show it was safe and generated an immune response. A CEPI-supported Phase II trial, separate from the upcoming planned trial, began in Kenya in 2025.
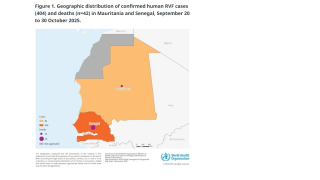
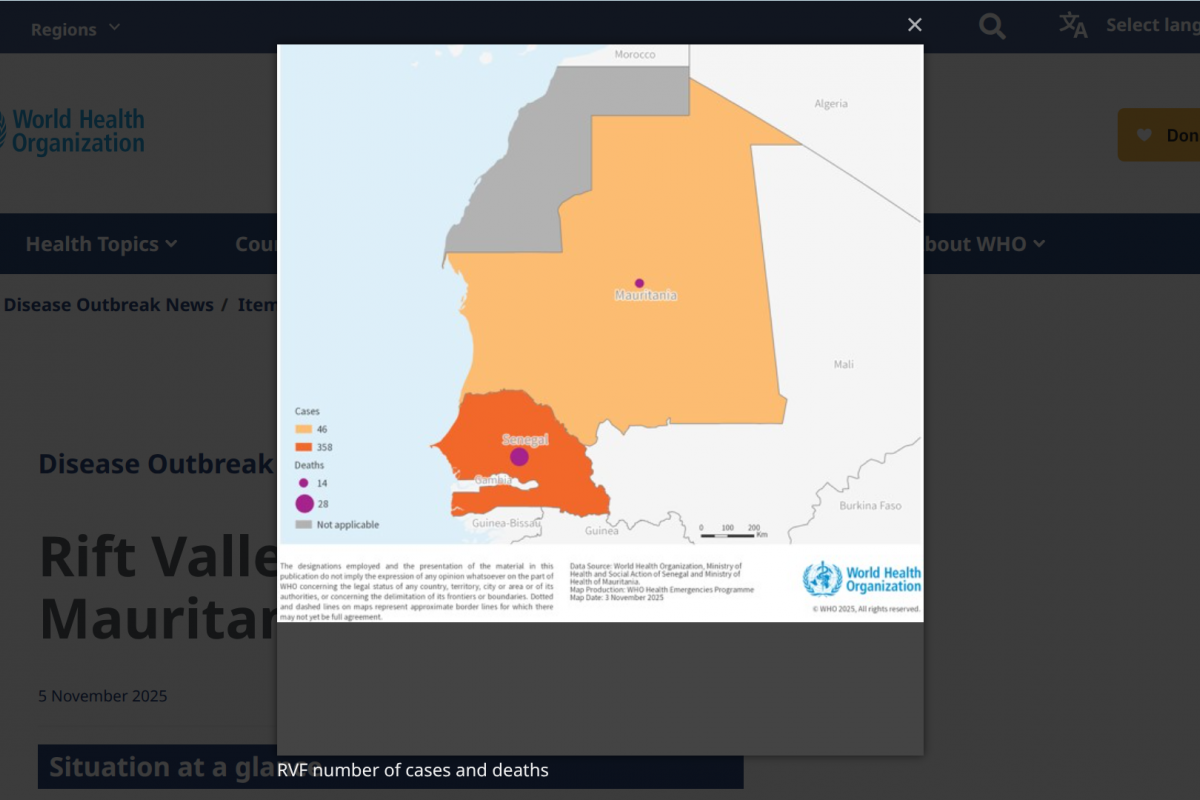
This collaboration arrives as a deadly RVF outbreak continues to impact communities in West Africa, particularly in Sénégal and Mauritania.
According to the latest available World Health Organization (WHO) data, DON584 from 2025, over 600 confirmed human cases, including 47 related deaths, have been reported.
Under the licensing agreement, CEPI is providing up to $3.5 million to support the development, manufacturing, and creation of a reserve for this RVF vaccine candidate.
In a press release, Dr. Umesh Shaligram, Executive Director at SII, added, "Producing these doses at speed is a necessary step to ensure that vaccine candidates like ChAdOx1 RVF can be delivered when and where they're most needed."
Rift Valley fever is a mosquito-borne zoonotic disease that primarily affects livestock but can spill over to humans through contact with infected animals or bites from infected mosquitoes. While most human cases are mild, severe infections can lead to brain inflammation, hemorrhagic fever, and death.
The disease also devastates pastoral communities by causing massive livestock losses, impacting livelihoods and food security in affected regions across Africa and the Middle East.
This collaboration exemplifies proactive "100 Days Mission" principles, positioning the world to respond more effectively to emerging infectious threats like RVF.
Until a vaccine becomes available, the U.S. CDC, WHO, and others recommend that travelers to affected areas follow guidance on mosquito prevention and avoiding contact with potentially infected animals.
Our Trust Standards: Medical Advisory Committee