Search API
With the continued overlapping incidence of chikungunya, dengue, malaria, Zika, and yellow fever diseases in the Region of the Americas, should international travelers be vaccinated before arriving in Florida?
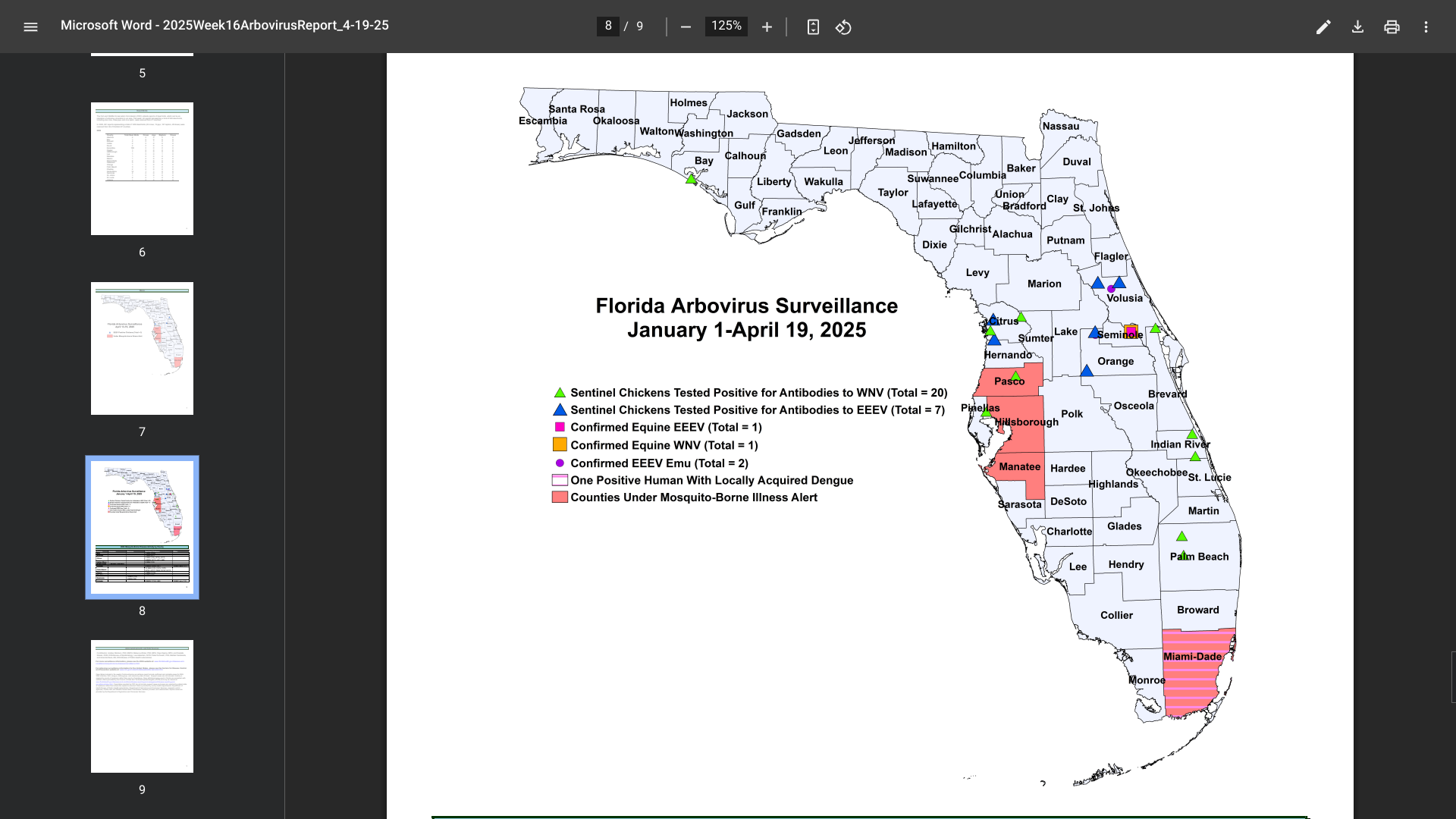
According to the Florida Department of Health (FDH) Arbovirus Surveillance update #16, dated April 19, 2025, numerous travelers have been diagnosed with vaccine-preventable diseases this year, particularly in the Miami and Tampa areas.
