Search API
Since the end of the recent pandemic, medical care has become a global industry. Research reveals that about 1.4 billion passengers are expected to fly in 2025.
The Medical Value Travel (MVT) population, which travels to countries seeking healthcare services, is experiencing rapid growth worldwide.
According to the Federation of Indian Chambers of Commerce (FICCI), the global MVT market size was valued at $115.6 billion in 2022 and is expected to reach approximately $286.1 billion by 2030.
In India, the MVT is expected to grow at a CAGR of 21.1% from 2020 to 2027 and is projected to reach $13 billion by 2026.
Bangladesh, Iraq, Maldives, Afghanistan, Oman, Yemen, Sudan, Kenya, Nigeria, and Tanzania account for about 88% of the total international patients visiting India.
The leading health services offered in India are for heart surgery, knee transplant, cosmetic surgery and dental care as the cost of treatment in India is considered to be the lowest in Asia.
The FICCI video stated on May 26, 2025, 'Realizing the increasing importance of global medical value travel and its significance to the Indian medical industry.'
Recent research indicates that tens of millions of people are not adequately vaccinated before visiting disease-endemic countries.
For example, in Europe, over 4% of returning international travelers who recently displayed symptoms may be infected with a mosquito-transmitted disease, such as chikungunya, dengue, or Zika.
When departing abroad in 2025, the U.S. CDC recommends that travelers consult with a travel vaccine expert about immunization options at least one month before departure.

ImmunityBio, Inc., today announced the signing of a strategic Memorandum of Understanding (MOU) with the Ministry of Investment of Saudi Arabia (MISA), King Faisal Specialist Hospital & Research Centre (KFSHRC), and King Abdullah International Medical Research Center (KAIMRC).
As of May 27, 2025, this multi-party collaboration will introduce the FDA-approved Cancer BioShield platform to Saudi Arabia and the broader Middle East, marking a new era of immune-restorative therapies for cancer patients.
Dr. Patrick Soon-Shiong, Founder, Executive Chairman, and Global Chief Medical and Scientific Officer of ImmunityBio, stated in a press release, “We are honored to work with KAIMRC, KFSHRC, and MISA to bring this transformative technology to the region."
"The BioShield platform changes the way we think about treating cancer, not by destroying the immune system but by restoring and activating it."
"The root cause of early mortality is the collapse of the immune system—lymphopenia is the disease, and cancer is a symptom."
"Together, by considering this a paradigm change, we can build a regional center of excellence for next-generation immunotherapies in which we activate the body’s natural defenses.”
The BioShield platform, powered by Anktiva (nogapendekin alfa inbakicept)—the world’s first FDA-approved IL-15 superagonist to stimulate the proliferation of NK and T cells (lymphocytes)—represents a paradigm shift in cancer care.
Unlike conventional treatments, such as chemotherapy and radiation, which kill and suppress natural killer immune cells, thereby paradoxically catalyzing further spread, BioShield protects and activates the immune system’s natural killer cells and T cells to restore immune function and prolong life.
For the first time in medicine, physicians can address the long-overlooked impact of lymphopenia (loss of NK and T cells), induced by current standards of care of chemotherapy, radiation, or by the cancer itself.
The BioShield is the first therapy in history to specifically address the protection and restoration of lymphocytes, represented by NK, CD8, and CD4 T cells—the most important cells in the body needed to fight cancer and infection.
Treating lymphopenia is an answer to premature death from life-threatening diseases such as cancer and sepsis, and, potentially, to aging and longevity in health.
As of May 2025, Anktiva plus BCG Vaccine is available at various clinical sites in the U.S.

