Search API
The Centre for Health Protection (CHP) of the Department of Health in Hong Kong announced today that no new cases of chikungunya fever (CF) have been reported.
The total number of confirmed CF cases in 2025 remains at 82.
As of January 14, 2026, the CHP's press release confirmed that 11 of these cases were local infections, while the others were imported from various areas.
Local cases of CF were reported following visits to the Tsing Yi Nature Trails and surrounding areas near Hong Kong in November last year, prompting the closure of the trails until further notice.
However, since the confirmation of the previous local case on December 10, 2025, no additional local cases have been reported.
Relevant government departments are conducting extensive efforts to control and prevent mosquito populations along the trails.
The CHP urges the public to refrain from entering the area to minimize the risk of contracting CF and to avoid exposure to the chemicals being used in the mosquito control operations.
While the U.S. Centers for Disease Control and Prevention (CDC) has not issued a Travel Health Advisory for Hong Kong, it did publish a Level 2 advisory in 2025, which has since been removed.
When departing from the United States for Hong Kong or any at-risk destination in 2026, chikungunya vaccination services are offered at travel health clinics located in most cities.
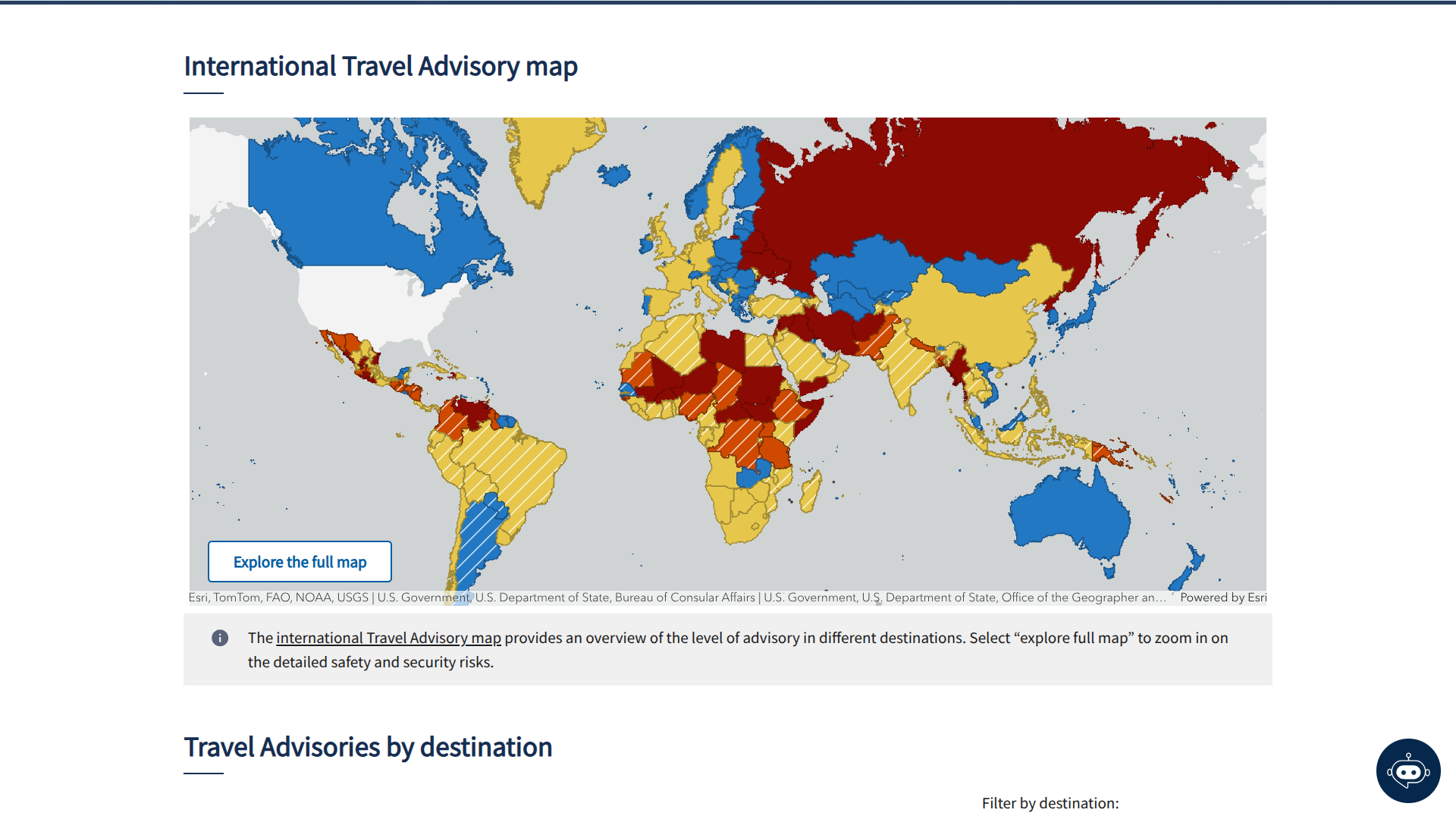
The U.S. Department of State has issued its highest-level warning, Level 4: Do Not Travel, for 22 destinations due to life-threatening risks. These locations are subject to ongoing security assessments, and the reasons for the warnings vary by destination.
