Search API
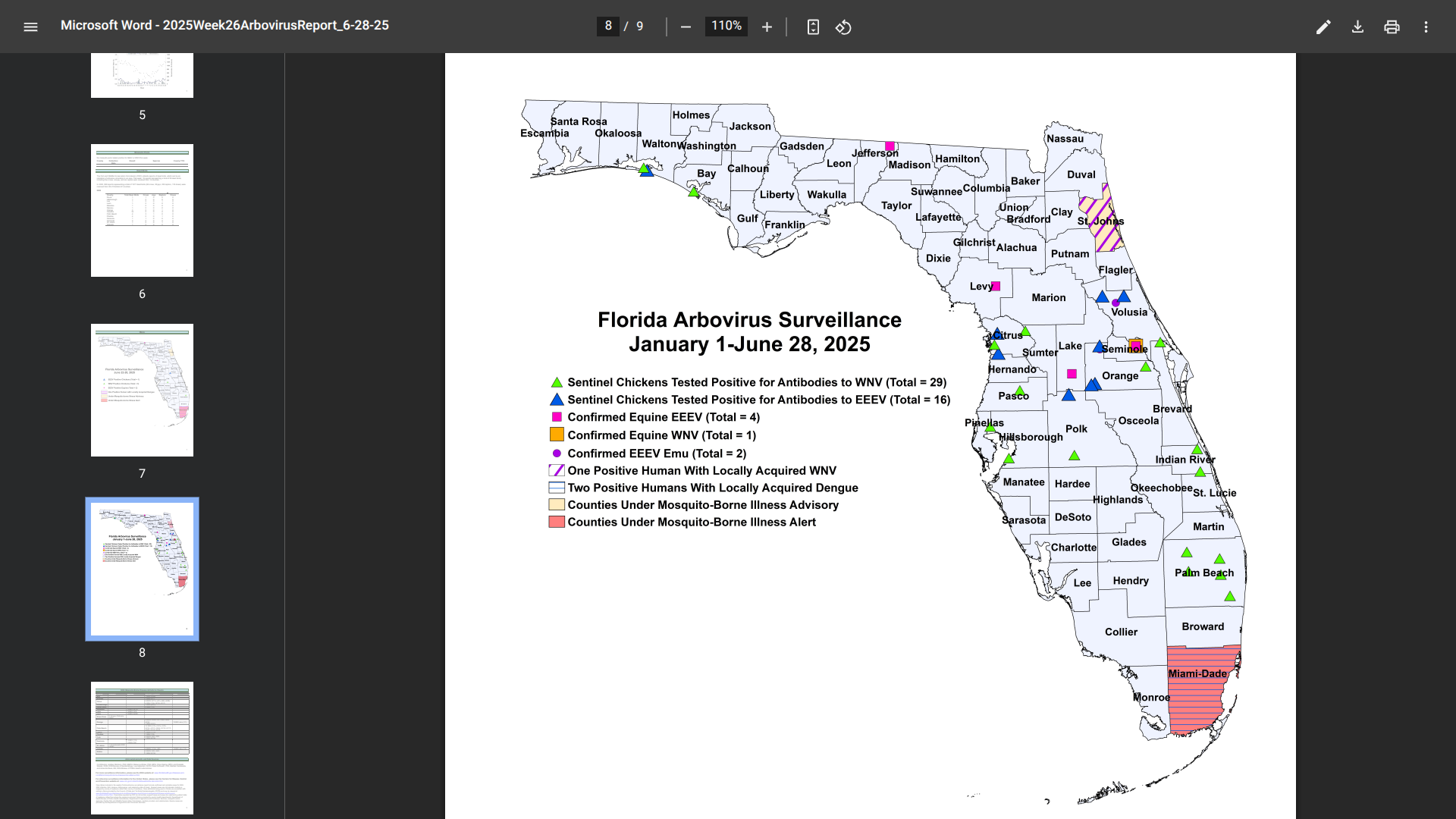
Florida health officials recently reported additional cases of mosquito-transmitted and travel-related chikungunya, dengue, and malaria in 2025.