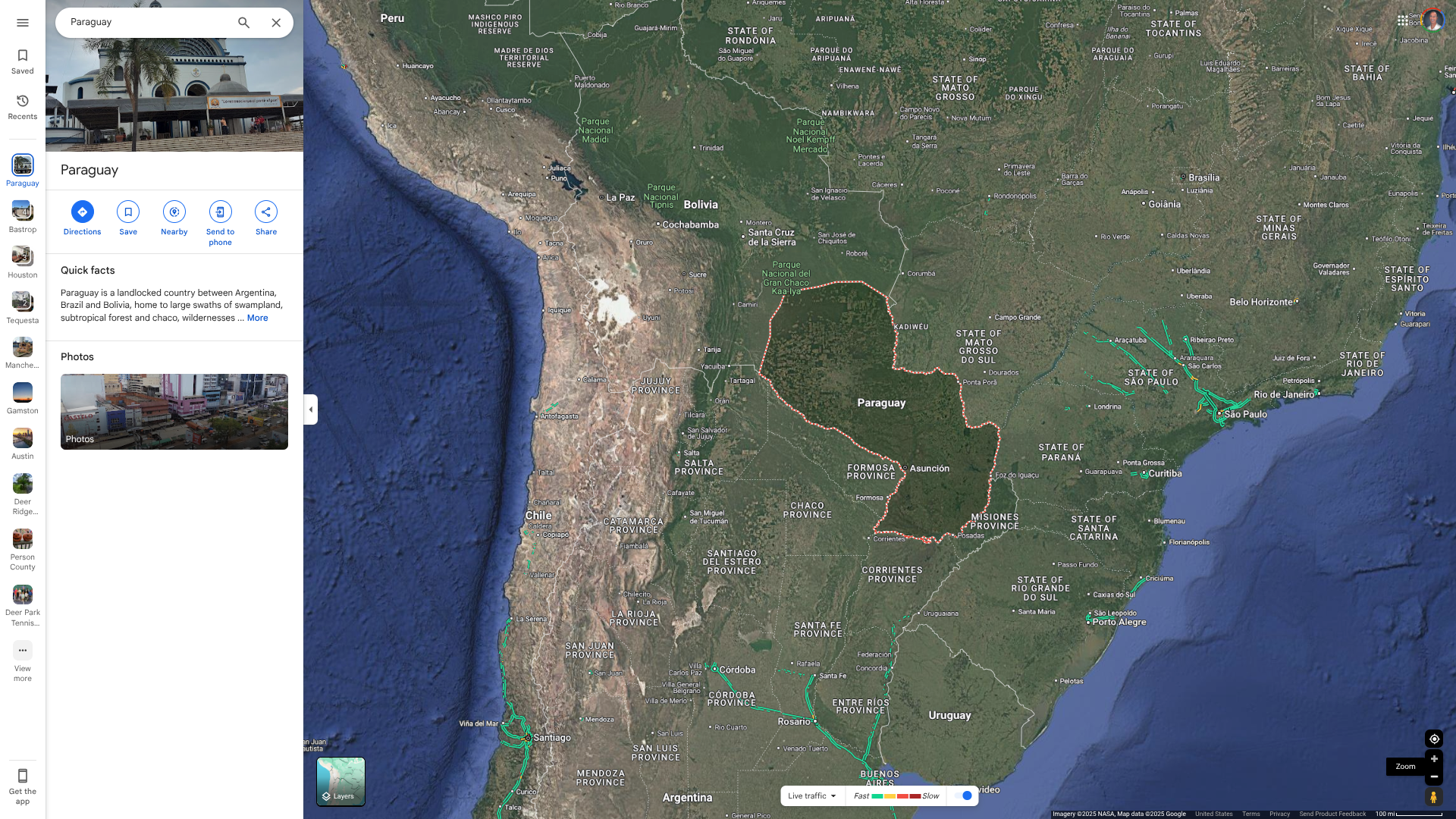
According to a recent blood donor study, about 37% of the residents of the Republic of Paraguay may be infected with the Chikungunya virus.
A research article published in the journal Medical Virology on May 13, 2025, stated that serum samples from 546 blood donors across seven regional districts and Asunción were collected from March to May 2023.
Anti-CHIKV IgG prevalence was 37.2%, with men showing a seroprevalence nearly 10% higher than women, but no significant age-related differences were observed.
Regional variation in CHIKV seroprevalence was not substantial.
In conclusion, this study suggests a high seroprevalence of CHIKV in Paraguayan blood donors, with the notable CHIKV prevalence underscoring the effects of recent outbreaks.
As of May 26, 2025, the U.S. CDC says there has been evidence of CHIKV transmission in Paraguay within the last 5 years.
During 2024, about 2.2 million people visited this South American country.
Furthermore, the CDC says certain travelers visiting Paraguay may be considered for Chikungunya vaccination. CHIKV vaccines are offered at travel clinics and pharmacies in the United States.
The São Paulo State Health Department recently confirmed that Dengue is a seasonal disease. During the rainy season, a high transmission rate can be observed. Throughout Brazil, Dengue is the most critical arbovirus disease transmitted by arthropod vectors.
