Search API
North Carolina health officials have confirmed three cases of measles in Buncombe County, which is home to the city of Asheville, a leading tourist destination. This marks the first confirmed instances of measles in the area in decades.
The North Carolina Department of Health and Human Services (NCDHHS) and Buncombe County Health and Human Services announced these cases on January 6, 2026. The cases involve three siblings who recently traveled to Spartanburg County, South Carolina, where a rapidly expanding measles outbreak has been occurring since late 2025.
These children began to show symptoms approximately one to two weeks after visiting the measles outbreak area, which is about 100 miles away.
Additionally, a potential public exposure occurred when the infected children visited the Mission Hospital Emergency Department waiting room (509 Biltmore Ave., Asheville, NC 28801) between 2:00 a.m. and 6:30 a.m. on January 4, 2026.
About two dozen potentially exposed individuals have been contacted by health officials.
Furthermore, anyone who was present during that time and has not been reached should call Buncombe County Public Health at 828-250-6100 for guidance.
And measles vaccines are readily available at primary care providers, pharmacies, and the Buncombe County Immunization Clinic located at 40 Coxe Ave., Asheville.
Across North Carolina, five confirmed measles cases have been reported since late December 2025, including additional cases in Polk and Rutherford counties.
According to NCDHHS, as of January 14, 2026, all but one of these NC cases are linked to travel from the South Carolina outbreak.
The outbreak in Spartanburg County has surged dramatically.
As of January 13, 2026, the South Carolina Department of Public Health reported 434 cases, with a significant increase of 124 new cases in just a few days. Most cases involve unvaccinated children.
Throughout 2025, measles outbreaks were identified in Texas, Utah, and other states. As well as record-setting outbreaks in Canada and Mexico.
Numerious health agencies recommend ensuring you are immune to measles before visiting an outbreak area in 2026.
Mexican health authorities recently confirmed the country's first case of mpox caused by clade Ib of the monkeypox virus (MPXV).
According to a report from the World Health Organization (WHO) on January 9, 2026, the affected individual is an adult male with a recent travel history to Germany. He is currently receiving medical care in Mexico.
As of January 13, 2026, there is no indication of community transmission of MPXV in Mexico, but officials are monitoring the situation closely.
This case represents a significant development in Mexico's mpox surveillance efforts. Previous cases in the country since the global outbreak in 2022 have mainly involved clade II strains, primarily concentrated in urban areas such as Mexico City and Quintana Roo.
Clade Ib, a subclade of clade I, has emerged notably in Africa and is associated with more severe cases compared to clade II variants in specific contexts. Clade Ib is characterized by sustained human-to-human transmission and has been linked to more severe outcomes in vulnerable populations, such as those with weakened immune systems.
Historically, clade I has a higher case-fatality rate (3–11%) than clade II.
However, improved healthcare access in non-endemic countries has so far reduced fatalities among imported cases, according to the WHO.
The WHO has reported over 100 travel-associated clade Ib cases in high-income countries across Africa, Asia, Europe, the Middle East, North America, South America, and Australia, with no deaths recorded in these imported cases to date.
Since November 2024, there have been 10 reported cases of clade I mpox (Ib) in the United States. Most of these cases occurred in individuals who had recently traveled to areas with known clade I outbreaks or were linked to such travelers, such as household contacts.
This specific case highlights the ongoing global circulation of clade Ib and underscores the role of international travel in the spread of the virus.
Health authorities, including the WHO, state that the JYNNEOS® (MVA-BN®) vaccine offers cross-protection across different mpox clades because it is based on the vaccinia virus, which confers broad immunity to orthopoxviruses.
The WHO says vaccine protection is expected to be effective across clades, with no evidence of significant immune escape in clade Ib.
In the United States, JYNNEOS is offered at various clinics as of early 2026.

