Search API
The Switzerland Federal Office of Public Health today reported its first case of Clade Ib mpox in an individual who had returned from Africa.
On April 8, 2025, the Swiss government stated that this person was in isolation and that there was no risk of infection to others.
The treatment of mpox consists primarily of treating its symptoms. In particularly severe cases, an antiviral therapy may also be conducted. The appropriate medicine is available in Switzerland in 2025.
Since September 2023, the World Health Organization has observed increased infections, especially of a new mpox variant designated Clade Ib, in Several African countries, including the Democratic Republic of the Congo.
The other well-known Clade II began infecting people worldwide in May 2022.
A preventive vaccination (Jynneos®) is assumed to be effective against Clade I infection. The vaccine is safe and highly effective in preventing severe mpox infections.
Jynneos is also available in numerous countries, such as the United States.

The South Korean Ministry of Food and Drug Safety recently approved BARYTHRAX, an anthrax vaccine jointly developed by GC Biopharma and the Korea Disease Control and Prevention Agency.
Traditional anthrax vaccines are made by attenuating Bacillus anthracis or culturing non-pathogenic Bacillus anthracis, which may contain residual toxin components. BARYTHRAX removes this risk and improves vaccine safety.
BARYTHRAX utilizes protective antigen (PA) proteins produced through genetic recombination techniques. With an anthrax infection, PA is a gateway for 2 Bacillus anthracis toxins, lethal factor and edema factor, to enter host cells.
BARYTHRAX vaccination can train and stimulate an immune response to neutralize anthrax by utilizing PA proteins.
Eun-chul Huh, President and CEO of GC Biopharma, commented in a press release on April 9, 2025, "This achievement underscores our commitment to localizing critical medicines for public health and national security. GC Biopharma will continue leading efforts to ensure stable supplies of essential medical products, as we have been doing with other vaccines and blood products since our founding."
Anthrax, caused by Bacillus anthracis, is a class-1 infectious disease capable of surviving extreme conditions and spreading easily through airborne transmission. If untreated, its fatality rate can reach up to 97%, making it a significant threat as a potential biological weapon.
The MFDS's approval, supported by GC Biopharma's production capacity, will pave the way for the company to supply Korea's essential anthrax vaccine reserve.
In the United States, very few people get anthrax from infected animals or contaminated animal products. The U.S. CDC says The type of illness a person develops depends on how anthrax enters the body: through the skin, lungs, or gastrointestinal system.
Getting a vaccine or taking certain antibiotics after exposure to anthrax can help prevent illness.
As of April 9, 2025, the U.S. FDA has approved anthrax vaccines for those at risk of exposure to anthrax bacteria.
For example, CYFENDUS™ is a combination of BioThrax® (anthrax vaccine adsorbed) and CPG 7909, a synthetic short DNA sequence as a vaccine adjuvant.
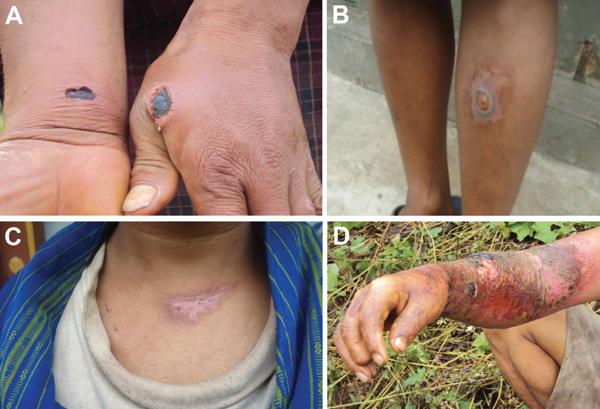
Shigellosis is the second leading cause of fatal diarrheal disease worldwide, strongly contributing to pediatric morbidity and mortality, without a U.S. FDA-approved vaccine available.
According to public health leaders and the Gates Foundation, developing an effective vaccine to prevent this deadly disease is essential in many areas worldwide.
To address this need, Valneva SE and LimmaTech Biologics AG today announced that the first participant has been vaccinated in a Phase 2 infant safety and immunogenicity study of Shigella4V2 (S4V2), the world's most clinically advanced tetravalent bioconjugate vaccine candidate against shigellosis.
Dr. Juan Carlos, Chief Medical Officer of Valneva, commented in a press release on April 9, 2025, "Seeing so many infants and children dying from shigellosis is not acceptable if it can be prevented with a vaccine."
