Search API
Measles outbreaks have been reported in Europe this year, and cases in England have significantly increased in the first few weeks of 2026.
The UK Health Security Agency (UKHSA) reported on February 19, 2026, that 130 laboratory-confirmed cases of measles had been reported this year.
This indicates a rise of 34 cases since the previous weekly update.
The surge is primarily driven by an ongoing outbreak in North London, particularly in the borough of Enfield, where 50 cases have been confirmed. Nearby Haringey has recorded 10 cases, while Birmingham in the West Midlands accounts for 23 cases, making up 18% of the total.
Overall, 68% of cases are concentrated in London.
Fortunately, no measles-related deaths have been reported in 2026.
UKHSA officials urge parents and guardians to check their children's vaccination status and ensure they receive the MMR vaccine. Catch-up vaccination campaigns are currently underway, and vaccination records are published quarterly in health protection reports. Public health leaders emphasize that vaccination remains the most effective way to prevent the spread of measles.
With over 20 million annual visitors to London, this UKHSA advice is essential.
As measles cases continue to rise in London, visitors to these areas of England should consult a healthcare professional about vaccination options.
As a frequent airplane traveler who is concerned about catching the flu in crowded cabins with recycled air, I am thrilled by recent research on CR9114. This nasal spray delivers influenza-specific monoclonal antibodies.
This innovative product, if approved, could be ideal for high-exposure situations, such as flying during flu season, especially for travelers.
While modern commercial airplanes have high-efficiency ventilation systems that recirculate cabin air through HEPA filters, they don't remove all viruses. If an infected passenger is on board—especially one who is coughing or sneezing—flu virus transmission can still occur.
Published on February 4, 2026, in Science Translational Medicine, the study, led by researchers from Harvard in collaboration with Leyden Laboratories, demonstrates that the intranasal delivery of the broad-spectrum hemagglutinin antibody CR9114 is both safe and feasible.
In two Phase 1 trials involving healthy volunteers, repeated dosing (twice daily, given the approximately 3-hour nasal half-life) achieved sustained high antibody levels in the nose.
These recovered antibodies retained strong neutralizing activity against both influenza A and B viruses.
Importantly, this nasal spray is not a replacement for seasonal influenza vaccines but rather may offer a rapid, short-term immunoprophylactic option. By delivering high local antibody concentrations directly at the virus's entry point with small doses, CR9114 has shown early promise.
Previous preclinical tests on mice and macaques confirmed its safety and effectiveness against influenza challenge.
These findings lay the groundwork for future efficacy trials and provide proof of concept that intranasal antibodies can elicit efficacious passive immunity against influenza viruses.
For someone who dreads having post-flight sniffles turn into illness, this nasal spray could become an essential item to carry on board before flying.

As Spring Break 2026 travel plans are being finalized, many travelers are taking steps to avoid mosquito-borne diseases like Dengue fever.
To help inform these decisions, the U.S. Centers for Disease Control and Prevention (CDC) has recently updated its global Dengue travel health notice. This notice highlights elevated risks in 15 countries across Asia, the Americas, Africa, and the Pacific.
The updated Level 1 Travel Health Notice, revised on February 17, 2026, reaffirms existing alerts. It emphasizes that Dengue remains a year-round threat in many tropical and subtropical regions.
According to the CDC, the following countries pose elevated risks for travelers: Afghanistan, Bangladesh, Colombia, the Cook Islands, Cuba, Guyana, the Maldives, Mali, Mauritania, New Caledonia, Pakistan, Samoa, Sudan, Timor-Leste, and Vietnam.
And the unfortunate leader in the Americas remains Brazil, which has reported over 133,00 cases and 8 related fatalities since the start of 2026.
In the United States, most Dengue cases are travel-associated. However, some limited local transmission has occurred in states such as Florida, Texas, and California, where mosquitoes capable of spreading the virus are present.
So far in 2026, Florida has reported travel-related Dengue cases.
In 2025, 429 cases of Dengue were reported among individuals who had traveled internationally, with 311 cases among travelers from Cuba and 21 from Puerto Rico.
