Search API
With Lyme disease season soon to spike in the Northeast, two pharmaceutical companies recently announced very positive news about a vaccine candidate.
Pfizer Inc. and Valneva SE announced on March 23, 2026, topline results from the Phase 3 VALOR "Vaccine Against Lyme for Outdoor Recreationists" clinical trial (NCT05477524) of its investigational 6-valent OspA-based Lyme disease vaccine candidate PF-07307405 (LB6V, formerly known as VLA15).
In the pre-specified analyses: Efficacy of 73.2% from 28 days post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 15.8, 93.5); Efficacy of 74.8% from 1-day post-dose 4 (season 2) in reducing the rate of confirmed Lyme disease cases compared to the placebo arm (95% CI 21.7, 93.9)
The press release stated, 'Given the clinically meaningful efficacy and the fact that the 95% confidence interval lower bound was above 20 in the second pre-specified analysis, Pfizer is confident in the vaccine's potential and is planning submissions to regulatory authorities.'
"Lyme disease can cause potentially serious consequences – where individuals and families face symptoms that can disrupt daily life, work, and long-term health – and there is currently no vaccine available," said Annaliesa Anderson, Ph.D., Senior Vice President and Chief Vaccines Officer, Pfizer. "The efficacy shown in the VALOR study of more than 70% is highly encouraging and creates confidence in the vaccine's potential to protect against this disease that can be debilitating."
As of late March 2026, the U.S. CDC reports that the bacteria that cause Lyme disease are transmitted to humans by blacklegged (Ixodes) ticks. Infected ticks must be attached for over 24 hours to transmit the infection, so that prompt removal can prevent transmission. These ticks are most commonly found in forested areas of the northeastern, north-central, and mid-Atlantic states, as well as parts of the Pacific Coast.
The CDC reports that recent estimates using other methods suggest that approximately 476,000 people may be diagnosed and treated for Lyme disease each year in the United States. This number likely includes patients who are treated based on clinical suspicion but do not actually have Lyme disease.
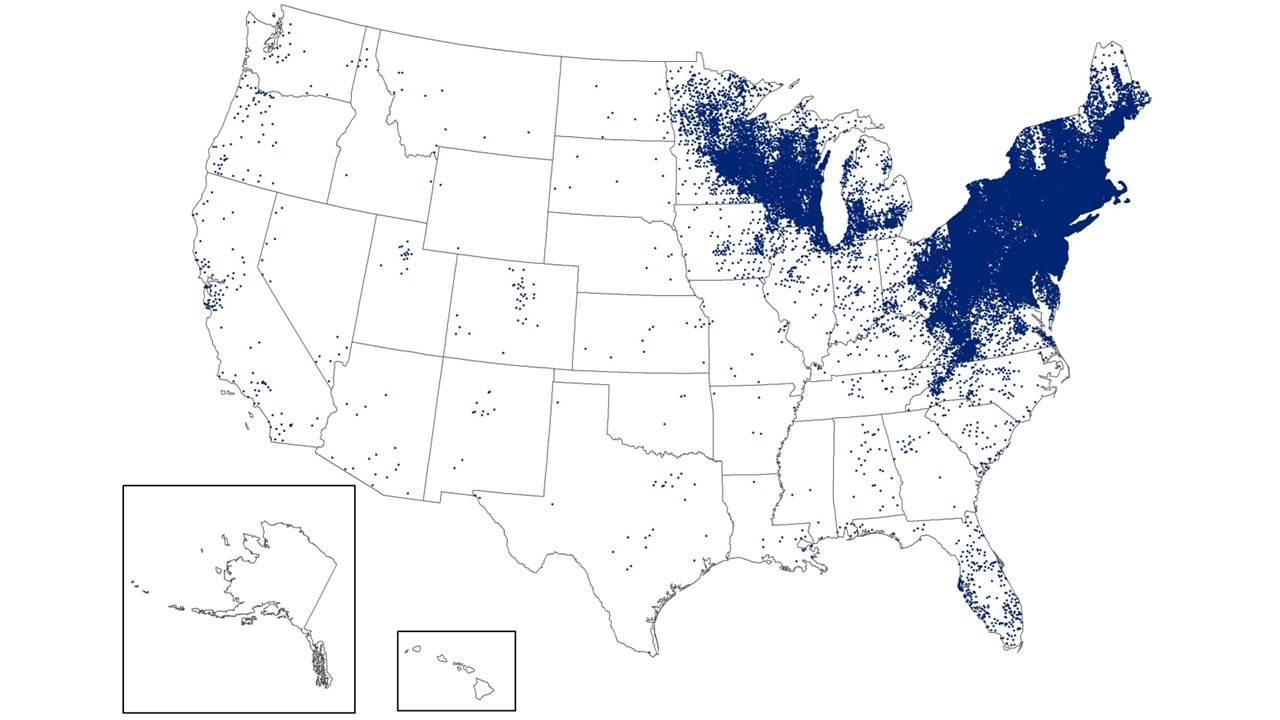
In response to the Meningitis B (MenB) outbreak in Kent, the UK Health Security Agency (UKHSA) is expanding its efforts to provide preventive antibiotic treatment and vaccinations.