Health authorities in West Bengal, India, recently issued a high alert after detecting two suspected cases of the Nipah virus (NiV) at the Virus Research and Diagnostic Laboratory of the Indian Council of Medical Research at AIIMS Kalyani.
The cases, identified on January 11, 2026, involve two nurses who work at a private hospital in Barasat, in the North 24 Parganas district, near the Bangladesh border.
Both patients are in critical condition and are receiving ventilator support while in isolation, with preliminary tests indicating a possible Nipah infection.
According to local media reports on January 13, 2026, the Nipah virus was first identified during the 1998-1999 Malaysian outbreak and has since caused sporadic, high-fatality outbreaks in South Asia.
Previous confirmed NiV cases in India were reported from Kerala state, specifically in the districts of Malappuram and Palakkad, along India's southwest coast.
According to the WHO's Disease Outbreak News in August 2025, four confirmed NiV cases, including two deaths, were reported in 2025.
As of 2026, there is no licensed Nipah vaccine available for humans, but research has accelerated due to the virus's priority status from the World Health Organization.
Key vaccine candidates include subunit vaccines targeting the fusion protein that have shown promise in Phase I trials for safety and immune response.
The University of Oxford launched the world's first Phase II trial in late 2025.
Other research platforms are exploring mRNA and peptide-based approaches.
According to research, vaccine experts estimate that a licensed vaccine could be available within five years, says Don Hackett, publisher of Vax-Before-Travel.com.
In a significant step toward enhancing global epidemic preparedness, the Serum Institute of India (SII), today announced it has partnered with the University of Oxford under the support of the Coalition for Epidemic Preparedness Innovations (CEPI) to establish an investigational reserve of the ChAdOx1 RVF vaccine candidate against Rift Valley fever (RVF).
This is an essential initiative as no RVF vaccine is currently available.