Additionally, 62 locally acquired Dengue cases were reported along Florida's east coast in 2025.
Preventive measures are crucial for anyone traveling to or living in at-risk areas, says the CDC.
As of February 19, 2026, Dengue vaccination options for U.S. travelers remain limited.
The only Dengue vaccine previously approved in the United States, Dengvaxia, has been discontinued due to low global demand, with remaining supplies in places like Puerto Rico set to expire by mid-2026.
Internationally, other vaccines, such as Qdenga, are authorized in over 40 countries, and newer approved vaccines, like Brazil's single-dose Butantan-DV, are being rolled out through national programs.
Travelers should consult travel vaccination clinics for up-to-date vaccination advice when visiting at-risk countries in 2026.

The Public Health Agency of Canada (PHAC) recently aligned its travel health guidance with that of the U.S. Centers for Disease Control and Prevention (CDC), issuing Level 2 travel notices for Bolivia, Cuba, Seychelles, Sri Lanka, and Suriname due to increased chikungunya virus activity.
As of February 19, 2026, these Level 2 advisories, which call for enhanced precautions, address the higher-than-expected transmission rates of the mosquito-borne illness in these destinations.
These advisories follow an epidemiological alert issued by the Pan American Health Organization (PAHO) on February 10, 2026, which warned of a sustained increase in chikungunya cases across various parts of the Americas since late 2025 and into early 2026.
The alert highlights the re-emergence of local transmission in areas that had been free of the virus for several years, including regions of the Guiana Shield and parts of Florida.
Chikungunya, primarily spread by infected mosquitoes, causes symptoms such as sudden fever, severe joint pain, muscle aches, headaches, nausea, fatigue, and rashes. While rarely fatal, the disease can cause prolonged joint discomfort in some individuals.
In December 2013, the virus was detected for the first time in the Americas Region.
According to PAHO data and related reports, by early 2026 (specifically epidemiological week 4, around late January to early February), 7,150 chikungunya cases had been reported regionally, including 2,351 confirmed cases and 1 death.
Cuba reported a significant number of suspected cases early in the year, with updates indicating that 1,457 were suspected as of late January.
The PHAC and the CDC recommend that travelers to these countries take necessary precautions, such as avoiding mosquito bites. They note that vaccination against chikungunya is available at travel clinics as of February 2026.
The Chicago Department of Public Health (CDPH) is currently monitoring a cluster of invasive meningococcal disease (IMD) cases.
Since January 15, 2026, ten adult cases and two related fatalities have been confirmed.
As of mid-February 2026, this Chicago outbreak is primarily affecting the West Side and Southwest Side of the city, with case numbers far exceeding the typical 0-2 cases reported in January over the past decade.
All tested isolates from this outbreak are identified as serogroup Y, which has been the dominant strain in Chicago since 2022.
CDPH recommends the MenACWY vaccination (Menveo, MenQuadfi) for individuals experiencing homelessness and encourages hospitals to vaccinate at-risk patients upon discharge.
Nationwide, IMD cases have been increasing sharply since 2021. In 2024, there were 503 confirmed and probable cases (based on preliminary data), marking the highest annual total since 2013.
According to CDPH, Neisseria meningitidis, the bacteria that cause IMD, is transmitted through direct contact with saliva. However, it typically requires close and prolonged contact to spread.
For the most accurate and up-to-date national figures, please visit the CDC's meningococcal disease surveillance webpage.

The Pan American Health Organization (PAHO) recently announced it has partnered with the Ministry of Health, Wellness, and the Environment of Saint Vincent and the Grenadines to promote the rollout of the human papillomavirus (HPV) vaccine for school-aged children.
On February 11, 2026, PAHO stated that HPV vaccination is a crucial component of cervical cancer prevention, as persistent infections with high-risk HPV types account for the majority of cervical cancer cases throughout the Caribbean Islands.
This collaboration is particularly important in Saint Vincent and the Grenadines, where cervical cancer poses a significant public health challenge.
Recent data revealed that 17 women were diagnosed with cervical cancer in 2024, marking an 88.9% increase from 2023. An additional 17 new cases were recorded in the first nine months of 2025.