"As such, the development of Shigella vaccines has been identified as a priority by the World Health Organization and, in line with our mission of developing vaccines against infectious diseases with unmet medical needs, we are focused on delivering a preventative solution against this deadly disease."
In the Phase 2 study S4V02, the safety and immunogenicity of S4V2 will be tested in approximately 110 nine-month-old infants to identify the best dose to be tested in a Phase 3 trial.
Sponsored and conducted by LimmaTech, S4V02 is a randomized, controlled, and blinded study conducted at a single study site in Kenya. Participants will receive a two-dose vaccination with one of two different vaccine dose levels of S4V2 or a control vaccine. Safety will be evaluated throughout the trial for approximately six months following the last vaccination.
Results of the phase 2 study, which is supported by funding from the Gates Foundation, are expected in the second half of 2025.
In November 2024, Valneva and LimmaTech launched a Phase 2b controlled human infection model (CHIM) study of S4V2 in healthy Shigella-naïve adults. This CHIM study forms part of the companies' staggered and risk-mitigating development strategy for S4V2, as it should provide the first results on efficacy before potentially advancing to further CHIM and Phase 3 studies.

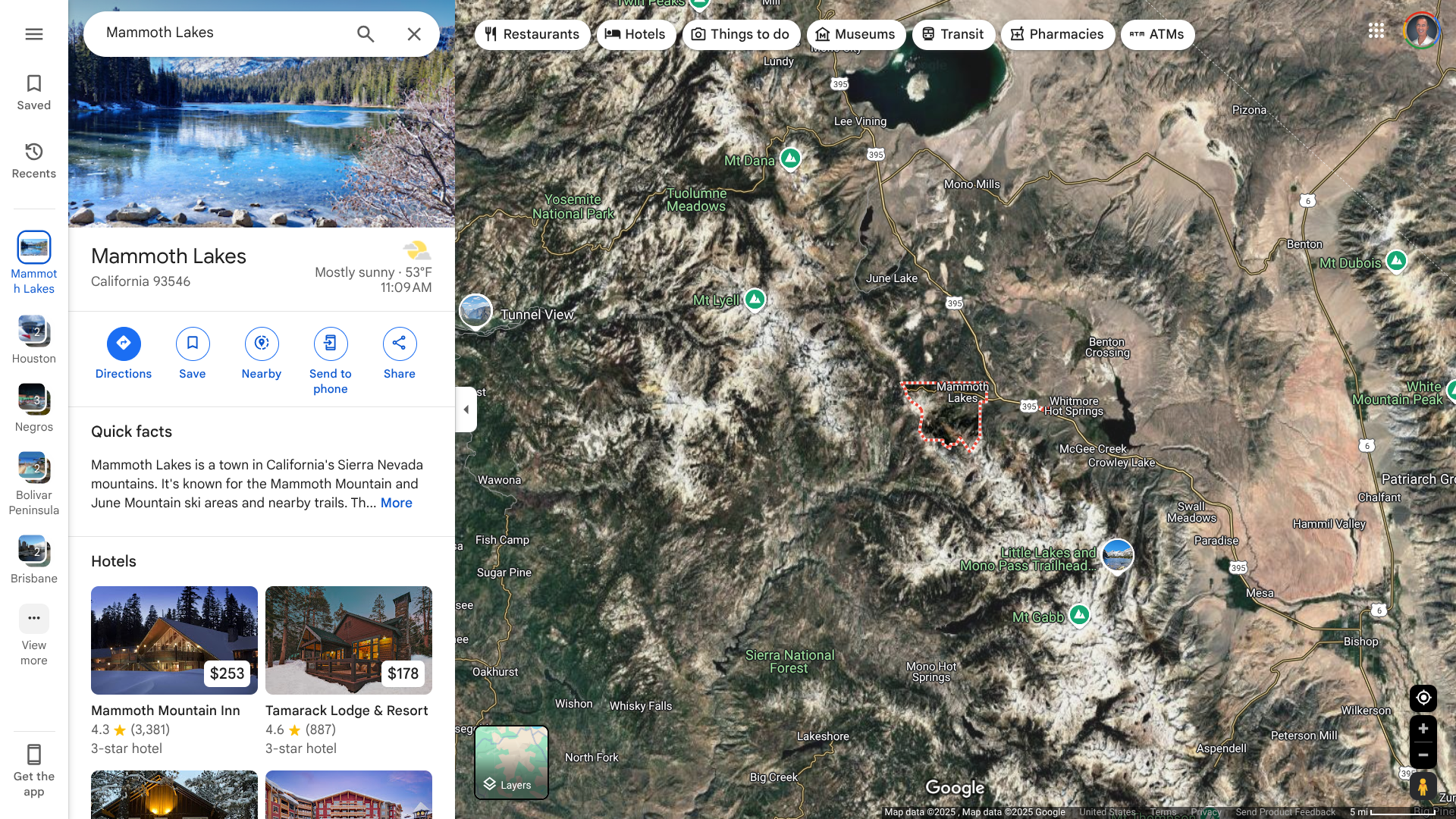
Mono County Public Health has confirmed a third fatality due to Hantavirus in the Town of Mammoth Lakes, a favorite outdoor destination for vacationers in Northeast California.