To notify people of these health risks, FDH has missed alerts for Hillsborough, Manatee, Miami, and Pasco counties in 2025.
For example, last year, 11 instances of chikungunya were reported in individuals with a travel history to Brazil (five), India (five), and Pakistan.
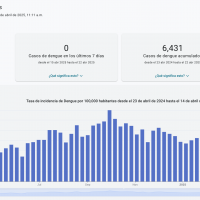
In 2025, sixty cases of dengue fever had already been reported among individuals who had traveled internationally, and one locally acquired case of dengue (DEN-3). During 2024, 1,016 travel-associated dengue cases were reported, primarily among visitors from Brazil, Cuba (567), and Puerto Rico.
Furthermore, 91 locally acquired dengue cases were reported from ten counties, including Miami-Dade (50), in 2024.
In 2024, 72 cases of travel-related malaria were reported in individuals with a history of visiting malaria-endemic areas, such as Africa (Nigeria) and Central and South America.
As of April 25, 2025, the U.S. Centers for Disease Control and Prevention, Canada Health, and the United Kingdom have not issued vaccination requirements for visiting Florida.
Of these mosquito-transmitted diseases, chikungunya vaccines are commercially available at most pharmacies and are recommended by various health agencies.
Merck today announced its financial results for the first quarter of 2025, which included a significant decrease in sales of its cancer prevention human papillomavirus (HPV) vaccines.
On April 24, 2025, Merck reported that GARDASIL/GARDASIL 9 vaccine sales declined 41% to $1.3 billion in 2025.
This decline is primarily due to lower demand in China, partially offset by higher demand in most international regions, particularly in Japan, as well as higher pricing and demand in the U.S.'
'Excluding China, sales grew 14%, or 16% excluding the impact of foreign exchange.'
Robert M. Davis, chairman and chief executive officer, Merck, commented in a press release, “We are working with focus and urgency to both realize the full potential of our near-term opportunities and to rapidly progress the next wave of innovation that will positively impact the lives of patients and drive future value creation for all of our stakeholders.”
The GARDASIL-9 vaccine remains the leading HPV vaccine in the United States, recommended by the U.S. CDC, and offered at most pharmacies.

Novavax Inc. recently stated that it believes its Biologics License Application for its protein-based COVID-19 vaccine is approvable, based on conversations with the U.S. Food and Drug Administration (FDA) from April 1, 2025, to April 24, 2025.
In its press release on April 23, 2025, Novavax announced that the FDA had issued a formal request, asking the company to provide a postmarketing commitment to generate “additional clinical data” for the COVID-19 vaccine.
'We look forward to engaging with the FDA expeditiously to address the PMC request and move to approval as soon as possible,' concluded Novavax.
The World Health Organization granted the Novavax COVID-19 vaccine (Nuvaxovid™) Emergency Use Listing on December 17, 2021, and it remains available today in numerous countries.

Belize's Ministry of Health & Wellness recently announced the confirmation of locally transmitted malaria cases in Santa Elena Town and Cristo Rey Village, located in the Cayo District.
The initial case was detected on January 17, with additional instances detected on March 11 and April 5, 2025. Of the four confirmed cases in 2025, three are attributed to local transmission, and one is classified as imported from Guatemala.
Enhanced surveillance has been activated along the Western border, particularly in the Santa Elena neighborhoods of Santa Cruz and Hillview, as well as Cristo Rey Village.
As of April 23, 2025, these malaria cases mark the first instances of local transmission in over six years. However, other Central American countries have also reported outbreaks of malaria.
While this development is of concern, Belize remains committed to maintaining its malaria-free status, which was first achieved in June 2023.
