Search API
With the global resurgence of the highly contagious measles virus, Southeast Asia is being affected by infected international travelers, including the city-state of the Republic of Singapore. In early 2026, Singapore's Communicable Diseases Agency (CDA) reported a significant increase in measles cases.
Despite historically high vaccination rates, measles remains a significant public health concern in Singapore.
By mid-February 2026, the CDA had recorded 13 confirmed measles cases for the year, including 11 in January alone, surpassing the 11 cases reported for the entire 2024 and nearing the 27 cases seen in 2025.
The CDA emphasized that all of the January cases involved individuals who were not fully vaccinated with the measles, mumps, and rubella (MMR) vaccine.
The vaccine has been compulsory for children since 1985, with the two-dose MMR regimen introduced in 1998.
While Singapore's robust public health system and high baseline coverage have so far limited widespread outbreaks among the six million residents, officials warn that sustained vigilance, including prompt reporting of suspected cases and strong adherence to vaccination, is essential to prevent outbreaks from escalating in our interconnected world.
As of February 22, 2026, proof of measles vaccination is not required for international visitors arriving in Singapore.
Researchers at Stony Brook University have developed an oral vaccine candidate based on a genetically modified, attenuated strain of Listeria monocytogenes. This vaccine stimulates powerful anti-tumor immune responses directly in the gut.
Led by immunologist Brian Sheridan, PhD, the research shows that the vaccine generates tumor-specific CD8+ T cells in the gastrointestinal tract.
In mouse models of colorectal cancer, the oral vaccine significantly improved tumor control, especially when combined with immune checkpoint inhibitors. This combination increases the infiltration of cancer-killing T cells into tumors.
Published on February 5, 2026, in the Journal for ImmunoTherapy of Cancer, the study presents a potential new strategy to overcome immunotherapy resistance in colorectal cancer, which is one of the leading causes of cancer-related deaths worldwide.
"The clinical significance of our laboratory findings is underscored by the vaccine performance in treating established tumors," says Dr. Sheridan. "While this vaccine alone initially curtailed local tumor growth, its true potential was revealed when combined with existing immune checkpoint inhibitors."
"This combination therapy led to profound tumor control in the model and suggests that the vaccine can effectively 'turn on' the immune system in tumors that were previously resistant to standard immune therapy," he explained in a press release.
Furthermore, the method demonstrated that oral immunization combined with immune checkpoint inhibitors induced the accumulation of tumor-specific CD8 T cells within the tumor microenvironment. These specialized immune cells remain stationed in the gut and provide immediate and long-lasting protection against cancer cells, a response not achieved by vaccination or immune checkpoint inhibitors alone.
