Pertussis vaccination during pregnancy has been implemented in many high-resource countries, and recent data from the United States, the United Kingdom, and South America demonstrate its effectiveness in reducing infant pertussis in the first two months of life.
What has not been well established is whether 'blunting' (maternal immunological interference) has clinical consequences.
Blunting of the infant’s subsequent response to primary immunization by maternally derived antibodies has been demonstrated for many antigens.
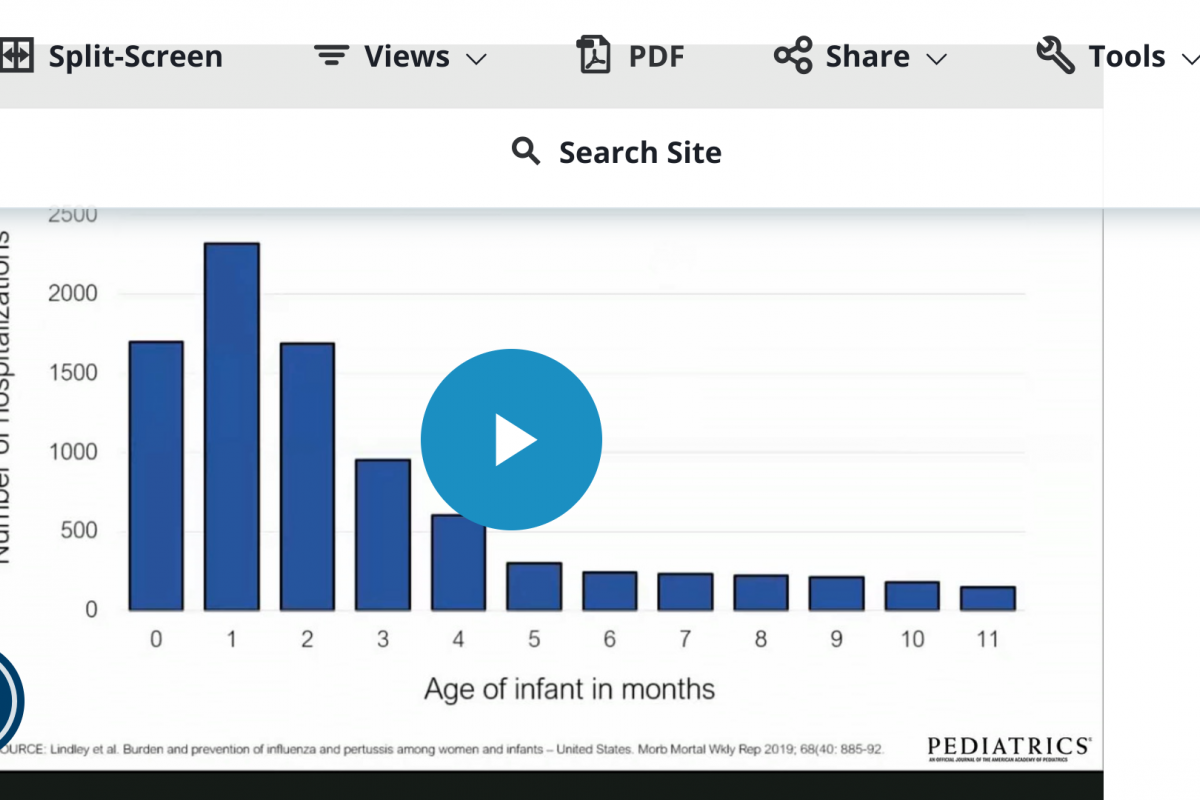
A new study published in the journal Pediatrics disclosed pertussis vaccination near 28 weeks' gestation was associated with a lower risk of infection among infants through 8 months of age.
These Australian researchers reviewed records from 2013 through 2017 to calculate the impact after infants received diphtheria, tetanus, and pertussis DTaP doses.
The vaccine effectiveness (VE) from maternal vaccination in infants younger than two months was 70.4% (95% confidence interval 50.5% to 82.3%).
Published on October 9, 2023, these researchers observed slightly lower VE point estimates for the third dose of infant pertussis vaccine among maternally vaccinated compared with unvaccinated infants (76.5% vs. 92.9%, P = .002) and did not observe higher rates of pertussis infection (hazard ratio, 0.70; 95% CI, 0.61–3.39).
The vaccine was given usually between 28 to 31 weeks gestation.
A commentary by Kathryn Edwards, MD, professor of pediatrics at Vanderbilt University Medical Center, stated, 'The consequences of maternally derived antibody on infant responses will need to continue to be monitored, as was done in the carefully conducted study of pertussis reported in this issue of Pediatrics. It will be critical to assess the burden of vaccine-preventable diseases and affirm that blunting from maternal immunization has no material impact on disease control.'