Over the past 25 years, nearly 18 million Brazilians have been infected with Dengue.
As of May 26, 2025, the state of São Paulo, with a population of 44 million, has reported about 617,000 of Brazil's 2.5 million Dengue cases this year. According to data, the state of São Paulo leads in the number of deaths from dengue fever, with 692 cases.
In February 2025, a public health emergency was declared to alert residents and international visitors of this continuing health risk.
Last year, about 2.1 million Dengue cases and 2,100 related fatalities were reported in São Paulo.
This ongoing surge is related to Dengue virus serotype 3 (DENV-3) reemergence since the population is not immunized against this serotype.
While a second-generation Dengue vaccine will be limited in availability in 2025, Butantan Institute in São Paulo has been involved in developing an innovative vaccine that could become available in 2026.
Additionally, with the annual influenza season arriving in South America, beginning on May 27, 2025, the flu vaccination will be offered to the entire population in São Paulo's 645 municipalities over six months of age.
When visiting Brazil in 2025, the U.S. CDC recommends international travelers speak with a travel vaccine expert about various immunization options, such as chikungunya and yellow fever.

The Republic of Singapore's Ministry of Health (MOH) and Communicable Diseases Agency (CDA) recently updated the vaccination requirements for all pilgrims travelling to Saudi Arabia for Haj or Umrah pilgrimages, in consultation with Majlis Ugama Islam Singapura (MUIS).
This vaccination update, published on May 8, 2025, safeguards pilgrims' health throughout their journey and better protects them from infections while overseas.
Singapore pilgrims travelling to Saudi Arabia for the Haj pilgrimage in June 2025 also need to be vaccinated for influenza and COVID-19 before travel, in addition to meningococcal vaccination. Pilgrims are to ensure that:
Meningococcal vaccination: Pilgrims must complete their meningococcal vaccination at least 10 days before departure and provide proof of immunization.
COVID-19 vaccination: Pilgrims must show proof of either a single dose of the updated vaccine for the 2024-2025 season, completion of the primary vaccination series, or laboratory-confirmed recovery from a COVID-19 infection during 2024.
Influenza vaccination: Pilgrims must receive their influenza vaccination within 12 months before their travel date and provide proof of immunization.
In addition, the MOH and CDA recommend that pilgrims ensure they are updated with other nationally recommended vaccinations listed in the National Adult Immunisation Schedule and National Childhood Immunisation Schedule.