After a slow start, these serious diseases have been concentrated in southeast Florida, in the great Miami area, which welcomes millions of international visitors each year.
Currently, Miami-Dade County remains under a mosquito-borne illness alert.
As of June 28, 2025, Florida Health's Weekly Arbovirus Report confirmed a second, indigenous case of dengue in Miami-Dade County.
The most recent case has been identified as serotype DENV-4, while the early case was DENV-3.
In 2024, a total of 91 cases of locally acquired dengue have been reported across ten Florida counties.
Additionally, there have been four cases of chikungunya and 20 cases of malaria related to international travelers this year.
While there are no travel advisories issued for Florida's southeast coast, Canada and the UK both advise speaking with a travel vaccine expert before visiting at-risk areas in 2025.
The regional health agency in Grand Est, France, reported its first locally acquired case of chikungunya in Bas-Rhin.
As of July 1, 2025, epidemiological investigations indicated that this locally acquired chikungunya case had frequented the municipalities of Lipsheim and Fegersheim during their infectious period.
Unlike previous local cases, this area is located in France's northeast.
In June 2025, Occitane (Hérault and Gard), Provence-Alpes-Côte d'Azur (Var and Bouches-du-Rhône), Auvergne-Rhône-Alpes (Drôme), and the island of Corse (Corse-du-Sud) reported local chikungunya cases.
And in the southern Indian Ocean, France's departments have reported significant chikungunya outbreaks in 2025.
According to the World Health Organization and the United States, chikungunya is a vaccine-preventable disease, with two vaccines commercially available as of July 2025.