As of mid-January 2026, the State Department lists the following countries and areas under this critical advisory: Afghanistan, Belarus, Burkina Faso, Burma, Central African Republic, Gaza, Haiti, Iran, Iraq, Lebanon, Libya, Mali, Niger, North Korea, Russia, Somalia, South Sudan, Sudan, Syria, Ukraine, Venezuela, and Yemen.
These advisories, including reissuances in early January 2026, emphasize that U.S. citizens should avoid travel to these areas entirely.
In many of these areas, the U.S. government has very limited or no ability to assist American citizens during emergencies.
For U.S. citizens already present in Level 4 destinations, the Department strongly urges departure as soon as safe travel options become available, as the government's capacity to provide emergency support, including evacuations, remains constrained in these high-risk environments.
The State Department recommendations for all international travelers include enrolling in the free Smart Traveler Enrollment Program to receive real-time updates, alerts, and emergency assistance from the nearest U.S. embassy or consulate.
Seperately, as of January 15, 2026, the UK's Foreign travel advice offers similar information, including entry requirements, safety and security, health risks, and legal differences.
While the advisories focus on security threats rather than health risks, international travelers should cross-reference the U.S. Centers for Disease Control and Prevention Travel Health Notices and the UK Travel Health Pro for any vaccine-preventable disease outbreaks, such as chikungunya, dengue, yellow fever, or zika, which may overlap in some tropical regions
