Search API
In late December 2025, two disease alerts regarding dengue fever outbreaks offered conflicting advice for travelers.
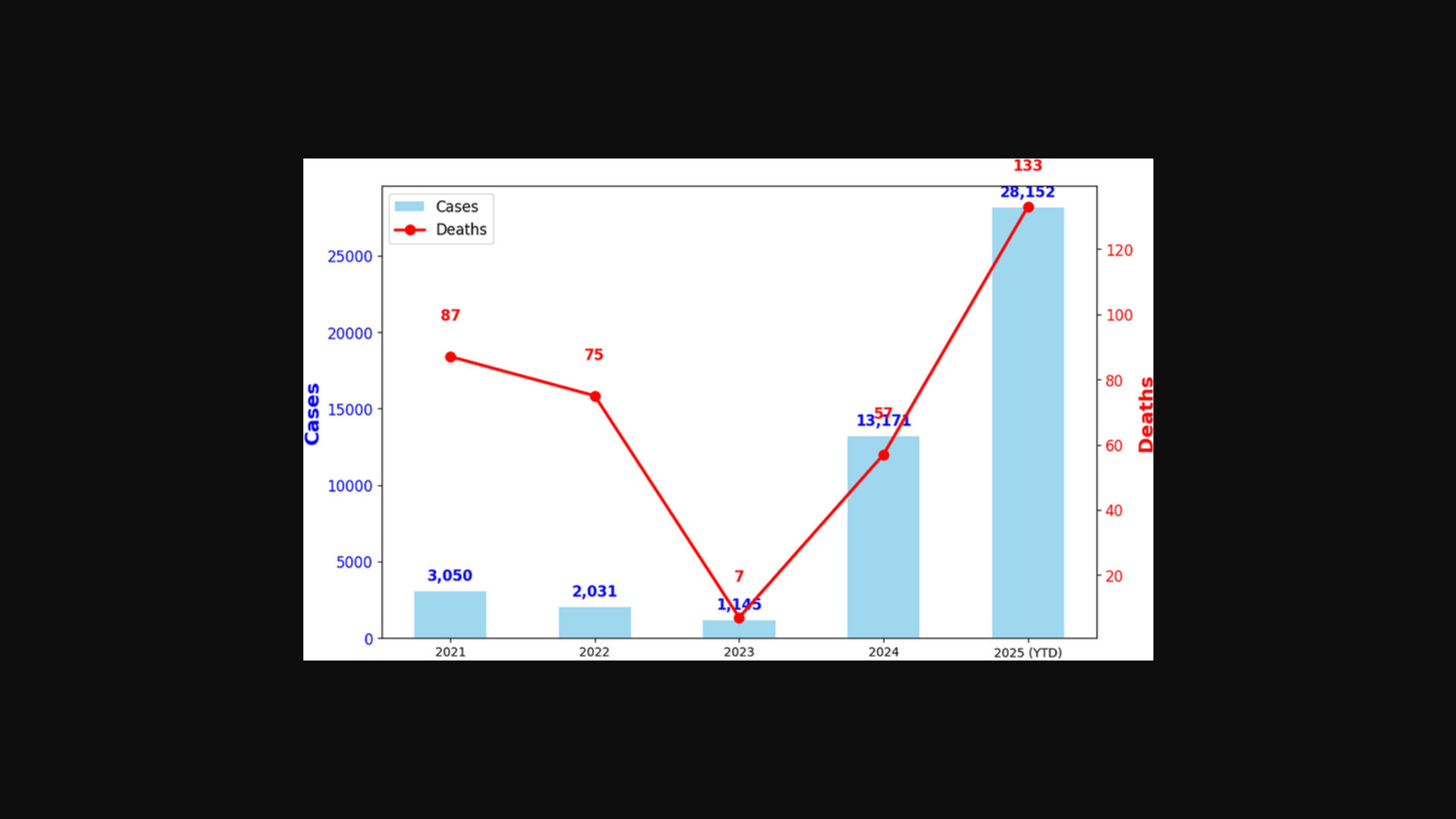
According to the Florida Department of Health (FDOH) Arbovirus Surveillance report for Week 51, as of December 20, 2025, there have been 62 locally acquired dengue cases recorded statewide. The counties most affected include Brevard with 35 instances, Miami-Dade with 25 cases, and both Hillsborough and Pasco with 1 case each.