The Ministry says malaria is a febrile disease caused by parasites, transmitted to humans through the bite of an infected Anopheles mosquito. Infected individuals typically experience episodes of fever with chills and sweating every 48 to 72 hours.
The Ministry is actively testing individuals who currently have, or recently had, a fever within the past 30 days.
While malaria cases may continue in April, vaccines are not yet offered in the Region of the Americas.

According to recent travel advisories issued by the United States government, visiting the Republic of Uganda is not encouraged in 2025.
On April 23, 2025, the U.S. Department of State reissued a Level 3: Reconsider Travel advisory for Uganda, a country home to approximately 32 million people, located in East Africa.
The State Department says visitors should exercise increased caution in Uganda due to security risks and ongoing civil unrest.
If you decide to travel to Uganda, enroll in the Smart Traveler Enrollment Program to receive alerts from the U.S. Embassy in Kampala. This free digital offering makes it easier to locate people in an emergency.
From a health perspective, Uganda is confronting multiple disease outbreaks in 2025.
Building on sustained U.S. technical support to Uganda since the first day of the 2025 Ebola outbreak, the United States government has now donated 100 vials of monoclonal antibodies (mAb) to the Uganda Ministry of Health. As a breakthrough in medical science, mAb has the potential to significantly improve survival rates in patients with Ebola.
U.S. Ambassador Popp stated in a press release on April 14, 2025, “Providing these groundbreaking treatments demonstrates the United States’ strong commitment to innovation, scientific excellence, shared prosperity, and global health security. As we respond to outbreaks like Ebola, we build stronger global health security partnerships that benefit us all.”