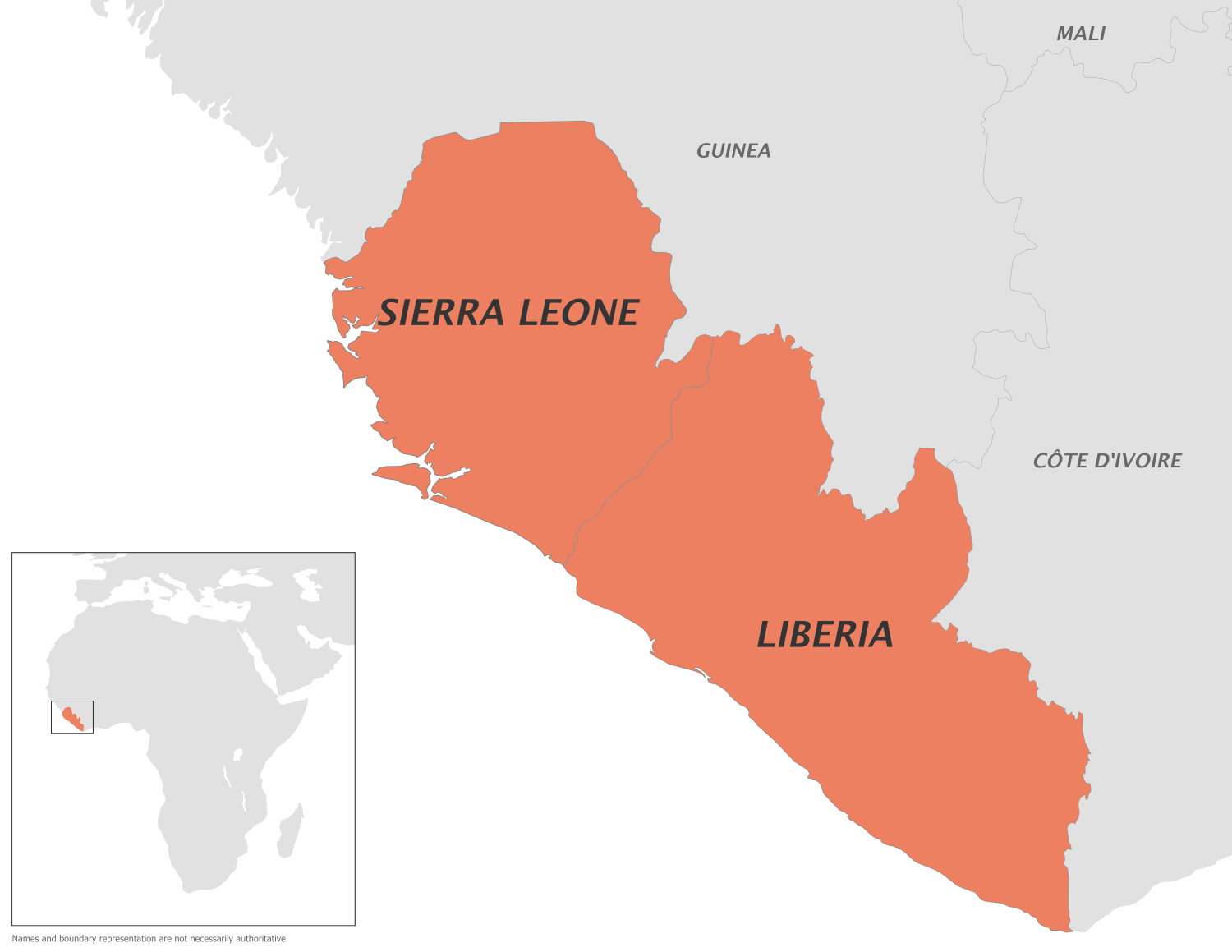
The U.S. Centers for Disease Control and Prevention (CDC) today issued a Level 2 Travel Health Notice, advising travelers to practice enhanced precautions due to the ongoing clade II Mpox outbreak in Liberia.
As of early November 2025, the West African country of Liberia has reported 2,447 suspected cases of Mpox since the outbreak began in September 2024. The virus is transmitted between clade IIa and clade IIb strains, with local human-to-human spread observed.
There are also cross-border links to neighboring countries such as Sierra Leone.
This CDC travel advisory, effective as of January 14, 2026, emphasizes the transmission of clade II Mpox from person to person, including through sexual and other intimate contact. The CDC has noted that both males and females are affected approximately equally.
To combat the outbreak, the Liberian Ministry of Health, in collaboration with partners such as the World Health Organization and the Africa CDC, has implemented enhanced response measures. These measures include a nationwide vaccination campaign to administer 42,720 vaccine doses to high-risk groups by the end of January 2026.
The CDC advises travelers to Liberia to review this notice and consider getting vaccinated against Mpox (JYNNEOS®) if they plan to engage in activities that may increase their risk.
Last year, approximately 80,000 international tourists visited Liberia.
This U.S. FDA-approved vaccine is offered at various travel clinics throughout the USA.
