Search API
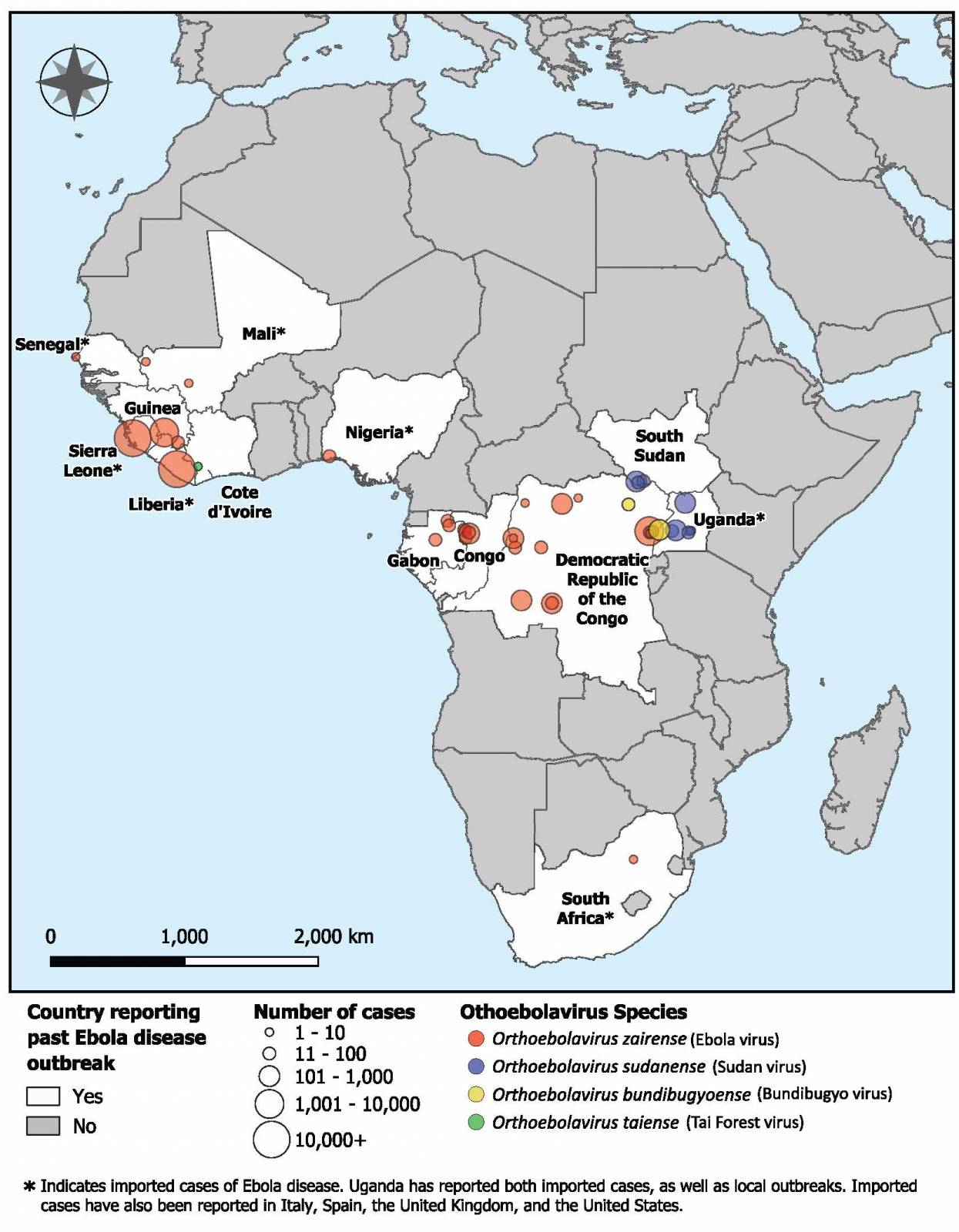
Ebola disease was first identified in 1976. Since then, outbreaks have emerged periodically and infected people in several African countries
A vaccine that has already received approval and is stockpiled for use against the Zaire ebolavirus could become more affordable and easier to deploy in low-resource settings due to a new collaboration between CEPI and MSD.
With the support of up to $30 million in funding from CEPI, MSD, also known as Merck in the United States and Canada, will utilize Hilleman Laboratories—a joint venture between MSD and Wellcome—to develop the Ervebo® Ebola vaccine with an updated manufacturing process.
Furthermore, SK bioscience and IDT Biologika will collaborate to develop the updated drug-substance process and the associated drug product.
"In a single decade the world has transformed Ebola from a global emergency to a disease that can be stopped in its tracks – and now CEPI's support will help to enable a sustainable and accessible supply of MSD's Zaire ebolavirus vaccine for years to come at a more affordable price," explains Dr. Richard Hatchett, CEO of CEPI, in a press release on January 21, 2026.