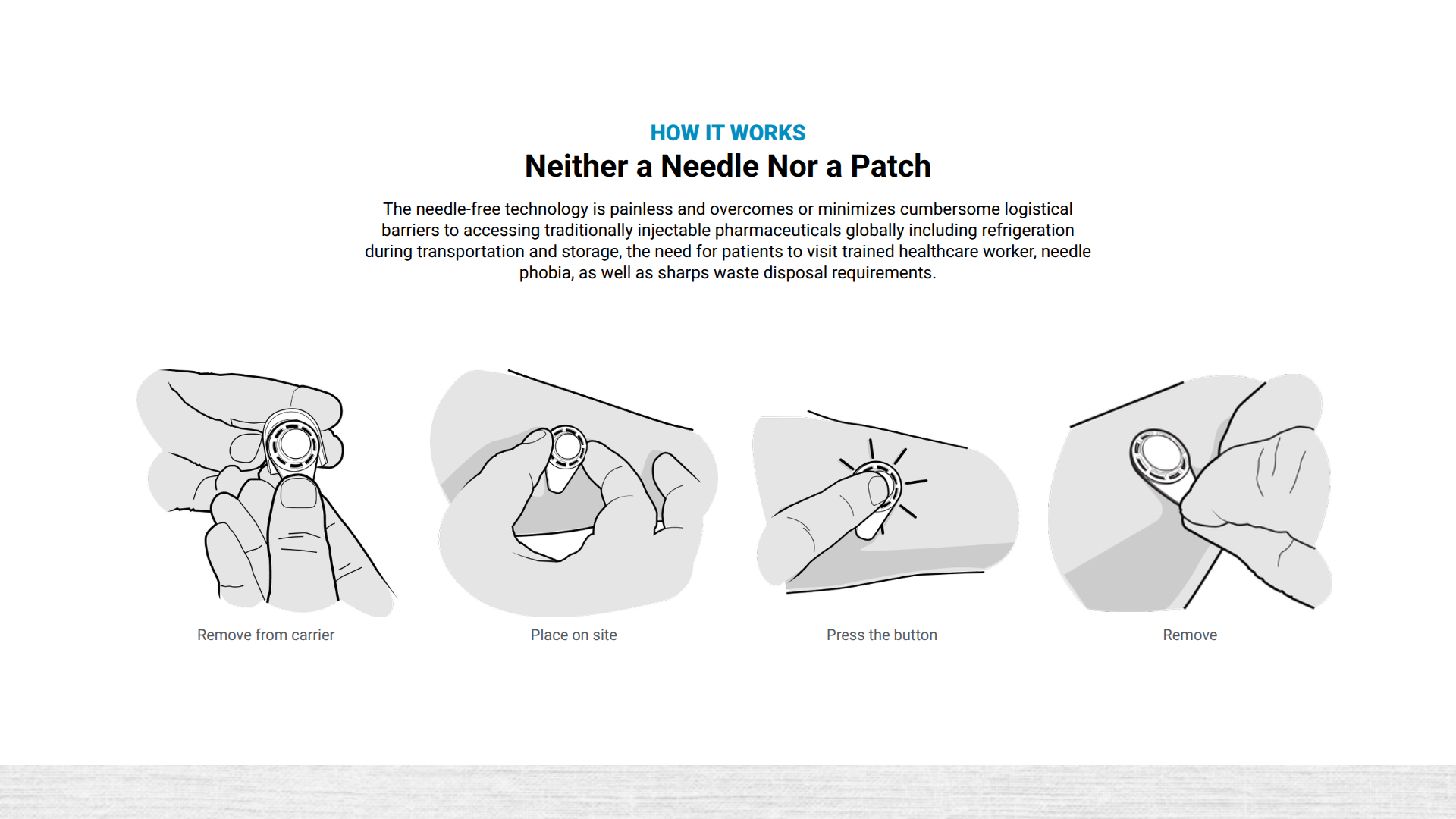
Emory University and Micron Biomedical recently announced the first clinical trial for a novel rotavirus vaccine, CC24.
This vaccine candidate is delivered using dissolvable microarray technology and is currently enrolling participants in the Phase 1 study, which launched in mid-June 2025.
Notably, this trial represents the first clinical evaluation of a drug or vaccine administered via patch or microarray, sponsored by the U.S. Centers for Disease Control and Prevention (CDC).
"CDC recognizes the potential of this groundbreaking clinical trial, which will test the safety of both our novel vaccine, CC24, in adults and the vaccine's delivery with 'patch' technology," says Dr. Demetre Daskalakis, Director of CDC's National Center for Immunization and Respiratory Diseases, in a press release.
"The trial marks significant progress in vaccine technology innovation and is a critical step toward saving more children from rotavirus illness and death."
Rotavirus infection is a leading cause of diarrheal deaths among children, particularly in low and middle-income countries where existing oral vaccines are often less effective. CC24 is a uniquely inactivated rotavirus vaccine developed by the CDC to provide an alternative to the oral administration of rotavirus vaccines.
Micron Biomedical's unique needle-free technology rapidly delivers vaccines and therapeutics via dissolvable microarray compounds that are painlessly pressed into the upper layers of the skin.
As of July 2, 2025, U.S.-FDA-approved rotavirus vaccines are available.