As of March 19, 2026, vaccination is now available for students (years 12 and 13) in schools and colleges with confirmed or probable cases of MenB.
Following an assessment by the local health protection team, antibiotics and vaccinations may also be offered to additional year groups. Students are encouraged to continue attending school as usual.
The vaccination extension also includes students at the University of Kent living on the Canterbury campus, close contacts of confirmed cases, and individuals who visited Club Chemistry in Canterbury in early March. The goal is to provide timely long-term protection for those at risk.
Professor Susan Hopkins from the UKHSA stated in a media release, "By extending the vaccination programme to those who have received antibiotics, we take an important step to protect those exposed."
Eligible individuals can request both the vaccination and antibiotics from their local GP.
Due to high demand for the private MenB vaccine, the NHS will release an additional 20,000 doses, enabling pharmacies to receive vaccines within 48 hours.
The UKHSA emphasizes that meningococcal disease, which includes meningitis and sepsis, is a serious but rare condition caused by meningococcal bacteria. Everyone is advised to remain vigilant for signs of severe illness and seek urgent medical attention if feeling unwell.
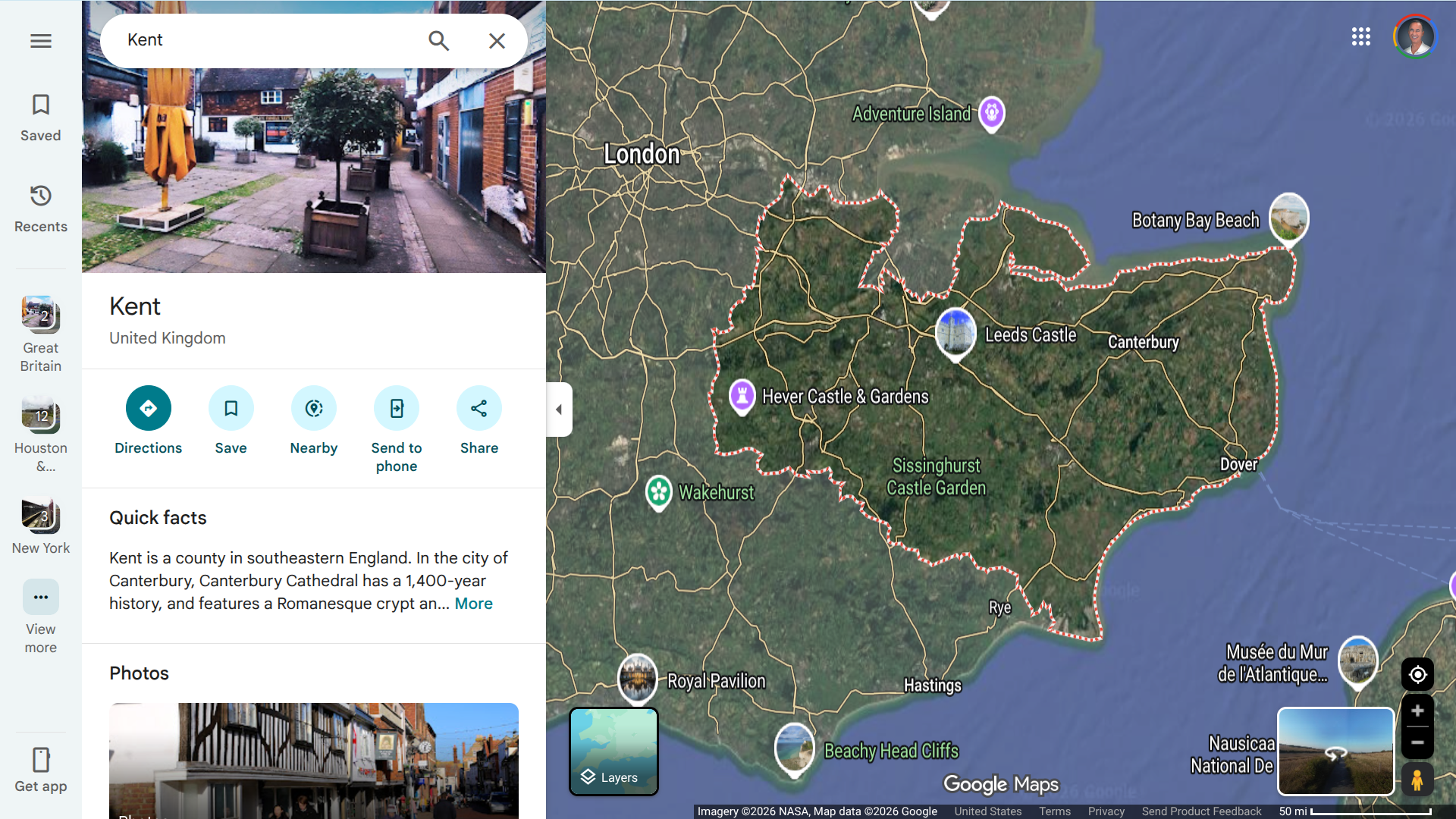
Since September 2022, over 1,000 confirmed and possible cases of shigellosis and other gastrointestinal infections have been reported among travelers returning from the Republic of Cabo Verde to Europe, the United Kingdom, and the United States.
New cases continue to emerge in late March 2026, more than three years into the outbreak.
This information comes from the latest update by the European Centre for Disease Prevention and Control (ECDC). In an epidemiological alert published on March 18, 2026, the ECDC reported a total of 766 confirmed and suspected cases of shigellosis across 13 EU/EEA countries, the UK, and the US.