Regarding the virus subtypes, the FDOH reported that DENV-3 accounted for 44% of the cases, while DENV-4 accounted for 21% in 2025.
Additionally, Florida has reported 415 Travel-Associated Dengue cases in 2025 who had traveled to a dengue-endemic area in the two weeks before onset.
However, today, the U.S. CDC reissued a global Level 1 Travel Health Notice identifying 12 countries experiencing dengue outbreaks late in 2025.
As of December 23, 2025, the CDC included Cuba, which is a neighbor of Florida. However, it did not address the health risk posed by this mosquito-borne virus in the southeastern counties of Florida.
Furthermore, neither the CDC nor the FDOH recommends the use of a dengue vaccine for prevention.
Currently, a first-generation dengue vaccine (Dengvaxia) is being tested in children in Puerto Rico, and the second-generation QDenga vaccine is offered in about 30 countries.
While the newly authorized third-generation, single-dose Butantan-DV vaccine is scheduled for release in Brazil in 2026, international travelers are optimistic that its availability will expand next year, including in Florida.
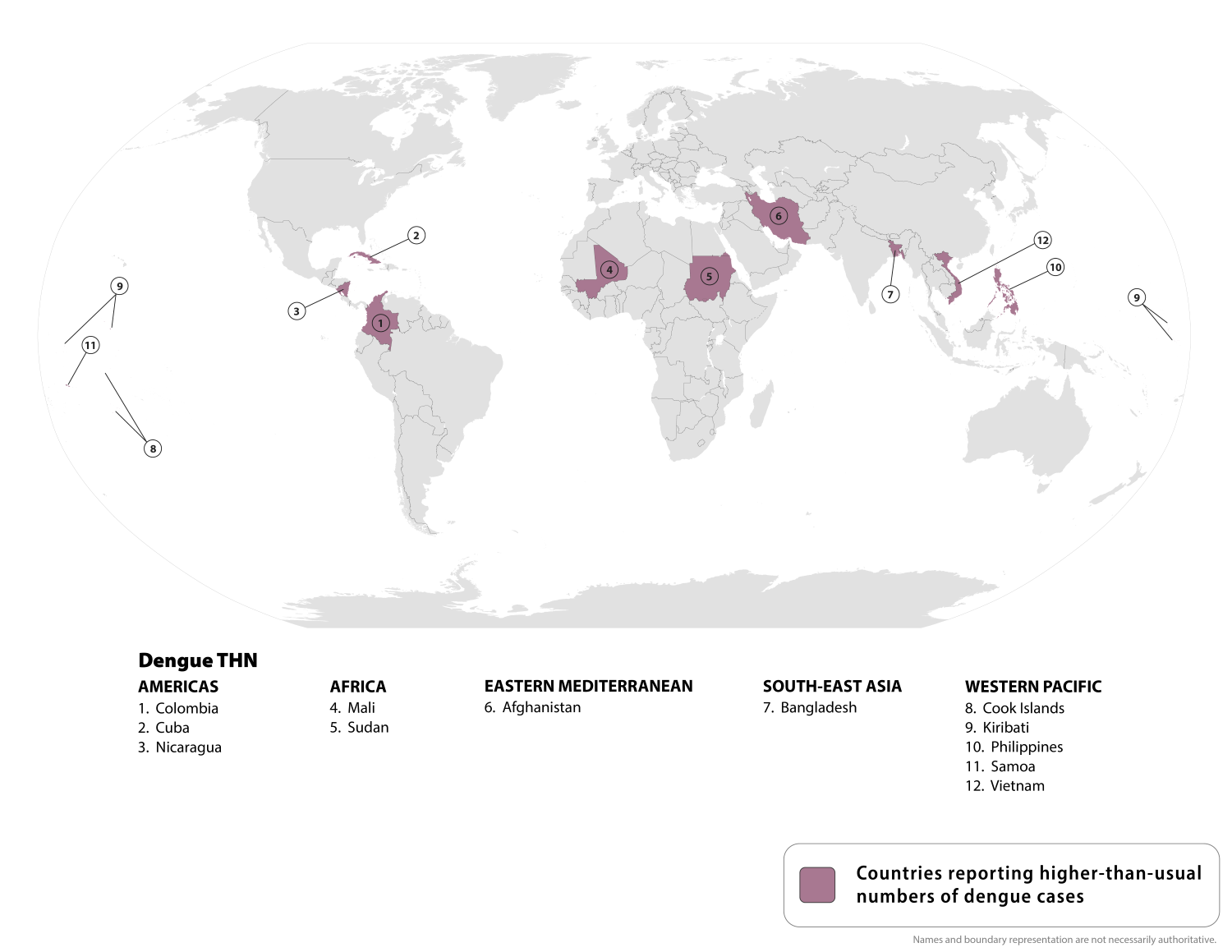
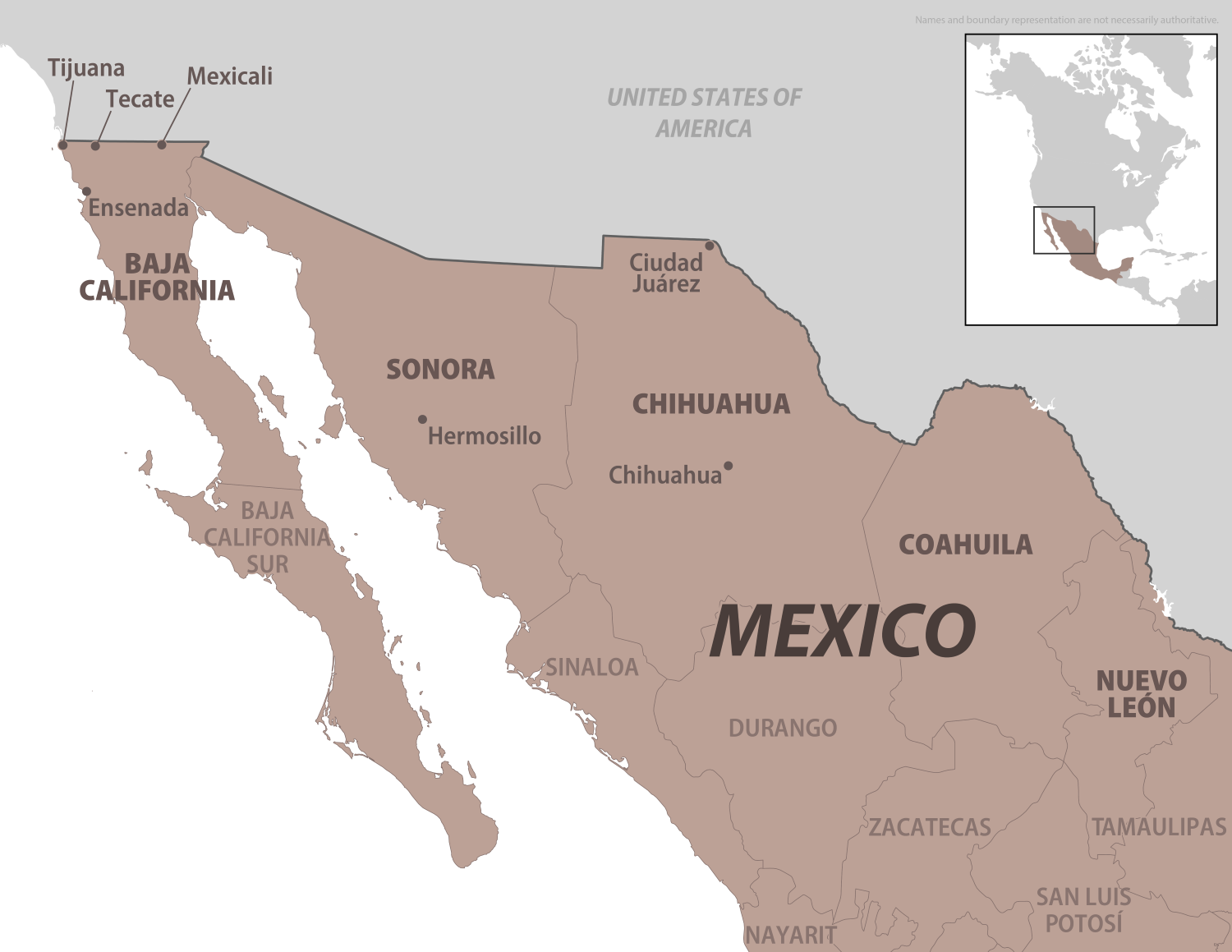
The U.S. government has issued a Level 1 Travel Health Notice regarding an outbreak of Rocky Mountain spotted fever (RMSF) in northern Mexico, specifically in the states of Baja California, Sonora, Chihuahua, Coahuila, and Nuevo León, which has been causing Illness and death in late 2025.