While the results are promising in preclinical models, human trials will be necessary to assess safety and efficacy.

Seasonal influenza activity remains elevated across much of the United States, though trends vary regionally, with some areas seeing declines in influenza A and rises in influenza B cases.
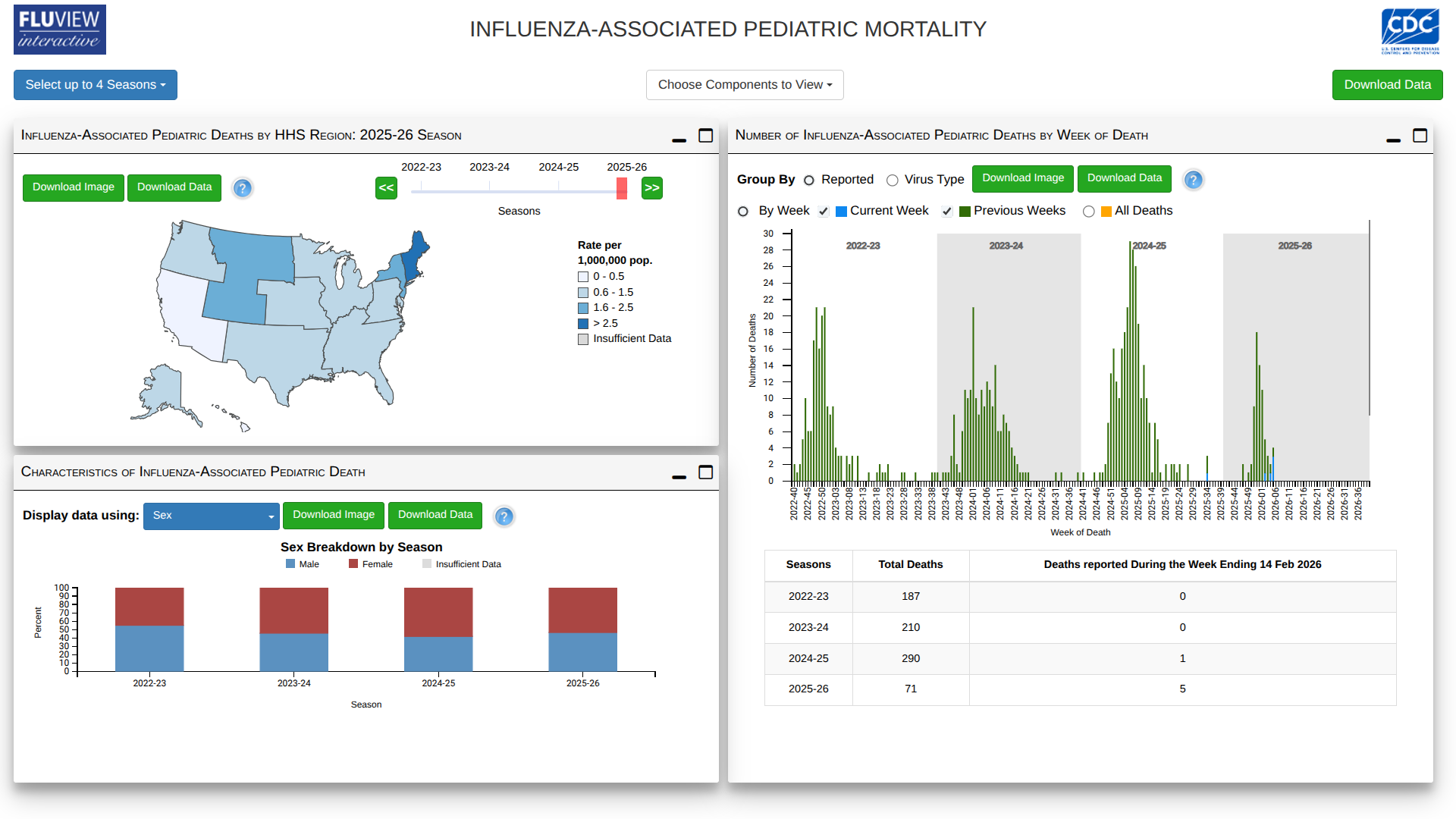
Unfortunately, the U.S. Centers for Disease Control and Prevention (CDC) has reported six new influenza-associated pediatric deaths in its FluView surveillance update for Week 6 of 2026.
This data highlights the ongoing serious risks posed by the virus to children.
Of the reported deaths on February 14, 2026, five occurred during the current 2025-2026 flu season, bringing the total number of pediatric fatalities this season to 71. These deaths happened during Weeks 2, 4, and 5 (ending January 17, January 31, and February 7, 2026).
Four deaths were linked to influenza A viruses, three of which were subtyped as A(H3N2).
Additionally, one death from the previous 2024-2025 season was reported this week, associated with influenza A(H1N1), bringing that season's final pediatric death total to 290.
Following an update to the U.S. childhood immunization schedule issued by the CDC on January 5, 2026, the annual influenza vaccine for children is now recommended based on shared clinical decision-making between parents or guardians and healthcare providers, rather than as a routine recommendation for all children aged 6 months and older.
Among the children eligible for vaccination and with known vaccination status, approximately 90% of the reported pediatric deaths this season occurred in those who were not fully vaccinated against influenza.
Health officials stress that this approach allows families to consider individual risk factors—such as age, underlying medical conditions, and community flu activity—when deciding whether to vaccinate. The CDC continues to emphasize that flu vaccination remains a crucial prevention tool during ongoing virus circulation.
Vaccination coverage data from the previous 2024-2025 season highlights ongoing challenges: only 50.2% of U.S. children aged 6 months through 17 years received at least one dose of the flu vaccine—the lowest coverage in the past 15 seasons.
As of early February 2026, interim coverage for the current 2025-2026 season stands at approximately 47.5%, similar to the same point last season.
The CDC urges parents to speak with their child's healthcare provider promptly about flu vaccination and to seek early antiviral treatment if flu symptoms develop in high-risk children.
As of February 21, 2026, flu shots remain available at clinics and pharmacies throughout the USA. However, the nasal flu shot is sold out for this season.
As the 2025-2026 influenza season progresses to its later stages, FluMist® —the only nasal spray flu vaccine in the United States—has officially sold out.
The sell-out of FluMist reflects typical end-of-season patterns, as it has a shorter shelf life compared to injectable flu vaccines.
FluMist, a live attenuated influenza vaccine (LAIV), does not require needles and is approved for healthy individuals aged 2 to 49.
According to AstraZeneca's FluMist website, the flu shot is "no longer available to order for the 2025-2026 flu season."