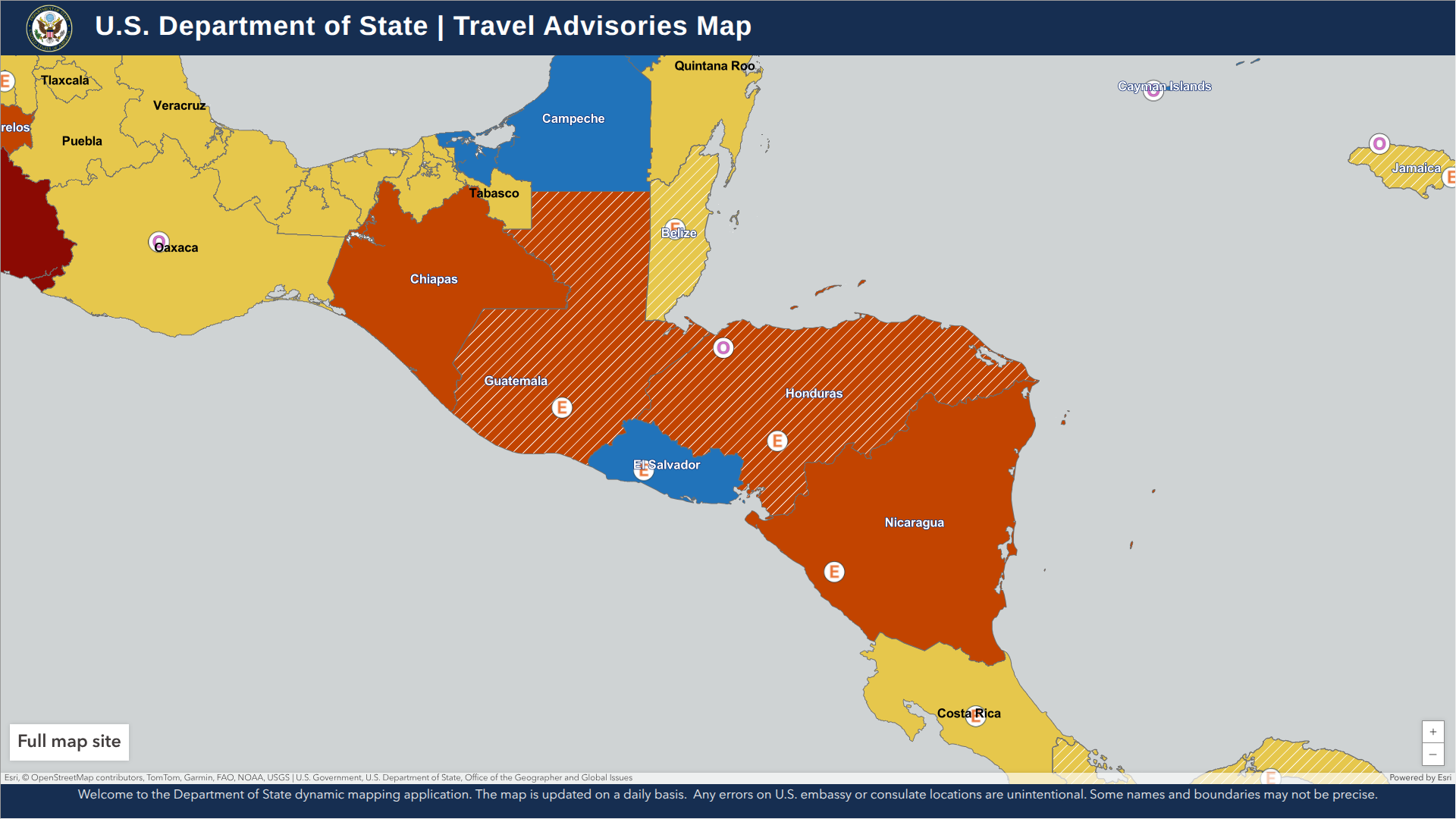
According to the Secretary of State at the Ministry of Foreign Affairs and International Cooperation, Honduran authorities are requiring documentation to show proof of a measles vaccine (MMR) for travelers coming from various countries, including the United States.
As of June 28, 2025, this new requirement applies to all foreign travel to the Republic of Honduras, whether by land, sea, or air.
The U.S. Embassy in Honduras stated that if someone cannot receive the vaccine, they will need to provide proof of a negative measles test result 72 to 80 hours before the flight.
This means that cruise ship passengers visiting this Central American country may need to carry proof of their vaccination.
In addition to measles vaccination, the U.S. CDC recommends being protected against chikungunya, dengue, malaria, typhoid, and Zika when visiting Honduras.