As of December 23, 2025, the Centers for Disease Control and Prevention (CDC) announced that ticks carrying the bacteria that cause RMSF have been found in several cities in northern Mexico.
These cities include Tecate, Tijuana, Ensenada, and Mexicali in Baja California, as well as Hermosillo in Sonora, Ciudad Juárez, and Chihuahua City in Chihuahua. The CDC warns that RMSF can be deadly if not treated promptly.
A similar CDC advisory was issued in 2023.
In the U.S., RMSF cases vary, but typically range from a few hundred to several thousand, says the CDC.
Since no protective vaccines are available as of 2025, travelers to these affected areas must take precautions. People can protect themselves by using a registered insect repellent on exposed skin and clothing.
Additionally, it is essential to check their bodies and their children's bodies for ticks daily, as well as their clothing. Ticks can be tiny, and their bites may be painless, so it is possible to overlook a tick bite. If you find a tick on your body, the CDC advises removing it as soon as possible.
The Transportation Security Administration (TSA) today announced it is gearing up for a record-setting holiday travel season.
On December 22, 2025, the TSA stated it is fully staffed and ready to screen a projected 44.3 million travelers at the nation's airport security checkpoints between Friday, December 19, 2025, and Sunday, January 4, 2026.
The TSA expects the heaviest traveler volume will be on Sunday, December 28, 2025.
The busiest day last year was December 27, when TSA screened just under 2.85 million travelers.
TSA Senior Official Performing the Duties of the Deputy Administrator, Adam Stahl, commented in a press release, "We continue to enhance the traveler experience by deploying new technologies and procedures t..."
One of these enhancements is REAL ID.
More than 94% of passengers already use their REAL ID or other acceptable forms of identification.
The TSA began enforcing the REAL ID Act in May 2025, and starting in February 2026, the agency will refer all passengers who do not present an acceptable form of ID and still want to fly to an option to pay $45 to use a modernized alternative identity verification system, TSA ConfirmID, to establish identity.
Another enhancement launched in July: TSA PreCheck® "Serve with Honor, Travel with Ease" benefits for the military and uniformed service community. This program includes expedited lanes for service members, free TSA PreCheck for surviving immediate family members of military servicemen and women who lost their lives in the line of duty, and TSA PreCheck discounts for spouses of military and uniformed service members.
With various disease outbreaks currently impacting international travelers, the U.S. CDC recently updated Travel Health Notices for chikungunya, dengue, measles, polio, and yellow fever. Many of these diseases have U.S. FDA-approved vaccines available at travel clinics in late December 2025.

With measles cases increasing by about 29% in 2025, the Republic of the Philippines Department of Health (DOH) plans to launch a measles vaccination campaign for children aged 6 months to 5 years, called 'Ligtas Tigdas', in 2026.
This vaccination campaign is essential to protect residents and visitors in 2026, as the Department of Tourism says the Philippines is ready for increased tourism.
According to the Philippine News Agency reporting on December 20, 2025, the first phase of the measles vaccination campaign will take place in Mindanao and the Bangsamoro region from January 19 to February 13, 2026.
The second phase will extend to Luzon and Visayas in June.
In the Philippines, the DOH recorded 4,843 measles-rubella cases in 2025. This data is 29% higher than the 3,748 cases logged in 2024.
In 2025, the Philippine Food and Drug Administration reported the circulation of counterfeit Speeda human rabies vaccine. This product may not be effective in the prevention of rabies and could contain harmful ingredients.
Additionally, the U.S. CDC included the Philippines in Dengue and Measles Travel Health Notices in 2025.
A recent Eurosurveillance rapid communication details Ireland's first known outbreak of the monkeypox virus (MPXV) clade 1b, involving four linked cases from August to October 2025, with cases related to a Dublin hospital.
On December 18, 2025, in Volume 30, Issue 50, the researchers wrote that, to the best of our knowledge, the first reported nosocomial transmission of MPXV clade Ib outside Africa occurred alongside the ongoing rise in MPXV clade Ib importations in countries outside Middle Africa.