As of April 3, 2025, Mono County has recorded 27 cases since it was first reported here in 1993, the most in California. Twenty-one of these infections affected county residents, and six occurred among visitors who were infected in Mono. In
As of 2022, 864 cases of Hantavirus disease were reported in the United States.
According to the U.S. CDC, Hantavirus is a serious and often fatal illness contracted through infected deer mouse droppings, urine, or saliva. It most commonly occurs in the late spring or summer.
“A third case of Hantavirus Pulmonary Syndrome (in 2025), each of which has been fatal, is tragic and alarming,” said Dr. Tom Boo, Mono County Public Health Officer, in a press release.
“We don’t have a clear sense of where this young adult may have contracted the virus. The home had no evidence of mouse activity. We observed some mice in the workplace, which is not unusual for indoor spaces this time of year in Mammoth Lakes.
We haven't identified any other activities in the weeks before illness that would have increased this person's exposure to mice or their droppings.
We’ve been aware of this suspected case for weeks, but obtaining testing has taken time. The occurrence of three cases in a short period has me worried, especially this early in the year.
We’ve gone about a month without additional suspect cases, but remain concerned about the increased activity.
We believe deer mouse numbers are high this year in Mammoth (Eastern Sierra). An increase in indoor mice elevates the risk of Hantavirus exposure.
As far as we know, none of these deceased individuals engaged in activities typically associated with exposure, such as cleaning out poorly ventilated indoor areas or outbuildings with a lot of mouse waste. Instead, these folks may have been exposed during normal daily activities in the home or the workplace.
Many of us encounter deer mice daily, and there is some risk. We should pay attention to the presence of mice and be careful around their waste,” stated Dr. Boo.
Without a preventive vaccine available in 2025, the county says to avoid eating food that may have been contaminated by rodents and always wash your hands thoroughly after any potential exposure.
The UK Health Security Agency (UKHSA) today announced it had detected a confirmed human case of Clade Ib mpox, in which the person who resides in the North East of England had no reported travel history and no reported link with previously reported mpox cases in the UK.
England has reported 11 mpox cases this year.
As of April 7, 2025, the UKHSA says the mpox risk to the UK population remains low, as clade Ia and Ib mpox are no longer classified as a high-consequence infectious disease.
To date, all previous cases in the UK have either been linked to someone who has traveled to an affected country or been linked to someone who has.
UKHSA Mpox Incident Director Dr Gillian Armstrong commented in a media release, "The risk to the UK population from mpox remains low."
"The majority of people who have presented with symptoms report close physical contact, including massages or sex, before developing symptoms."
"While mpox infection is mild for many, it can be severe for some, and UKHSA is committed to preventing its spread within the UK."
Currently, the UK government has authorized an approved mpox vaccine known as IMVAMUNE® (JYNNEOS®, MVA-BN®).
Whether you have travelled or not, it is essential to remain alert to the risks. Anyone who thinks they may have mpox should contact NHS 111 for advice on what to do, says the UKHSA.
Recently, the U.S. CDC updated its Level 2 Travel Health Advisory, saying, 'There is ongoing person-to-person transmission of mpox in Burundi, Central African Republic, Democratic Republic of the Congo, Kenya, the Republic of the Congo, Rwanda, Tanzania, Uganda, and Zambia.'
'Person-to-person transmission has occurred through various means during this outbreak.'