Users are encouraged to sign up for notifications ahead of the next influenza season.
As of February 20, 2026, AstraZeneca has not disclosed exact dose numbers for FluMist, as product-specific distribution and administration data are typically reported after the season ends. In other countries, FluMist® is known as Fluenz® Tetra.
Historically, FluMist has accounted for a smaller market share than injectable alternatives.
A key highlight of this season was the launch of FluMist Home in August 2025, which allowed for self- or caregiver administration.
This direct-to-consumer service enabled eligible individuals to order the vaccine online through a medical screening process, with home delivery in 34 states, covering approximately 80% of the eligible population.
Meanwhile, as the flu season continues to peak in some states, injectable vaccines remain available from healthcare providers and pharmacies across the USA. For the latest updates, visit cdc.gov/flu, flumist.com, or consult your healthcare provider.

Seasonal influenza continues to circulate at elevated levels across the United States in mid-February 2026, with health officials from the Centers for Disease Control and Prevention (CDC) noting a shift in the dominant strains driving infections.
According to the CDC's latest Weekly U.S. Influenza Surveillance Report for 2026, seasonal influenza activity remains high nationally. Influenza A activity is decreasing, while influenza B activity is increasing both nationwide and in most regions of the country. Influenza A(H3N2) viruses have been the most commonly reported subtype this season so far.
However, these trends show notable regional variability.
Regional differences are particularly evident in supplementary tracking tools.
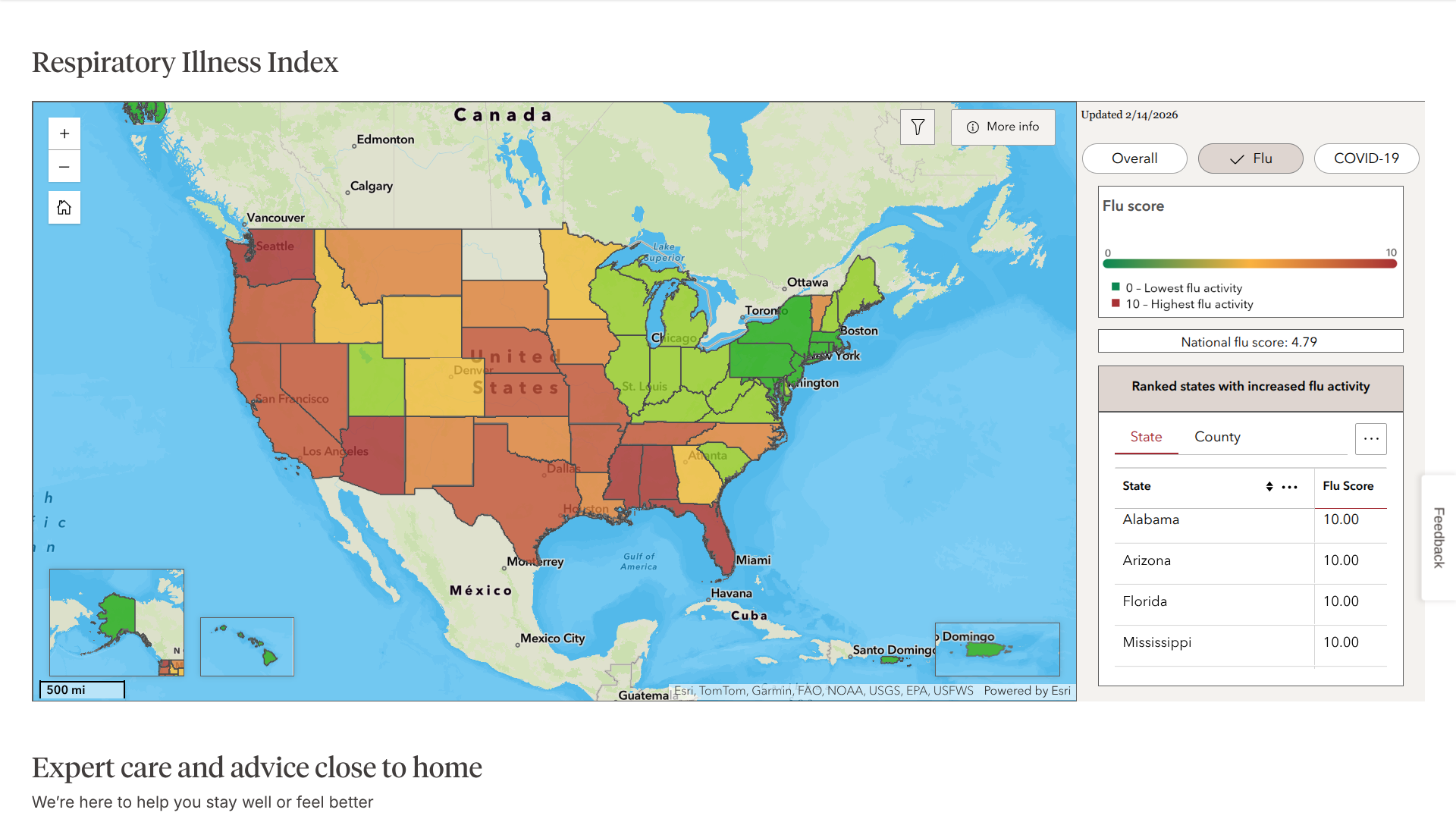
The Walgreens Respiratory Index©, which monitors respiratory illness trends using prescription, testing, and over-the-counter product data from Walgreens locations, illustrates this variability in mid-February 2026.