This cluster highlights the ongoing possibility of mpox transmission from travel-associated cases. It emphasises the need for a high level of clinical suspicion for mpox in cases with relevant clinical symptoms and travel to countries where mpox is currently circulating.
The Irish outbreak originated with a traveler returning from Pakistan and spread via sexual, household, and nosocomial (in a healthcare setting) transmission. The cases were genetically identical and related to a sequence from Oman in February 2025.
The close relationship between the Oman and Irish sequences highlights the role of international travel in the spread of clade 1b and the potential for global transmission, the researchers added.
Since the second declaration of a public health emergency of international concern for mpox was lifted in September 2025, both MPXV clades I and II, and their subclades, continue to circulate globally, the World Health Organization (WHO) Disease Outbreak News reported in early December 2025.
The WHO considers there to be community transmission of the Mpox virus in various European countries, such as Belgium, Germany, and Spain.
In Ireland, as of December 21, 2025, the MVA-BN (JYNNEOS) vaccine is authorized in response to mpox outbreaks.
And when departing for Ireland or other mpox-risk countries, the U.S. CDC recommends international travelers speak with a travel vaccine advisor regarding immunization options.
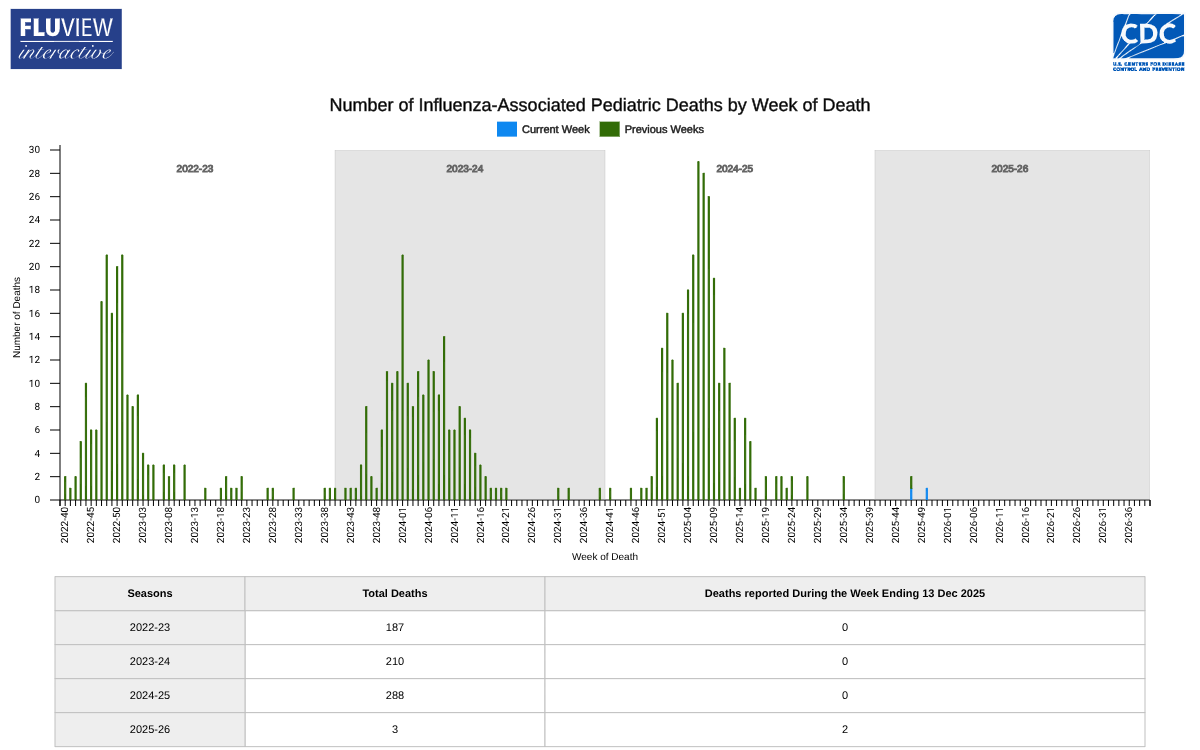
Influenza activity in the United States has recently spiked across the country, with 17 jurisdictions now reporting high or very high influenza-like illness (ILI) activity. This news comes as Americans prepare for holiday gatherings, a period when flu transmission often peaks.
According to the Centers for Disease Control and Prevention (CDC) week #50 FluView surveillance report, this indicator and others signal the earnest start of the 2025-2026 flu season.