About 15 million people visited central Africa last year, many traveling from the U.S. and the UK.
The CDC, like the UK, recommends mpox vaccination for at-risk individuals.
The Pan American Health Organization (PAHO) recently updated its Dengue fever outbreak data dashboard, highlighting the continued surge of cases in 2025.
Within the Americas, the Federative Republic of Brazil's state of São Paulo is on track to exceed last year's record numbers.
As of April 8, 2025, São Paulo has reported approximately 522,000 dengue cases and 390 related fatalities, with an additional 475 deaths currently under investigation by the health ministry.
For all of 2024, over 9.6 million dengue cases and 5,441 related fatalities were reported throughout Brazil.
While Dengue has become endemic in the U.S. territories of Puerto Rico and the U.S. Virgin Islands, most cases in the United States are travel-related.
The U.S. Centers for Disease Control and Prevention (CDC) reported on April 2, 2025, that 1,346 travel-related Dengue cases and one local case have been reported in 30 jurisdictions this year.
Most of these cases were identified in southern Florida.
With over 6 million people visiting Brazil in 2024, and many international travelers returning to the U.S., identifying Dengue infections has become a challenge.
To offer updated guidance on Dengue testing, the CDC recently issued a Health Alert Network (HAN) Health Update to provide additional information to healthcare providers about the ongoing risk of dengue virus infections and updates to testing recommendations in the United States.
The CDC urges healthcare providers to continue taking steps to prevent, detect, diagnose, and respond to Dengue, as described in the June 2024 HAN Health Advisory (CDCHAN-00511).
Currently, the CDC does not recommend any Dengue vaccine in the U.S. However, second-generation Dengue vaccines are available in about 40 countries.

The Department of Mayotte, an overseas department of France, recently announced that, due to the first indigenous (local) case of chikungunya on the island, health authorities are activating level 2A of the ORSEC (emergency) plan.
This decision will increase surveillance of the epidemiological situation and measures to prevent more active viral (virus) circulation by mosquitoes in the territory.
As of April 7, 2025, Mayotte has reported four chikungunya cases this year.
As a reminder, about 900 miles away, Reunion Island has been facing a chikungunya epidemic, with 13,594 cases, including two related fatalities, since August 2024.
Mayotte's Regional Health Agency says all travelers to and from Reunion Island should protect themselves from mosquito bites.
Mayotte is located between Africa's east coast and Madagascar, one of France's 18 regions.
To protect people on Reunion, the local government has started vaccinating 40,000 residents with a U.S. FDA-approved vaccine.
Valneva SE's IXCHIQ® Chikungunya vaccine is commercially available in the U.S. and recommended for travelers to endemic areas in 2025.

Two powerful earthquakes recently struck the central part of the Republic of the Union of Myanmar's Sagaing Region near Mandalay. The full scale of damage, deaths, and injuries has not yet been ascertained.
The World Health Organization's (WHO) Public Health Situation Analysis, released on April 3, 2025, says preliminary assessments indicate high casualties and trauma-related injuries, with an urgent need for emergency care.
On April 2, 2025, the State Administration Council (SAC) reported that 3,003 people had died and 4,515 had been injured in the earthquake.
In the earthquake-affected areas, with limited access to safe water and worsening sanitation and hygiene conditions, the WHO says the risks for Acute Waterborne (AWD) - cholera outbreaks are assessed as very high.
Since July 2024, AWD and cholera cases have surged in at least nine states, including Ayeyarwady, Bago, and Mandalay.
Mandalay, which faced a cholera outbreak in October 2024, remains at high risk for further outbreaks due to infrastructure damage and overcrowded shelters, significantly increasing the likelihood of cholera and other waterborne disease outbreaks.
From June 22, 2024, to March 23, 2025, 12,195 AWD cases (not all confirmed as cholera) were reported, along with three deaths.
A vaccination campaign using oral cholera vaccine (OCV) has been conducted in Yangon, Mon, and Mandalay, providing 2.4 million doses.
As of April 7, 2025, the U.S. CDC says, 'OCV vaccination may be considered for children and adults traveling to areas of active cholera transmission.'
In the United States, OCVs such as DUKORAL® are available at travel clinics and pharmacies.
The WHO wrote, 'These deteriorating conditions, combined with overcrowded temporary shelters and reduced sanitation services, significantly increase the risk of cholera and other waterborne disease outbreaks in the affected areas.'