From 2020 to 2024, a total of 44 women succumbed to this vaccine-preventable disease.
In 2023, cervical cancer accounted for 12.7% of all cancers among women in Saint Vincent.
The situation is similarly alarming across the Caribbean region, including Cuba and Puerto Rico, which have one of the highest rates of cervical HPV infection globally, at approximately 16% among women, ranking second only to sub-Saharan Africa.
Every year, more than 78,000 women in the Americas are diagnosed with cervical cancer, leading to over 40,000 deaths, 83% of which occur in Latin America and the Caribbean.
Dr. Amalia Del Riego, PAHO/WHO Representative for Barbados and the Eastern Caribbean Countries, welcomed the strong community response to this initiative. "We congratulate the Ministry of Health for advancing the elimination strategy by actively engaging communities," she stated.
"This partnership builds on recent milestones in the country, including the introduction of HPV DNA testing for screening (launched in September 2025) and expanded access to diagnostic and treatment services."
These actions, from vaccination to screening and treatment, represent a holistic approach to eliminating cervical cancer, in line with PAHO and WHO regional and global goals for 2026.
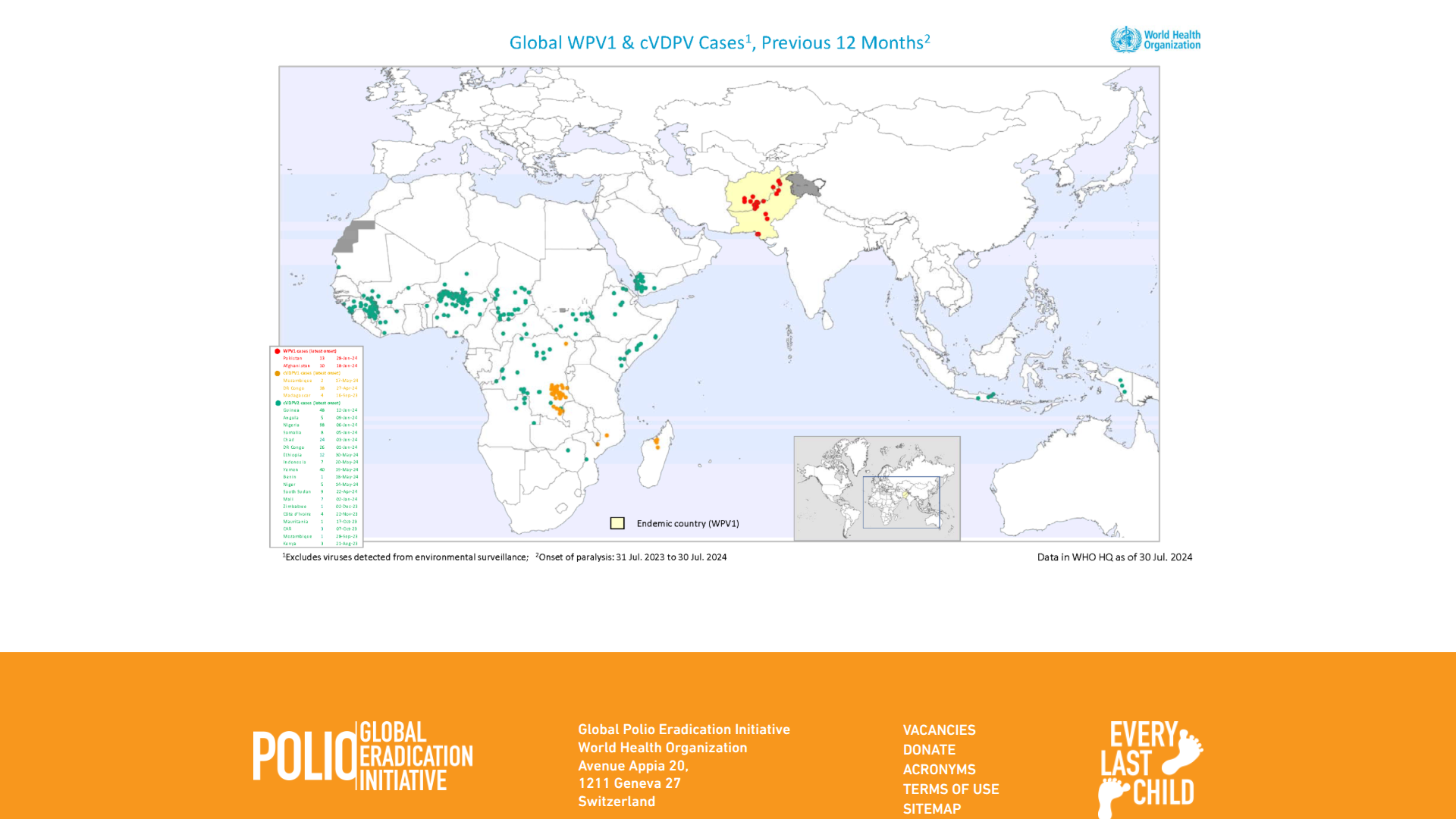
In a significant move to eliminate one of the world's last remaining polio-endemic threats, the Government of Japan has provided $6.3 million to support polio eradication and strengthen routine immunization services across all 34 provinces of Afghanistan.