"This deal brings together longstanding partners of CEPI with longstanding partners of MSD to boost global defences against one of the deadliest pathogens known to humankind, helping to save lives."
The existing MSD Zaire ebolavirus vaccine was developed in the midst of the 2014-16 West African Ebola crisis. Its manufacturing process is complex and potentially vulnerable to supply disruptions, making the vaccine expensive to produce and difficult to scale up. In addition, the vaccine must be stored in freezers at ultra-low temperatures of -70 degrees Celsius, creating substantial logistical challenges in the often remote, low-resource settings where Ebola outbreaks typically occur.
Currently, the Merck Ebola vaccine is not commercially available in the United States, but is maintained in the U.S. Strategic National Stockpile.
Florida health authorities have confirmed the first locally acquired case of chikungunya in 2026, occurring in Miami-Dade County.
This incident underscores the ongoing risks posed by this mosquito-borne virus, particularly given the significant number of travel-related cases.
For example, just a few days ago, thousands of travelers visited Miami-Dade to attend the NCAA football championship game.
As of January 17, 2026, the Florida Department of Health (DOH) reported 16 cases of chikungunya associated with travel. In the previous year, the state recorded 328 cases, with Miami-Dade County accounting for 214, significantly surpassing all other areas.
The vast majority (316) of Florida's travel-related cases originated from travelers returning from Cuba, where the virus has surged dramatically.
Cuba has reported more than 50,000 chikungunya cases in 2025, with all 15 provinces affected.
Nationally, the U.S. Centers for Disease Control and Prevention (CDC) has seen more than 400 chikungunya cases in 2025, with all but the two locally acquired instances linked to travel.
The CDC says the chikungunya virus, transmitted primarily by Aedes mosquitoes, causes symptoms in most infected individuals within 3–7 days of a bite. Common signs include high fever and severe joint pain, often debilitating. While the majority recover within a week, some experience persistent or chronic joint pain lasting months or even years.
As of January 21, 2026, there is no specific antiviral treatment for chikungunya.
However, a chikungunya vaccine has been approved by the U.S. FDA and is commercially available at travel clinics across the United States and in various other countries.
The CDC urges travelers to consult healthcare providers or travel medicine specialists before trips to affected destinations, use insect repellents, wear protective clothing, and eliminate standing water to reduce mosquito breeding.
Visitors to and residents in areas like South Florida should also maintain vigilance against local vectors, especially given the presence of competent Aedes mosquitoes.

Researchers at The University of Texas at El Paso (UTEP) have discovered unusually high levels of parasitic infection in the insects that transmit Chagas disease along the U.S.-Mexico border.
The kissing bugs were collected near homes and natural areas, raising concerns about the potential for local transmission of the disease in the Southwestern United States.
Led by Dr. Rosa Maldonado, a professor of biological sciences, the research team found that the infection prevalence in kissing bugs has significantly increased, from 63.3% in a study conducted in 2021 to 88.5% in the current study.
This alarming trend indicates that T. cruzi infections, which cause Chagas disease, are on the rise. It remains alongside dengue fever as one of the neglected diseases with the highest burden in the Americas.
"The infection prevalence in kissing bugs has risen significantly, indicating a marked upward trend for T. cruzi infections," Dr. Maldonado mentioned in a press release on January 6, 2026.
For this study, researchers collected kissing bugs from various locations, including El Paso County, Texas, and southern New Mexico, as well as from Franklin Mountains State Park, Central El Paso, and Canutillo.
The findings from this study suggest that Chagas disease is becoming an increasing public health issue in the Southwest. In this region, the disease was previously confined primarily to Latin America.
However, in 2025, Texas reported 22 human cases of Chagas disease, both imported and locally acquired.
Chagas disease is caused by the parasite Trypanosoma cruzi (T. cruzi) and is primarily spread by blood-feeding insects known as kissing bugs. This debilitating illness affects approximately 6 million people worldwide and can persist for decades in its chronic phase, often without symptoms.
However, it can lead to severe heart and gastrointestinal complications over time.
Previously, on March 20, 2025, researchers at UTEP successfully tested a faster, more sensitive, and more reliable method for diagnosing Chagas disease.
Furthermore, according to the World Health Organization, no approved vaccine exists to prevent Chagas disease. However, vaccine development is ongoing in 2026.
The current study, published in the journal Epidemiology & Infection, was conducted in collaboration with Dr. Priscila S. G. Farani, an assistant professor in UTEP's School of Pharmacy.
The Taiwan Centers for Disease Control (TCDC) continues to report a significant increase in acute viral hepatitis A (HepA) cases.