Confirmed on January 13, 2026, SII will produce up to 100,000 doses of the investigational ChAdOx1 RVF vaccine. An initial 10,000 doses are earmarked for a potential upcoming clinical trial to evaluate the candidate's safety and immunogenicity in outbreak-affected regions. The remaining doses will be maintained as a strategic investigational reserve, ready for rapid deployment in future outbreaks to generate critical clinical data and accelerate evidence for licensure.
ChAdOx1 RVF has completed Phase I trials. Results published from the UK and Uganda trials show it was safe and generated an immune response. A CEPI-supported Phase II trial, separate from the upcoming planned trial, began in Kenya in 2025.
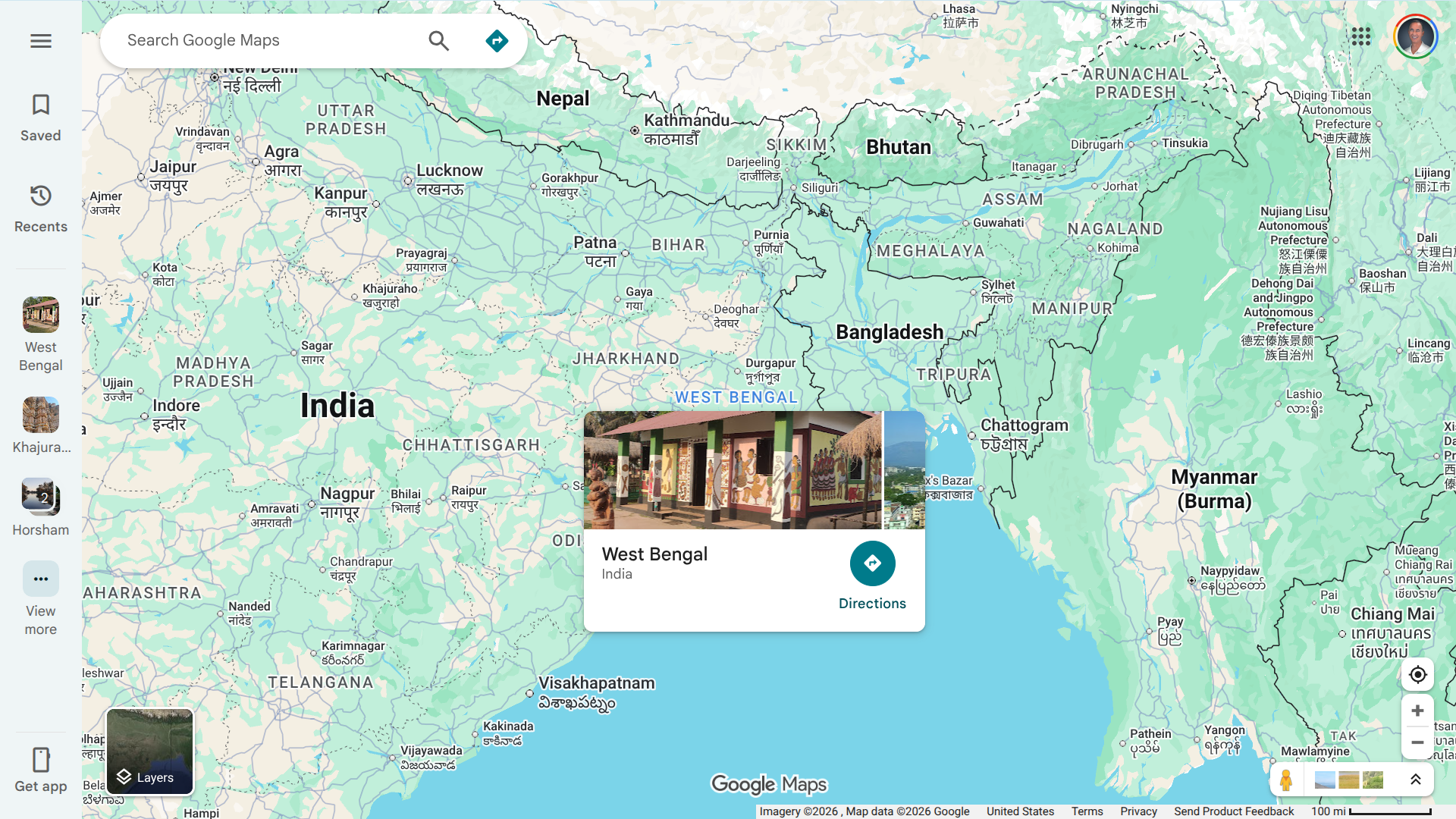
This collaboration arrives as a deadly RVF outbreak continues to impact communities in West Africa, particularly in Sénégal and Mauritania.
According to the latest available World Health Organization (WHO) data, DON584 from 2025, over 600 confirmed human cases, including 47 related deaths, have been reported.
Under the licensing agreement, CEPI is providing up to $3.5 million to support the development, manufacturing, and creation of a reserve for this RVF vaccine candidate.
In a press release, Dr. Umesh Shaligram, Executive Director at SII, added, "Producing these doses at speed is a necessary step to ensure that vaccine candidates like ChAdOx1 RVF can be delivered when and where they're most needed."
Rift Valley fever is a mosquito-borne zoonotic disease that primarily affects livestock but can spill over to humans through contact with infected animals or bites from infected mosquitoes. While most human cases are mild, severe infections can lead to brain inflammation, hemorrhagic fever, and death.
The disease also devastates pastoral communities by causing massive livestock losses, impacting livelihoods and food security in affected regions across Africa and the Middle East.
This collaboration exemplifies proactive "100 Days Mission" principles, positioning the world to respond more effectively to emerging infectious threats like RVF.
Until a vaccine becomes available, the U.S. CDC, WHO, and others recommend that travelers to affected areas follow guidance on mosquito prevention and avoiding contact with potentially infected animals.
The Texas Department of State Health Services (DSHS) today announced it is enhancing wildlife rabies prevention efforts in far West Texas.