For local assistance during July 2025, the U.S. Embassy Tegucigalpa is available to U.S tourists.
Additionally, the Department of State stated in 2024, "Reconsider travel to Honduras due to civil unrest." The Smart Traveler Enrollment Program is available to travelers interested in receiving digital travel alerts while abroad.
In June 2025, the WHO's Dengue Situation Update #723 confirmed that the Western Pacific Region continues to face a high burden of mosquito-borne arboviral diseases, particularly Dengue.
Within the Philippines, Quezon City (QC) declared a Dengue outbreak in February 2025
The QC government today published updated data regarding the ongoing outbreak.
The QC Epidemiology and Surveillance Division (QCESD) dashboard reported on July 1, 2025, that 5,762 dengue cases were reported in 2025, with QC's District 2 confirming the highest number of cases.
The QCESD stated that most fatal dengue cases (23) involved children and young women.
To alert international travelers visiting QC, a metro area with about 3 million residents located north of Manila, the U.S. Centers for Disease Control and Prevention included the Philippines in its Global Dengue Outbreak Advisory issued in June 2025.
Without a preventive vaccine available in the U.S., the CDC recommends that travelers to risk areas should prevent mosquito bites by using an EPA-registered insect repellent, wearing long-sleeved shirts and long pants when outdoors, and sleeping in an air-conditioned room or one with window screens.

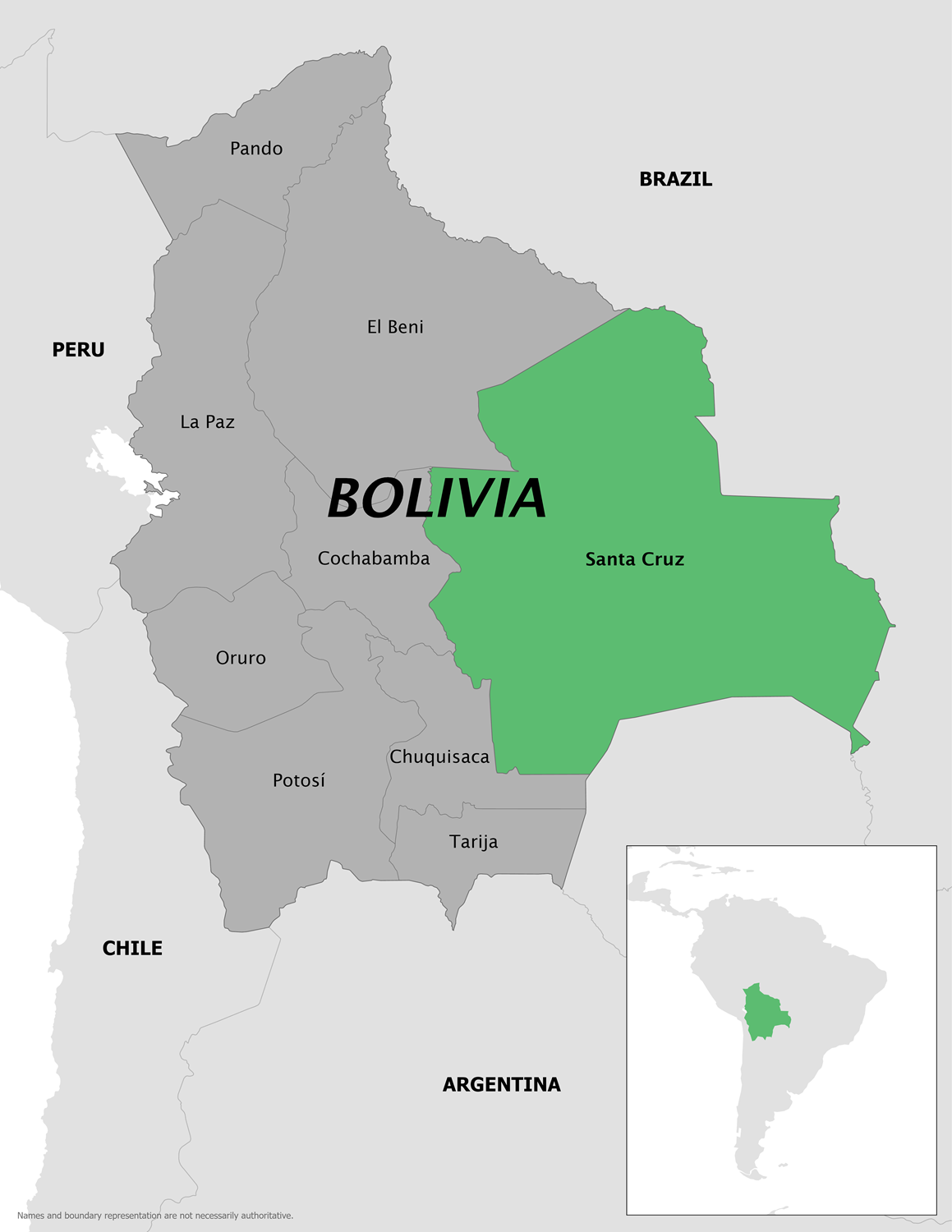
Like many countries in South America, the Plurinational State of Bolivia has been confronted with a multi-year outbreak of Chikungunya.
To alert international travelers visiting Bolivia, the U.S. Centers for Disease Control and Prevention (CDC) issued a Level 2 Travel Health Advisory on June 26, 2025, stating that this year's Chikungunya outbreak is centered in Bolivia's Santa Cruz department.
According to recent data, Bolivia welcomed around 984,000 international tourists in 2024.
Data released at the end of June 2025 indicates Bolivia has reported 3,863 Chikungunya cases this year and 505 cases in 2024.
So far in 2025, about 38 people have returned to the U.S. infected with the Chikungunya virus.
The CDC advises that if you are pregnant, you should reconsider travel to the affected areas, especially if you are nearing the time of delivery. Mothers infected around the time of delivery can pass the virus to their baby before or during delivery.