And will enhance the ongoing efforts to end the current Sudan Ebola Virus outbreak in Uganda, as no vaccines or therapeutics have been approved for the prevention or treatment of SVD in 2025.
Additionally, the CDC has included Uganda in its polio and mpox travel advisories and recommends pre-arrival vaccinations for several routine and travel-related diseases, such as yellow fever, mpox, and cholera.
Travel vaccines for these diseases are commercially available at clinics and pharmacies in the United States.
When the World Health Organization announced polio outbreaks remained a Public Health Emergency of International Concern for an additional three months, other health agencies reacted with more detailed disclosures focused on outbreaks.
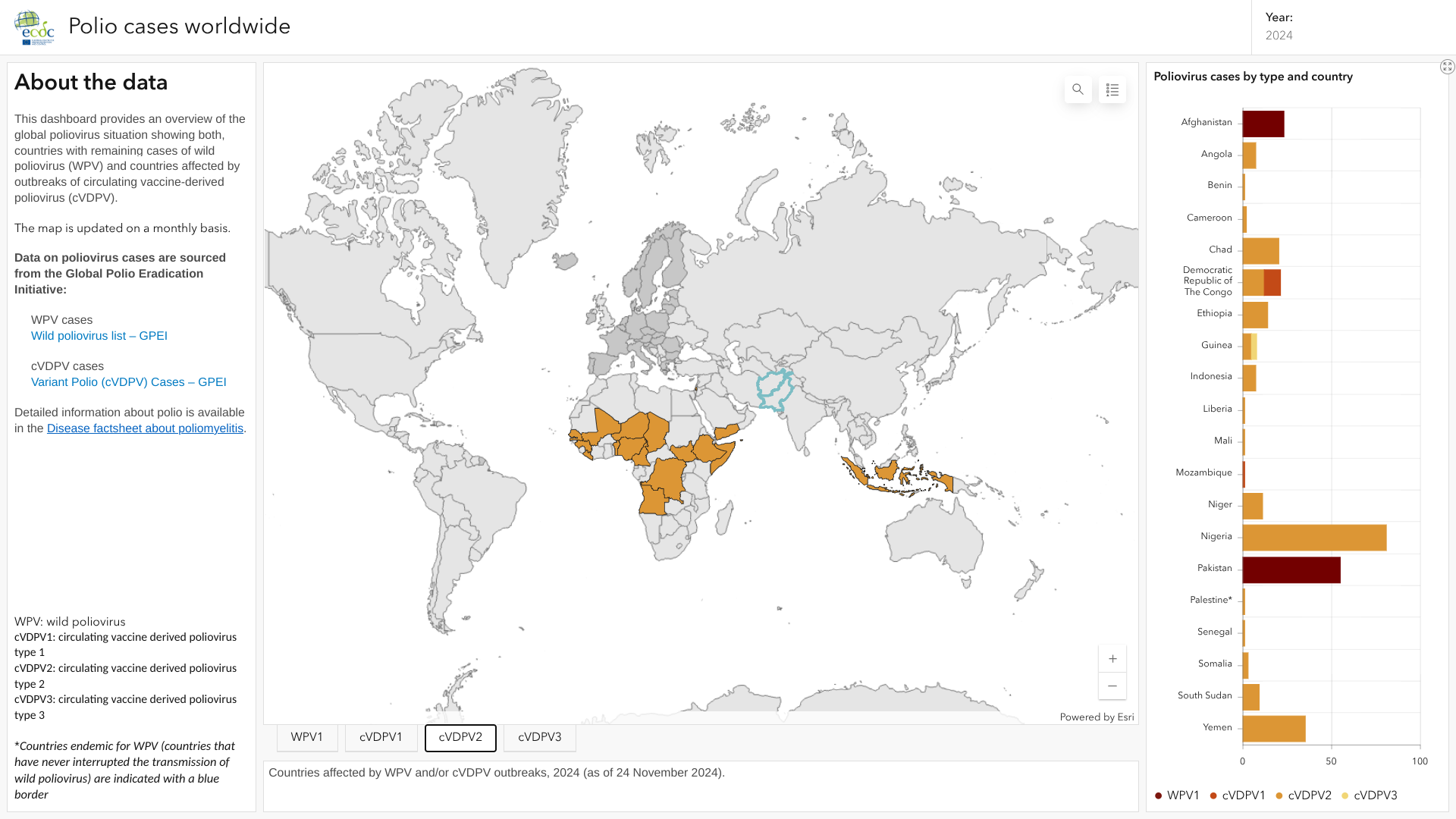
For example, on April 22, 2025, the U.S. Centers for Disease Control and Prevention (CDC) reissued a Global Polio Alert—Level 2 advisory regarding polio outbreaks and poliovirus detections in 38 countries.
And the updated European CDC dashboard revealed that vaccine-derived poliovirus type 2 (cVDPV2) was detected in wastewater systems of 14 cities across five European countries in 2025.
In the Pacific Ocean area, a cVDPV2 environmental sample has been reported from Lae City, Morobe Province, Papua New Guinea. Located north of Australia, this country previously confirmed that poliovirus was circulating in 2018.
Both the CDC and ECDC recommend that adults who have previously completed the routine polio vaccine series receive a single, lifetime booster dose of polio vaccine before traveling to any destination listed.
In the United States, the IPV polio vaccine is available for purchase at most pharmacies.