Announced on January 13, 2026, the 32nd annual Oral Rabies Vaccination Program (ORVP) aims to protect both people and animals by vaccinating wildlife along the Texas-Mexico border.
Funded by the State of Texas and others, the ORVP has successfully reduced rabies cases significantly since it began over 30 years ago.
Last year, DSHS reported 19 confirmed positive rabies cases in animals from January to June 2025.
Additionally, the ORVP is now expanding to address the Arizona fox rabies variant from New Mexico, located 150 miles away.
Aerial bait distribution, a key component of the program, will begin from Alpine on January 16, 2026, and from Del Rio International Airport on January 21, weather permitting.
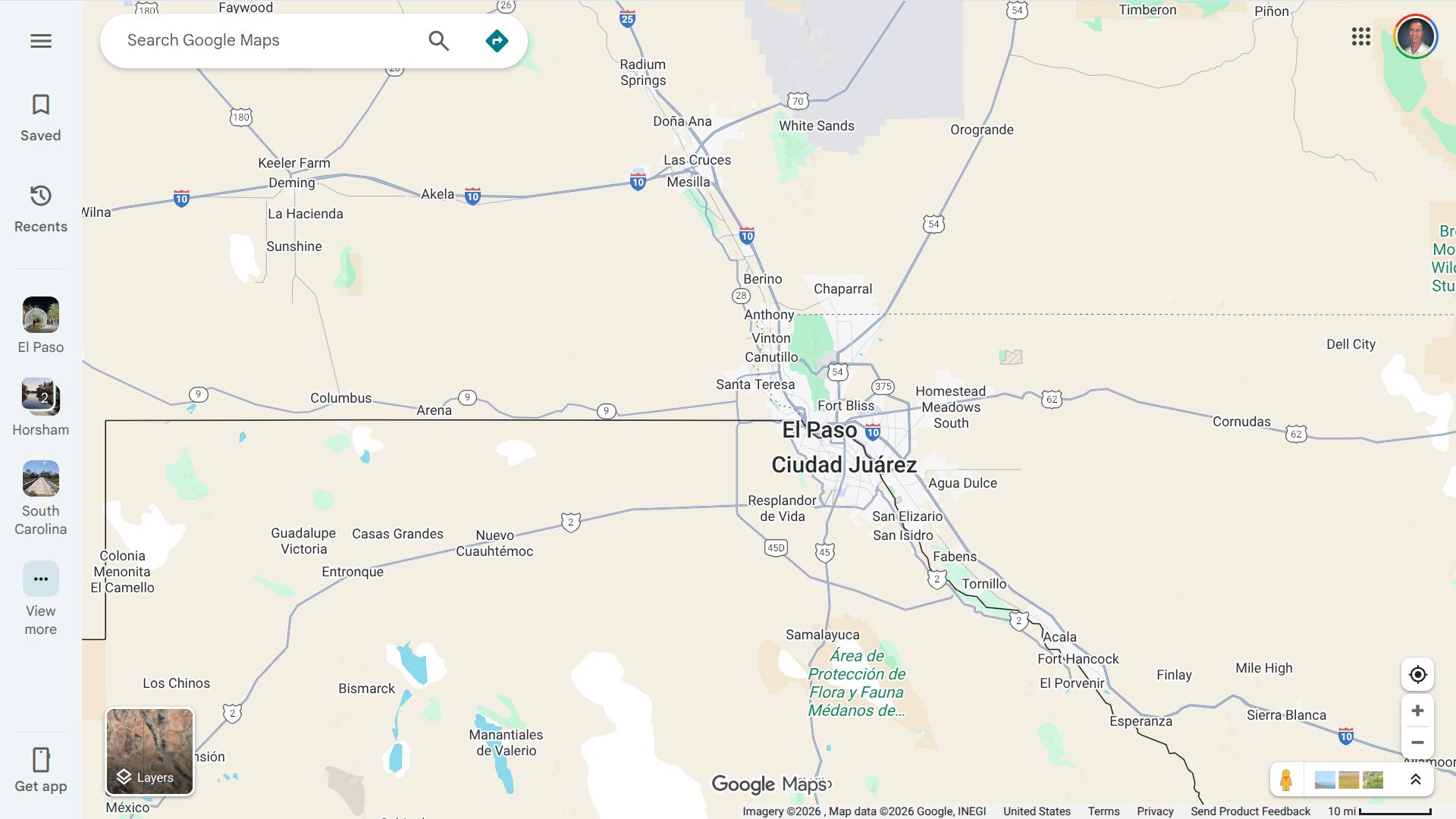
These efforts will occur in the Border Maintenance Zone, covering 19 counties, including El Paso, Hudspeth, and Cameron, as well as targeted urban areas around El Paso.