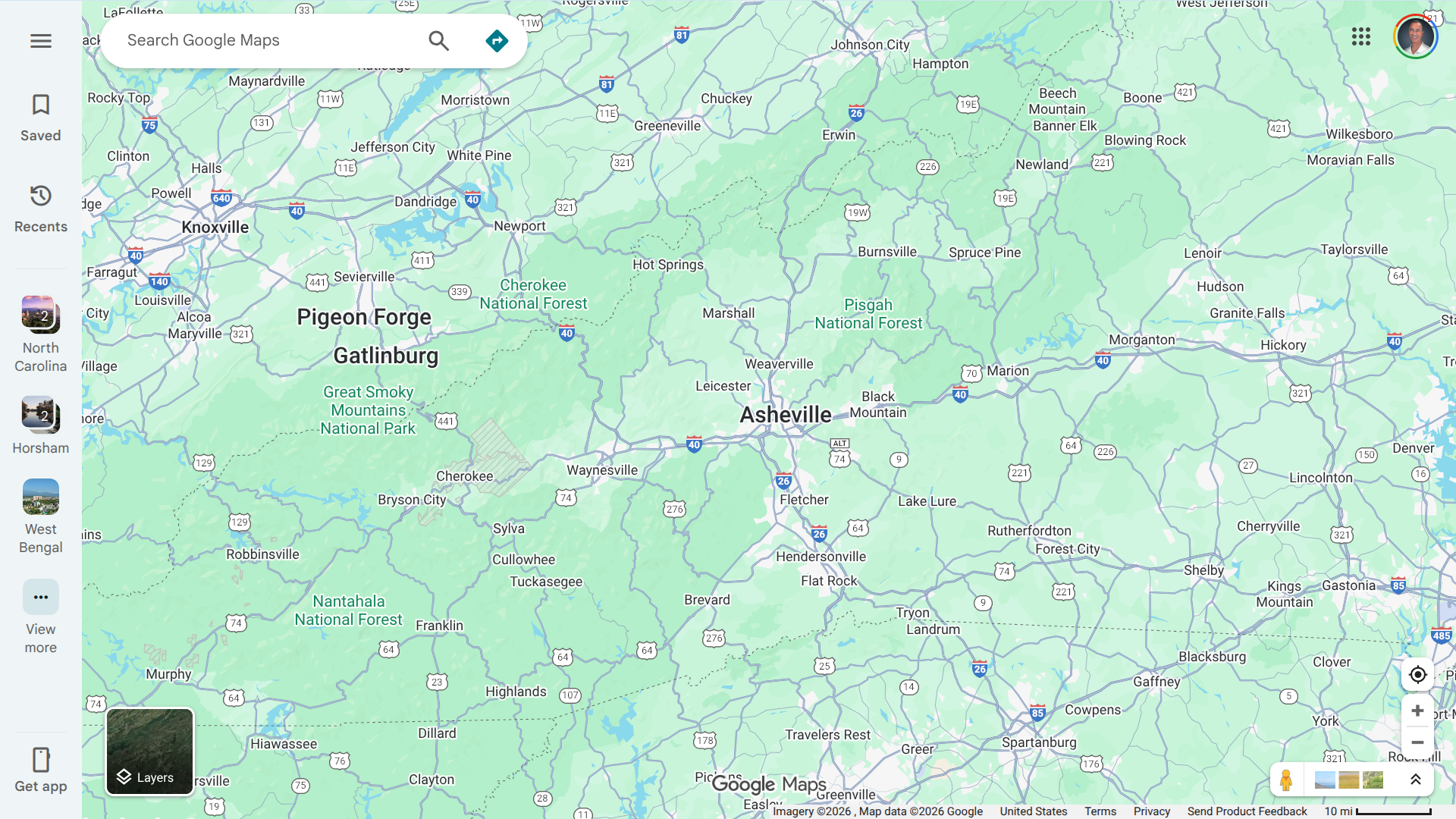
North Carolina health officials have confirmed three cases of measles in Buncombe County, which is home to the city of Asheville, a leading tourist destination. This marks the first confirmed instances of measles in the area in decades.
The North Carolina Department of Health and Human Services (NCDHHS) and Buncombe County Health and Human Services announced these cases on January 6, 2026. The cases involve three siblings who recently traveled to Spartanburg County, South Carolina, where a rapidly expanding measles outbreak has been occurring since late 2025.
These children began to show symptoms approximately one to two weeks after visiting the measles outbreak area, which is about 100 miles away.
Additionally, a potential public exposure occurred when the infected children visited the Mission Hospital Emergency Department waiting room (509 Biltmore Ave., Asheville, NC 28801) between 2:00 a.m. and 6:30 a.m. on January 4, 2026.
About two dozen potentially exposed individuals have been contacted by health officials.
Furthermore, anyone who was present during that time and has not been reached should call Buncombe County Public Health at 828-250-6100 for guidance.