The surge that began in spring 2025 has led to the highest number of reported cases in nearly a decade and remains unabated as of January 20, 2026.
Official surveillance data from the Taiwan National Infectious Disease Statistics System show that there were a total of 484 confirmed acute HepA cases for the entire year of 2025.
Demographic data from this period reveal that approximately 82.8% of cases occurred in males, with the highest proportion (36.2%) among adults aged 30-39 years. Additionally, 40% of the cases in 2025 were linked to sexual contact.
In the early weeks of 2026, an additional 22 cases have already been recorded, with individuals aged 20-29 accounting for about 28% of these cases.
Historical outbreaks show HepA cases concentrated in northern Taiwan, spreading southward over time.
The TCDC has emphasized that hepatitis A antibody prevalence in Taiwan remains notably low, particularly among younger and middle-aged adults. This leaves large segments of the population vulnerable and contributes to the continued spread of the virus, which spreads through the fecal-oral route, often via contaminated food or water, or through close contact with infected individuals.
For real-time statistics and official guidance, please refer to the Taiwan National Infectious Disease Statistics System or the CDC website. It is crucial to maintain vigilance regarding hygiene, safe practices, and vaccination to mitigate this ongoing increase in cases.
According to the U.S. CDC, unvaccinated travelers aged 1 year or older traveling to Taiwan should speak with a healthcare provider about pre-trip vaccinations. Travelers allergic to a vaccine component should receive a single dose of immune globulin, which provides adequate protection for up to 2 months, depending on the dosage given.
Unvaccinated travelers who are over 40 years old, are immunocompromised, or have chronic medical conditions planning to depart for a risk area in less than 2 weeks should receive the initial dose of vaccine and, at the same appointment, receive immune globulin, says the U.S. CDC.
These HepA immunization services are offered at travel clinics in the United States in 2026.
Clemson University officials were recently informed by the South Carolina Department of Public Health (DPH) of a confirmed case of measles in an individual affiliated with the University.
As of January 17, 2026, the individual has isolated per DPH requirements, and DPH is conducting contact tracing with individuals who may have been exposed and outlining isolation and quarantine protocols.
DPH will provide email notifications to any individuals identified as possible contacts who may need to quarantine. A person infected with measles is contagious for 4 days before and after the rash begins. Isolation of an actively infectious case lasts until four full days after the onset of the rash, and DPH determines the dates of isolation.
If a person without documented immunity receives a dose of the MMR vaccine within 72 hours after the last exposure, they do not have to quarantine.
According to the most recent data from Student Health Services, nearly 98% of main campus Clemson students have provided proof of immunity. Recent data indicated that about 30,000 students and staff were affiliated with the University in 2026.
Statewide, South Carolina is experiencing a significant measles outbreak, with the total reaching 558 cases as of January 19, 2026, centered in Spartanburg County and extending into North Carolina in cities such as Asheville.