This 12-month initiative, announced on February 11, 2026, will support the procurement and distribution of oral polio vaccines (OPV) to reach more than 12 million Afghan children.
The novel OPV (nOPV2) has been deployed about 2 billion times in recent years.
Afghanistan is one of only two countries, along with Pakistan, where wild poliovirus type 1 is still circulating, posing a serious risk of paralysis and death to unvaccinated children.
While significant progress has been made, with reported cases dropping sharply in recent years, transmission continues in the country's high-risk southern regions.
According to official data from the Global Polio Eradication Initiative and local health authorities, wild polio cases in Afghanistan decreased from 25 in 2024 to 13 in 2025.
As of early February 2026, no new wild polio cases have been reported in Afghanistan this year.
Globally, the total number of wild polio cases reached around 44 in 2025, comprising 13 in Afghanistan and 31 in Pakistan, a decline from 99 cases the previous year.
This trend highlights the positive impact of ongoing vaccination efforts.
This renewed financial support from Japan reinforces hope that Afghanistan will soon join the list of polio-free nations, thereby protecting future generations of children from this preventable disease.
A clinical-stage biotechnology company recently announced that the first participants have been dosed in the company's Phase 1A clinical trial evaluating Centi-Flu 01, a pan-influenza universal flu vaccine.
According to Centivax, the Phase 1A represents a key milestone toward a new kind of flu vaccine designed to provide broader, more reliable protection than standard seasonal vaccines, protecting against currently circulating strains, future strains, and pandemic strains.
The first data from this studyis expected within the year.
Unlike conventional seasonal influenza vaccines, which must be reformulated annually to attempt to match predicted circulating strains, Centi-Flu 01 is designed to focus both antibody and cellular immune responses on conserved regions of the influenza virus that cannot mutate and are shared across strains and distance subtypes.
This approach aims to generate broad, consistent, and durable immunity against both seasonal and pandemic influenza.
Sawsan Youssef, PhD, founder and Chief Science Officer of Centivax, stated in a press release on February 12, 2026, "A universal influenza vaccine allows us to be proactive—moving from annual guesswork to predictable, durable response."
In addition to safety, the study will evaluate efficacy based on established correlates of protection, using the gold-standard hemagglutination inhibition (HAI) assay against a panel of more than twenty flu strains—including currently circulating strains, historical mismatch strains, seasonal guidance strains, and pandemic strains—in a direct head-to-head comparison with existing standard-of-care flu vaccines.
Because the HAI assay is the same correlate-of-protection used to license seasonal flu vaccines, positive data will provide a clear benchmark demonstrating the candidate's ability to deliver broad protection with a single vaccine.
This type of innovative flu shot is essential because current vaccines are suboptimal at preventing virus transmission.
The 2024-2025 seasonal flu vaccine (trivalent formulation provided moderate protection in the United States, according to interim estimates from the U.S. CDC. Seasonal influenza effectiveness estimates among children and adolescents was 32%, 59%, and 60% in outpatient settings.