And measles vaccines are readily available at primary care providers, pharmacies, and the Buncombe County Immunization Clinic located at 40 Coxe Ave., Asheville.
Across North Carolina, five confirmed measles cases have been reported since late December 2025, including additional cases in Polk and Rutherford counties.
According to NCDHHS, as of January 14, 2026, all but one of these NC cases are linked to travel from the South Carolina outbreak.
The outbreak in Spartanburg County has surged dramatically.
As of January 13, 2026, the South Carolina Department of Public Health reported 434 cases, with a significant increase of 124 new cases in just a few days. Most cases involve unvaccinated children.
Throughout 2025, measles outbreaks were identified in Texas, Utah, and other states. As well as record-setting outbreaks in Canada and Mexico.
Numerious health agencies recommend ensuring you are immune to measles before visiting an outbreak area in 2026.
Mexican health authorities recently confirmed the country's first case of mpox caused by clade Ib of the monkeypox virus (MPXV).
According to a report from the World Health Organization (WHO) on January 9, 2026, the affected individual is an adult male with a recent travel history to Germany. He is currently receiving medical care in Mexico.
As of January 13, 2026, there is no indication of community transmission of MPXV in Mexico, but officials are monitoring the situation closely.
This case represents a significant development in Mexico's mpox surveillance efforts. Previous cases in the country since the global outbreak in 2022 have mainly involved clade II strains, primarily concentrated in urban areas such as Mexico City and Quintana Roo.
Clade Ib, a subclade of clade I, has emerged notably in Africa and is associated with more severe cases compared to clade II variants in specific contexts. Clade Ib is characterized by sustained human-to-human transmission and has been linked to more severe outcomes in vulnerable populations, such as those with weakened immune systems.
Historically, clade I has a higher case-fatality rate (3–11%) than clade II.
However, improved healthcare access in non-endemic countries has so far reduced fatalities among imported cases, according to the WHO.
The WHO has reported over 100 travel-associated clade Ib cases in high-income countries across Africa, Asia, Europe, the Middle East, North America, South America, and Australia, with no deaths recorded in these imported cases to date.
Since November 2024, there have been 10 reported cases of clade I mpox (Ib) in the United States. Most of these cases occurred in individuals who had recently traveled to areas with known clade I outbreaks or were linked to such travelers, such as household contacts.
This specific case highlights the ongoing global circulation of clade Ib and underscores the role of international travel in the spread of the virus.
Health authorities, including the WHO, state that the JYNNEOS® (MVA-BN®) vaccine offers cross-protection across different mpox clades because it is based on the vaccinia virus, which confers broad immunity to orthopoxviruses.
The WHO says vaccine protection is expected to be effective across clades, with no evidence of significant immune escape in clade Ib.
In the United States, JYNNEOS is offered at various clinics as of early 2026.

Health authorities in West Bengal, India, recently issued a high alert after detecting two suspected cases of the Nipah virus (NiV) at the Virus Research and Diagnostic Laboratory of the Indian Council of Medical Research at AIIMS Kalyani.
The cases, identified on January 11, 2026, involve two nurses who work at a private hospital in Barasat, in the North 24 Parganas district, near the Bangladesh border.
Both patients are in critical condition and are receiving ventilator support while in isolation, with preliminary tests indicating a possible Nipah infection.
According to local media reports on January 13, 2026, the Nipah virus was first identified during the 1998-1999 Malaysian outbreak and has since caused sporadic, high-fatality outbreaks in South Asia.
Previous confirmed NiV cases in India were reported from Kerala state, specifically in the districts of Malappuram and Palakkad, along India's southwest coast.
According to the WHO's Disease Outbreak News in August 2025, four confirmed NiV cases, including two deaths, were reported in 2025.
As of 2026, there is no licensed Nipah vaccine available for humans, but research has accelerated due to the virus's priority status from the World Health Organization.
Key vaccine candidates include subunit vaccines targeting the fusion protein that have shown promise in Phase I trials for safety and immune response.
The University of Oxford launched the world's first Phase II trial in late 2025.
Other research platforms are exploring mRNA and peptide-based approaches.
According to research, vaccine experts estimate that a licensed vaccine could be available within five years, says Don Hackett, publisher of Vax-Before-Travel.com.