For example, elderly and medically vulnerable pilgrims should take the pneumococcal vaccine before their trip. For more information about nationally recommended vaccinations and available subsidies, please visit the CDA website.
Hajj is an annual Islamic pilgrimage to Mecca, Saudi Arabia, and Umrah is a pilgrimage to Mecca, located in the Hejazi region of Saudi Arabia.

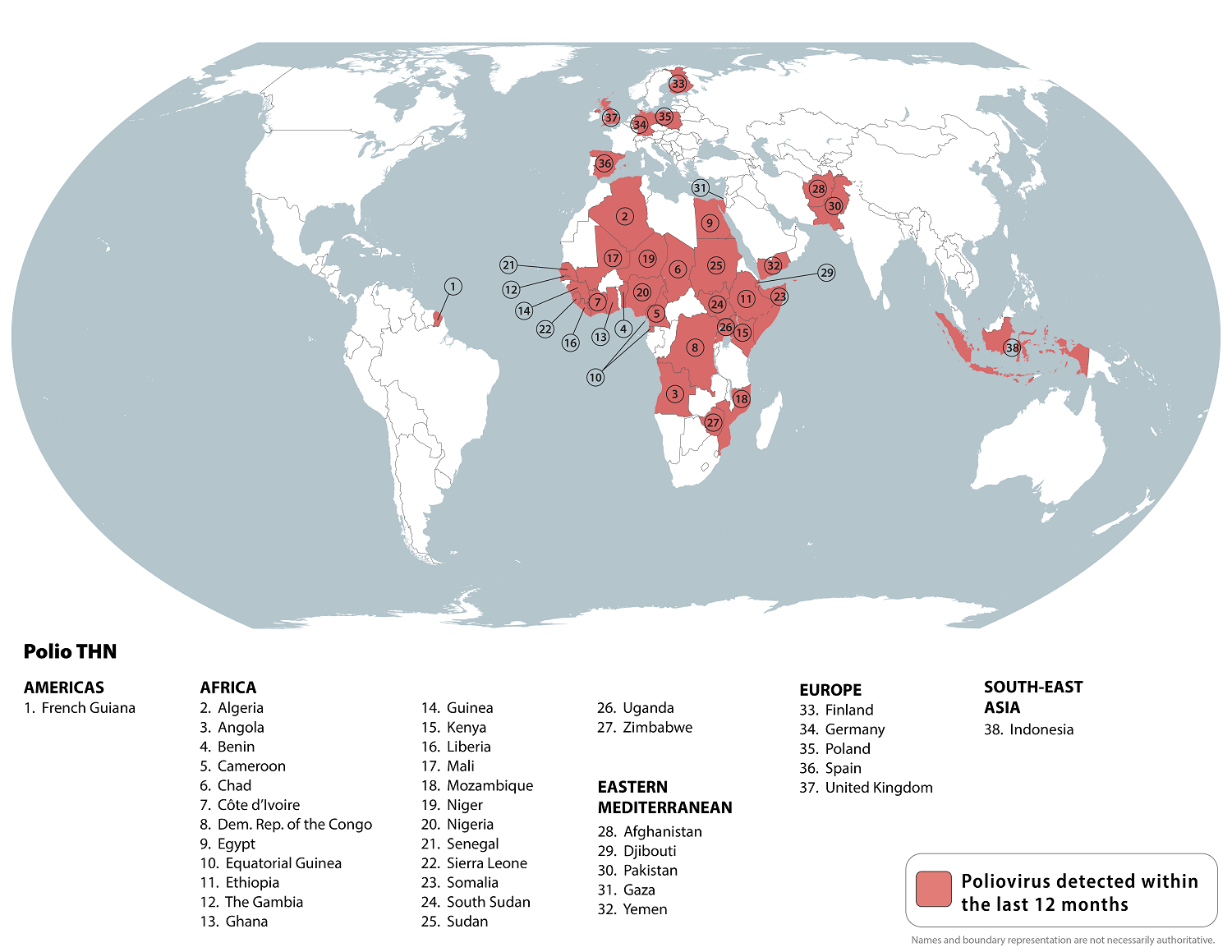
When the polio eradication campaign was launched in 1988, the ambitious goal was to eliminate the disease by 2000. Despite the campaign's remarkable progress, polio remains a global health challenge in 2025.
According to the World Health Organization (WHO), polio remains a global health concern in May 2025.
The WHO says there is no cure for polio, and infection prevention through vaccination is the best protection available.
Polio paralysis is caused by a human enterovirus called the wild poliovirus. However, less than 1% of wild poliovirus infections result in paralysis.
An article published in Clinical Infectious Diseases on May 21, 2025, says one significant misstep of the polio vaccination was the transition from the trivalent oral polio vaccine (OPV) to the bivalent OPV, which excluded the type 2 vaccine.
This switch occurred after the eradication of wild-type 2 poliovirus was declared in 2015.
This change was made to curb the emergence of circulating vaccine-derived polioviruses (cVDPV), which occur when the weakened virus in the vaccine mutates back to virulence and spreads in under-immunized populations.
Contrary to the optimistic modeling predictions, the exclusion of type 2 vaccine led to a dramatic increase in cVDPV cases, skyrocketing from just 6 cases in 2016 to more than 1,000 cases 3 years later, with more than 3,300 children paralyzed since the switch in 43 countries.
These researchers believe that the Inactivated Polio Vaccine (IPV) must be an essential part of polio eradication campaigns in the future.
Today, the U.S. CDC is partnering with other agencies through the Global Polio Eradication Initiative (GPEI) to help eradicate polio globally. Last week, the GPEI reported that Germany, Nigeria, Papua New Guinea, and Sudan confirmed cVDPV2-positive environmental samples and human infection.
As of April 2025, the CDC identified 38 countries reporting poliovirus detections.
In the U.S., vaccinations with the IPV are standard with the CDC's schedules, including a booster dose for certain people visiting poliovirus outbreak areas in 2025.
'Before any international travel, make sure you are up to date on your polio vaccines,' says the CDC.
As of May 25, 2025, polio vaccination services are offered at clinics and pharmacies nationwide.