The vaccine baits, small plastic packets dipped in fish oil and fishmeal, are designed to attract wildlife like coyotes and foxes but pose no threat to pets or livestock.
The two-week operation will involve six to nine flights per day, dropping about 693,600 baits at a density of 50 baits per square mile. Residents should leave found baits undisturbed and can report any concerns to DSHS at 1-877-722-6725 or 512-776-7676.
DSHS continues to monitor all Texas counties for rabies outbreaks, such as red foxes near Austin, Texas.
For more information, visit the DSHS Oral Rabies Vaccination Program page.
Furthermore, rabies vaccines are available from various clinics throughout the United States.
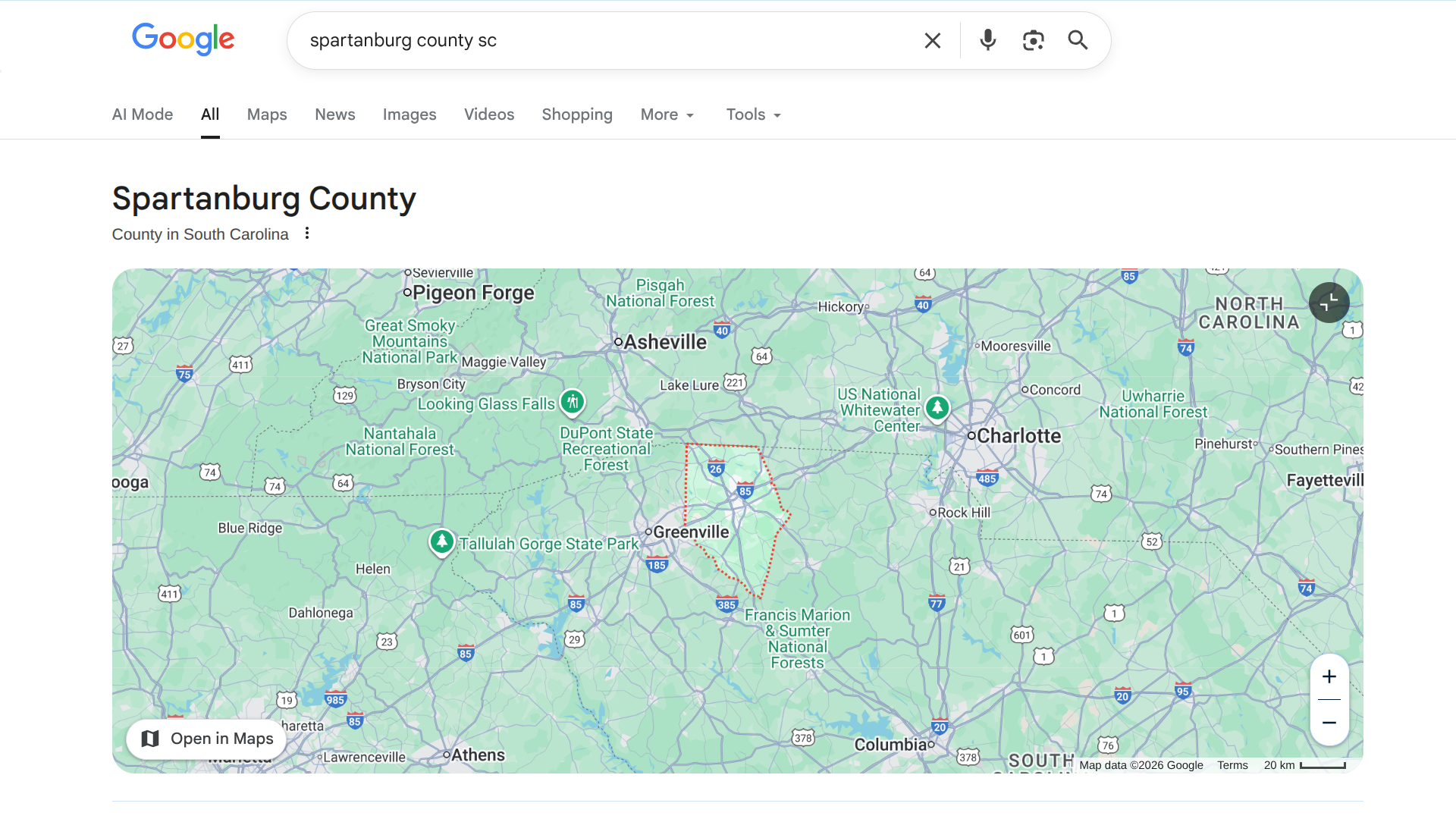
The South Carolina Department of Public Health (DPH) recently reconfirmed that an ongoing measles outbreak in the Upstate region during 2025 has extended into 2026.
On January 9, 2026, DPH reported 310 cases of measles in the current outbreak. Most of these measles cases were concentrated in Spartanburg County. Currently, about 256 infected people in SC were unvaccinated before infection.
DPH wrote Some cases are travel-related exposures or close contacts of known cases. Other cases have no identified source, suggesting that measles is circulating in the community and could spread further.