Newborns infected in this way or by a mosquito bite are at risk for severe illness, including poor long-term outcomes.
Furthermore, vaccination is recommended for most travelers who are visiting an area with a Chikungunya outbreak.
As of June 30, 2025, Chikungunya vaccines are approved for use by the CDC and are commercially available at travel clinics and pharmacies in the United States.
Current polio vaccines are made from either inactivated or weakened versions of the virus. These vaccines have been administered worldwide for many decades.
However, this approach presents challenges in certain situations. It highlights the need for an improved vaccine that does not rely on the virus itself, particularly as the global community strives to end poliovirus infections.
As of June 30, 2025, the U.S. Centers for Disease Control and Prevention identified poliovirus detections in 41 countries.
Developing vaccine formulations that do not use live viruses in their production would be highly beneficial, as it would eliminate the potential safety risks associated with handling and growing the virus, wrote Evaxion A/S in early June 2025.
To address this need, Evaxion received undisclosed funding from the Gates Foundation to help eradicate polio worldwide by exploring design options for a new and innovative vaccine.
This project will combine Evaxion’s leading and clinically validated AI-Immunology™ platform to identify and combine various antigens to combat the virus. Based on these findings, several new antigen constructs will be designed for selection and validation.
“We are thrilled to receive support from the Gates Foundation and help the world achieve the goal of completely eradicating polio. We are excited to apply our AI-Immunology™ platform to combat yet another infectious disease. The grant allows for further application and validation of our platform without adding to our operational spend,” says Christian Kanstrup, CEO of Evaxion, in a press release on June 3, 2025.
The inactivated (killed) polio vaccine (IPV) was developed in 1955 to produce antibodies in the blood that target all three poliovirus types, thereby preventing the spread of the virus, and has been offered in the U.S. since 2000.
IPV vaccinations are offered at clinics and pharmacies in the U.S.