As the 2025-2026 influenza season continues, recent data from the U.S. Centers for Disease Control and Prevention (CDC) indicate a significant decrease in flu activity nationwide.
The CDC's assessment framework classifies the current flu season as moderate across all age groups. Typically, flu seasons in the United States peak between December and February, sometimes extending into March or later.
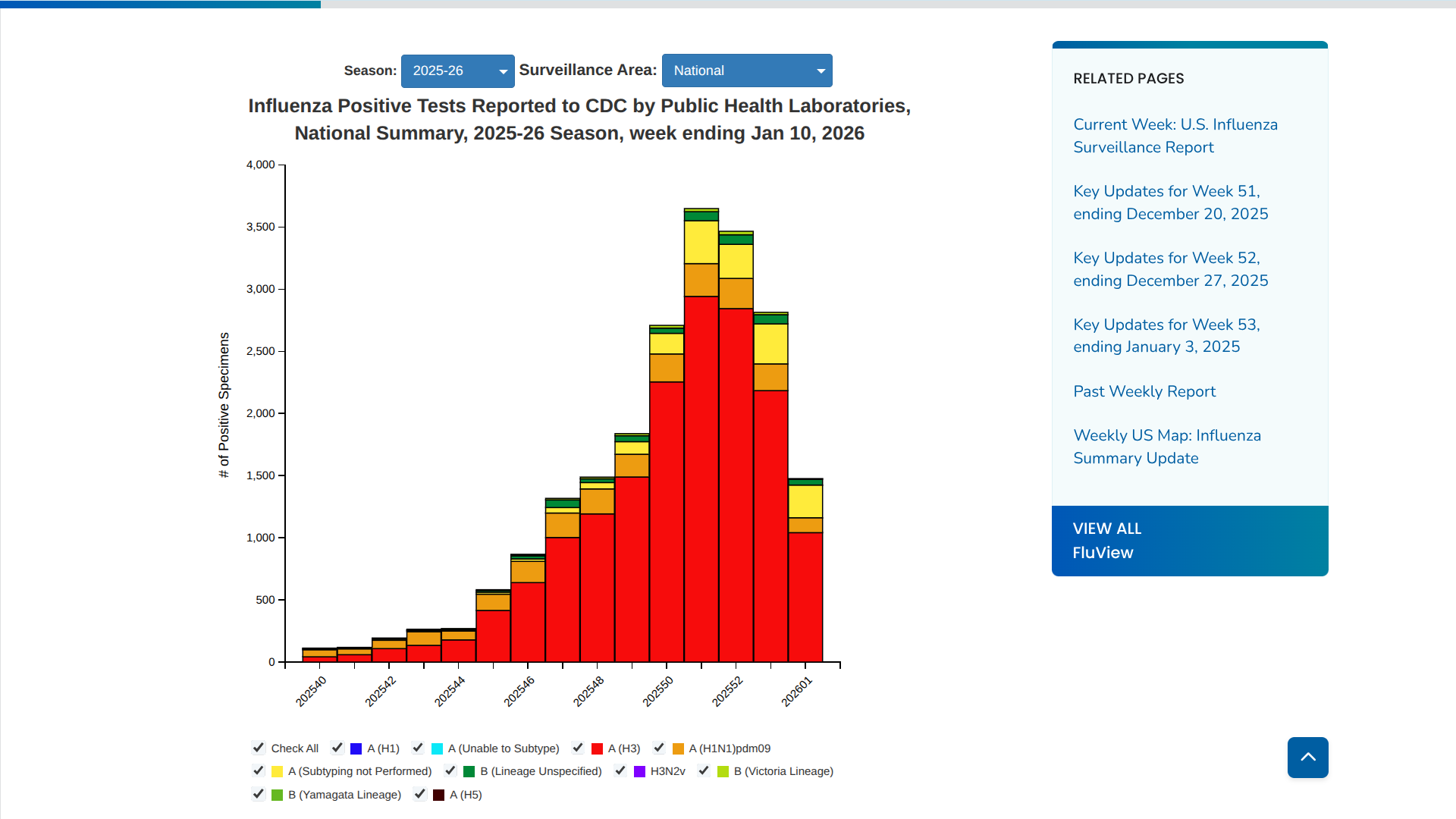
According to the CDC's latest FluView report for the week ending January 10, 2026, the number of confirmed positive influenza tests dropped by approximately 31% compared to the previous week.
This decline marks the most significant drop observed in this season so far.
The flu test positivity rate dropped to approximately 24.7% for the week ending January 3, down from 32.9% the previous week. However, experts caution that this could partially reflect reduced healthcare visits during the holidays rather than an actual peak.
Influenza A(H3N2) viruses are the most frequently reported influenza viruses so far this season.
These data mark a shift from earlier reports, such as the week ending December 27, 2025, when activity was still increasing throughout the USA.
The CDC also reported on January 19, 2026, that about 130 million flu shots had been distributed to clinics and pharmacies this flu season. Furthermore, these healthcare providers continue to recommend and offer flu shot services.
Indian health officials in western Madhya Pradesh recently implemented emergency measures in response to a localized outbreak of Guillain-Barré Syndrome (GBS), a rare autoimmune neurological disorder.
As of January 18, 2026, The Hindu reports indicate that there are at least 14 to 18 cases of GBS (confirmed and suspected), many of which are among children and adolescents.
Two people have died in 2026, and others were on life support, with some cases requiring ICU treatment.
Additional cases have been reported in adults, including a recent diagnosis in Neemuch city.
In response to the outbreak, two rounds of door-to-door screenings have been conducted by Health Department teams, ASHA workers, and Anganwadi staff to identify potential new cases early.
During early January 2026, Deputy Chief Minister Rajendra Shukla, who oversees health, visited the area to inspect preparations and directed that the government cover the treatment costs, including critical medications and injections.
Financial aid is also being provided to some families through the Red Cross Society.
GBS is often triggered by preceding infections, such as Campylobacter jejuni (which can result from contaminated food or water), respiratory viruses, or other pathogens; however, the exact cause of this current cluster is still under investigation.
Past GBS clusters in India have occasionally been linked to water quality issues or acute diarrheal diseases.
In 2025, Pune, Maharashtra, reported over 200 cases and 23 deaths linked to contaminated water sources.
For international travelers to India, including Madhya Pradesh, major health authorities such as the U.S. CDC have not issued specific travel health advisories related to this GBS outbreak. Standard travel vaccinations (hepatitis A, typhoid, and rabies, if at risk) are still recommended for travel to India in 2026.
Currently, there is no vaccine available to prevent Guillain-Barré Syndrome.