DPH has identified new public exposures at Jesse S. Bobo Elementary and began notifying potentially exposed students, faculty, and staff on December 30, 2025. Students from Jesse S. Bobo Elementary, Sugar Ridge Elementary, and Boiling Springs Elementary who quarantine successfully without becoming ill are scheduled to return to classes on January 10, 2026.
Separately, North Carolina has reported measles cases in early 2026 linked to travel/exposure in South Carolina, including three in Buncombe County, which is located north of Spartanburg County, SC.
As of January 12, 2026, measles vaccines are available at many primary care provider offices and pharmacies, as well as DPH Health Departments and the Buncombe County Immunizations Clinic at 40 Coxe Avenue, Asheville, NC.
Both county health departments say vaccination remains the best way to prevent measles and stop these outbreaks
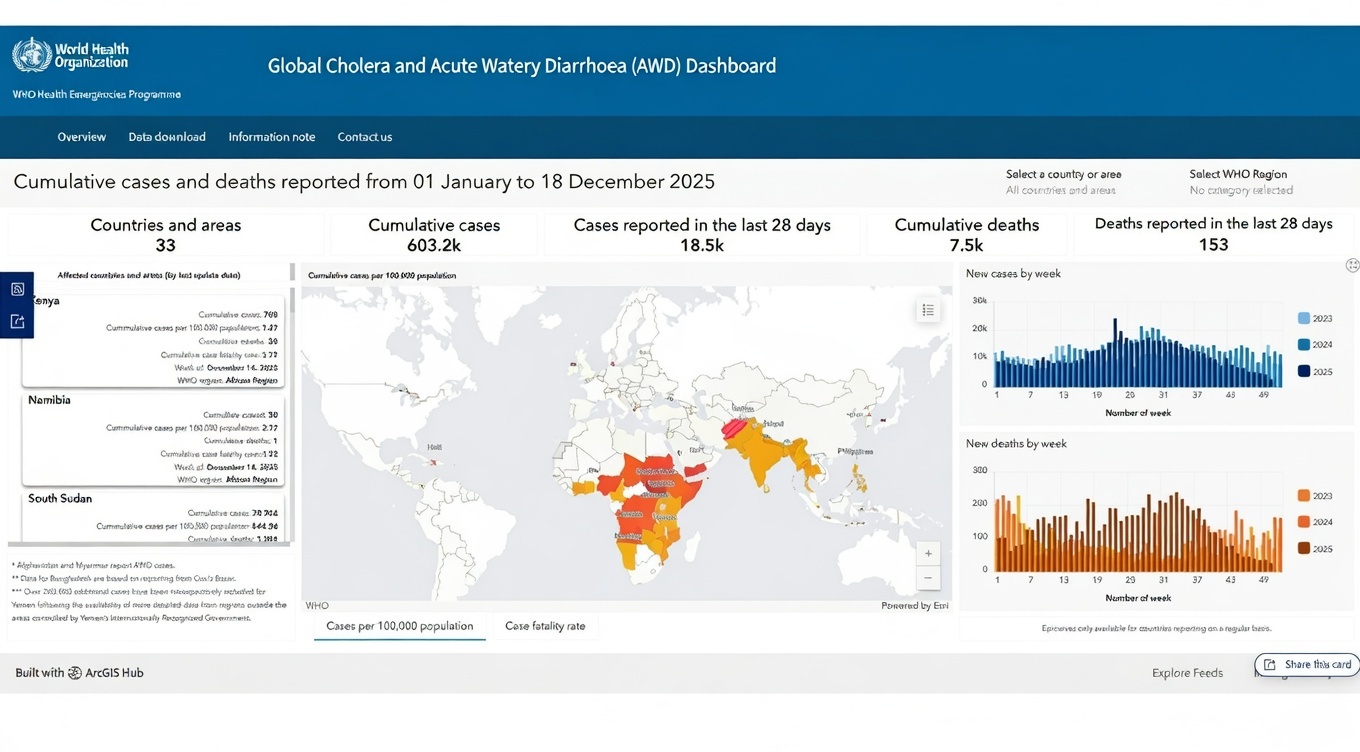
As we enter 2026, cholera outbreaks continue to be a significant global threat, despite decades of progress in controlling this severe disease.
The World Health Organization (WHO) has recorded seven cholera pandemics in the last two centuries, with the current seventh pandemic beginning in South Asia in 1961 and still ongoing.
As of December 31, 2025, the WHO has reported a total of 601,845 cholera cases and 7,671 deaths across 31 countries in Africa, Asia, the Middle East, and the Americas.
These numbers indicate persistent transmission across various WHO regions, particularly in the Eastern Mediterranean and African regions, which bear the most significant burden.
Preliminary data for 2025 suggest that case numbers and mortality rates are higher than in previous years. In lower-risk areas, such as the EU/EEA, cholera is uncommon and typically linked to travel from endemic regions.
According to the European Centre for Disease Prevention and Control (ECDC) on January 9, 2026, only 12 confirmed cases were reported across five EU/EEA countries in 2023.
This is a slight increase from 29 cases in 2022, two cases in 2021, and none in 2020.
All recent cases involved individuals with a travel history to affected areas. The WHO highlights vaccination as a crucial strategy in controlling cholera, especially for high-risk groups.
Oral cholera vaccines (OCVs), including WHO-prequalified options like Dukoral®, Euvichol-Plus®, and Euvichol-S®, are recommended for travelers at higher risk of exposure. Travelers planning to visit regions with active cholera transmission should consult travel health clinics to evaluate their personal risk factors and determine if an OCV immunization is appropriate in 2026.
When departing from the USA, OCVs are available at travel clinics in most states.
However, the WHO noted that increased OCV production has yet to meet growing global needs.