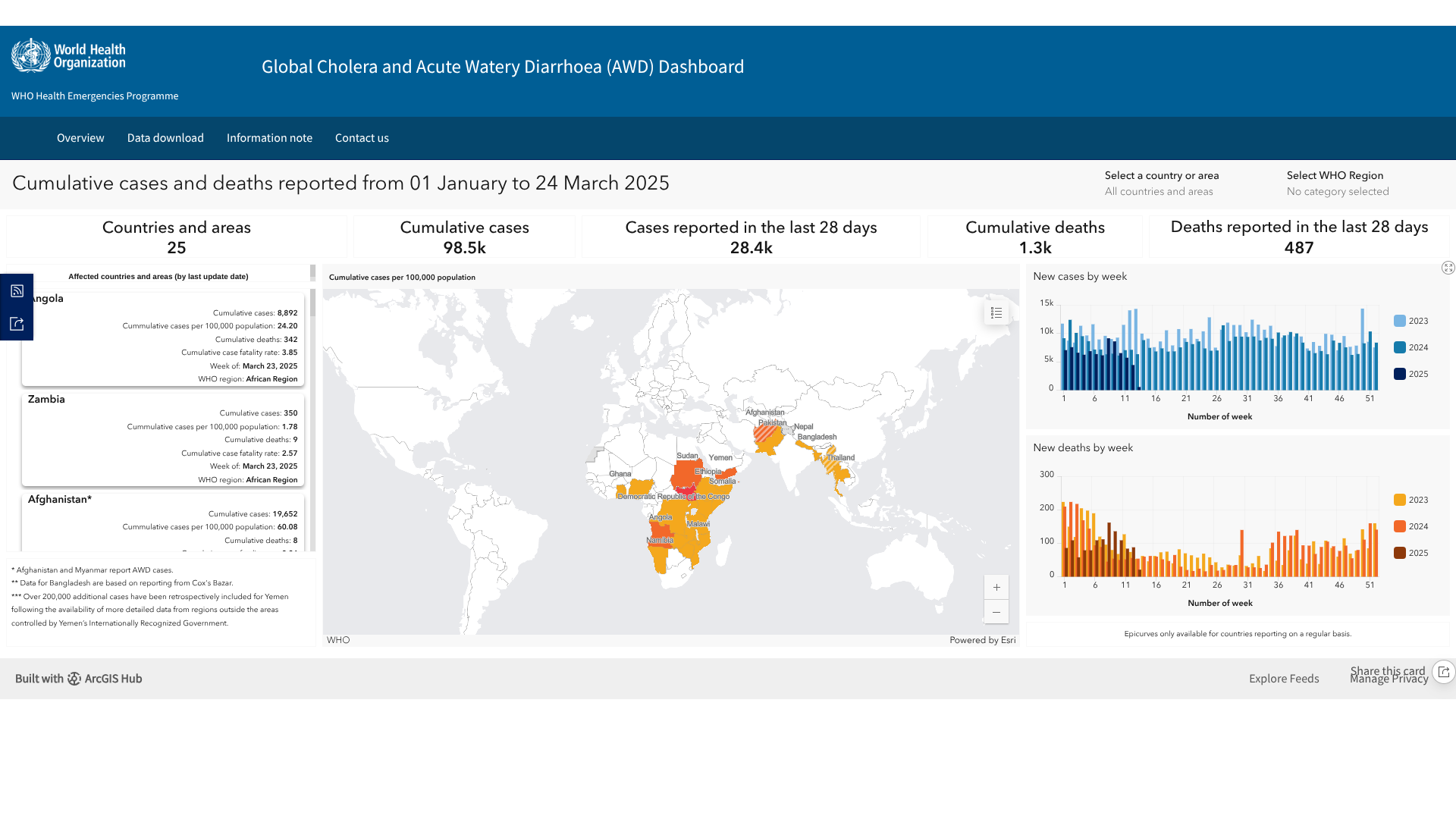
The World Health Organization (WHO) recently published its multi-country outbreak of cholera, external situation report #25, which indicates various countries continue to be severely impacted by this vaccine-preventable disease.
This year, a cumulative total of 116,574 cholera cases and 1,514 deaths were reported from 25 countries across three WHO regions, with the African Region recording the highest numbers.
In the Caribbean, a total of 1,298 suspected cholera cases, including 19 deaths, were reported in Haiti, especially noted in Cité Soleil and Arcahaie.
In March 2025, 517 cholera-related deaths occurred, highlighting an 8% decrease from February 2025.
The WHO announced some positive news on April 22, 2025: the average stockpile of Oral Cholera Vaccine (OCV) had stabilized at 5.2 million doses, marking the fourth consecutive month with a stockpile above the five million doses needed for the emergency stockpile at all times.
However, the growing global demand continues to exceed supply, hindering efforts to control cholera outbreaks, respond rapidly to the disease’s spread, and implement preventive campaigns, according to the WHO.
In the United States, travel clinics and pharmacies currently have ample access to OCVs.