Numerous states in the eastern portion of the country have shifted to green, indicating lower relative intensity compared to baseline levels. In contrast, states in western areas continue to show red, representing the highest intensity category.
The Index also highlights maximum intensity in several southern states, including Alabama, Arizona, Florida, and Mississippi, where respiratory illness activity—driven largely by ongoing flu circulation—remains at peak levels.
Health experts emphasize that while influenza A declines in many places, the rise in influenza B could sustain elevated activity or lead to prolonged circulation in certain regions.
Officials continue to recommend vaccination.
"This flu season is exceptionally active," commented Anita Patel, vice president of Pharmacy Services Development at Walgreens, in a January 2026 press release.
"To protect yourself and others, I encourage everyone to get vaccinated as soon as possible, wash your hands frequently, and if you're feeling symptoms, get tested right away," Patel says.
The CDC updates its FluView surveillance weekly, and activity levels can fluctuate. For the most current data, visit the CDC's FluView website.
Furthermore, when traveling abroad, check the CDC's Travel Health Notices for information on disease outbreaks in other countries and related travel vaccine options.
Measles outbreaks have been reported in Europe this year, and cases in England have significantly increased in the first few weeks of 2026.
The UK Health Security Agency (UKHSA) reported on February 19, 2026, that 130 laboratory-confirmed cases of measles had been reported this year.
This indicates a rise of 34 cases since the previous weekly update.
The surge is primarily driven by an ongoing outbreak in North London, particularly in the borough of Enfield, where 50 cases have been confirmed. Nearby Haringey has recorded 10 cases, while Birmingham in the West Midlands accounts for 23 cases, making up 18% of the total.
Overall, 68% of cases are concentrated in London.
Fortunately, no measles-related deaths have been reported in 2026.
UKHSA officials urge parents and guardians to check their children's vaccination status and ensure they receive the MMR vaccine. Catch-up vaccination campaigns are currently underway, and vaccination records are published quarterly in health protection reports. Public health leaders emphasize that vaccination remains the most effective way to prevent the spread of measles.
With over 20 million annual visitors to London, this UKHSA advice is essential.
As measles cases continue to rise in London, visitors to these areas of England should consult a healthcare professional about vaccination options.
As a frequent airplane traveler who is concerned about catching the flu in crowded cabins with recycled air, I am thrilled by recent research on CR9114. This nasal spray delivers influenza-specific monoclonal antibodies.
This innovative product, if approved, could be ideal for high-exposure situations, such as flying during flu season, especially for travelers.
While modern commercial airplanes have high-efficiency ventilation systems that recirculate cabin air through HEPA filters, they don't remove all viruses. If an infected passenger is on board—especially one who is coughing or sneezing—flu virus transmission can still occur.
Published on February 4, 2026, in Science Translational Medicine, the study, led by researchers from Harvard in collaboration with Leyden Laboratories, demonstrates that the intranasal delivery of the broad-spectrum hemagglutinin antibody CR9114 is both safe and feasible.
In two Phase 1 trials involving healthy volunteers, repeated dosing (twice daily, given the approximately 3-hour nasal half-life) achieved sustained high antibody levels in the nose.
These recovered antibodies retained strong neutralizing activity against both influenza A and B viruses.
Importantly, this nasal spray is not a replacement for seasonal influenza vaccines but rather may offer a rapid, short-term immunoprophylactic option. By delivering high local antibody concentrations directly at the virus's entry point with small doses, CR9114 has shown early promise.
Previous preclinical tests on mice and macaques confirmed its safety and effectiveness against influenza challenge.
These findings lay the groundwork for future efficacy trials and provide proof of concept that intranasal antibodies can elicit efficacious passive immunity against influenza viruses.
For someone who dreads having post-flight sniffles turn into illness, this nasal spray could become an essential item to carry on board before flying.