The CDC also reported on December 19, 2025, three influenza-associated pediatric deaths this season, bringing the total to three.
This data is actually encouraging when compared to the 2024-2025 flu season, when 288 influenza-associated pediatric deaths were confirmed.
The CDC wrote 'the public is urged to take preventive actions, including vaccination, frequent handwashing, covering coughs and sneezes, and staying home when sick. There are prescription flu antiviral drugs that can treat ILI; those should be started as early as possible and are especially important for patients at higher risk for flu-related complications.'
For more information, visit the CDC FluView report at www.cdc.gov/flu/weekly.
While two malaria vaccines are currently in use in Africa, a new, innovative vaccine candidate is advancing in human clinical trials. Results from a study highlight the potential of multistage vaccines to address both infection and transmission, offering a new approach in the fight against malaria.
A new phase 2 randomized controlled trial published in The Lancet Infectious Diseases reports encouraging levels of protection from an investigational multistage malaria vaccine, ProC6C-AlOH/Matrix-M, against controlled human malaria infection in adults with lifelong exposure to the mosquito-transmitted parasite.
The vaccine candidates, which target multiple stages of the Plasmodium falciparum life cycle, the protozoan responsible for the most severe form of malaria in humans, have demonstrated promising efficacy in a study conducted among healthy adults in a malaria-endemic region of Mali.
Researchers, including the Malaria Research and Training Center at the University of Sciences, Techniques, and Technologies of Bamako, evaluated the vaccine's safety, immunogenicity, and protective efficacy. Participants received three doses of the vaccine or a control, followed by controlled exposure to the malaria parasite.
ProC6C-AlOH/Matrix-M achieved an efficacy of 54% (95% CI 9–77, p=0·029) at 12 weeks after the final dose, and 76% (95% CI 36–91, p=0·0022) in a time-to-event analysis, indicating a promising level of protection.
The study authors note that further research is needed to evaluate the vaccine's performance against naturally acquired malaria and in younger age groups, including children.
In a related commentary published by The Lancet on December 16, 2025, researchers wrote, "Having a first multistage malaria vaccine with proven efficacy is a key achievement, as there is consensus in the malaria community on the need to cover as many parasite stages as possible."
This study's results are essential as malaria transmission occurs in 80 countries across five WHO regions. Last year, there were an estimated 282 million cases of malaria globally reported by the World Health Organization.
As of December 21, 2025, malaria vaccines, such as R21/Matrix-M™, are not offered in the United States.
Mpox cases in Africa surged in 2025, driven by circulating Clade Ib and Clade I viruses, with more than 42,000 confirmed cases by late November, according to the World Health Organization (WHO).
On December 5, 2025, the WHO reported community transmission of the mpox virus persists in Burundi, the Democratic Republic of the Congo, Kenya, Malawi, Mozambique, the Republic of Congo, Rwanda, South Africa, the United Republic of Tanzania, Uganda, and Zambia.
To help reduce these outbreaks, mpox vaccine donations have continued in late 2025.
UNICEF Supply recently posted on X that 20,000 MVA-BN (JYNNEOS) mpox vaccine doses arrived in the African country of Liberia, part of a 32,000-dose donation from Bavarian Nordic.
As of December 15, 2025, this shipment marks the first phase of a total donation of 32,000 doses to UNICEF, supporting Bavarian Nordic's commitment to protecting communities and ensuring vaccines reach those at risk.
Bavarian Nordic wrote, 'We are grateful to be part of this collective effort and thank everyone working to bring protection to communities affected by mpox. We will share further updates as the remaining doses reach their destinations.'
Previously, the Africa CDC received a donation of 110,000 doses of mpox vaccines to support the ongoing response to the mpox outbreak across the continent. During October 2025, the vaccines were allocated to Uganda, one of the countries most affected.
Paul Chaplin, President and CEO of Bavarian Nordic, commented in a press release, "Mpox remains a health emergency in Africa as evidenced by the continued spread across the continent and increase in the number of cases."
In the United States, the number of mpox cases has been reduced in most states. A factor in this positive trend is that the JYNNEOS vaccine is offered at health and travel clinics as of December 20, 2025.
The U.S. CDC says at-risk travelers should get their first monkeypox vaccine at least six weeks before traveling to outbreak areas in Africa if possible.