Additionally, around 300 cases of other gastrointestinal infections — including salmonellosis, cryptosporidiosis, giardiasis, campylobacteriosis, and pathogenic Escherichia coli — have also been identified.
Despite ongoing investigations by local, European, and international health authorities, the exact source of these infections, which are believed to be food- or waterborne, has not yet been identified. Most of the affected travelers had stayed at the same hotel chain in the Santa Maria region on the island of Sal, a popular all-inclusive resort destination.
Cabo Verde is an archipelago of islands and islets located in the Atlantic Ocean, about 385 miles off the coast of West Africa.
As of March 24, 2026, the United Kingdom has reported 263 cases, while the United States has reported 7 cases.
The ECDC assesses the likelihood of new infections among travelers visiting the Santa Maria region of Sal as moderate, as the source remains uncontrolled. The dominant strain identified is Shigella sonnei, which causes severe diarrhea (often bloody), fever, abdominal cramps, and dehydration. Some strains exhibit antibiotic resistance, complicating treatment.
Further cases are anticipated until effective control measures are put in place.
Travelers returning from Sal who experience persistent diarrhea, bloody stools, fever, or severe stomach cramps should seek medical attention immediately and inform their healthcare provider about their recent travel history.
Currently, no licensed vaccine is available for shigellosis or the other pathogens involved in this outbreak. However, promising candidates such as Valneva and LimmaTech's tetravalent Shigella4V2 (S4V2) bioconjugate vaccine candidate are advancing through Phase 2 clinical trials.
The smallest country in South America has reported a significant rise in chikungunya virus infections, with laboratory-confirmed cases exceeding 1,357 as of early March 2026.
According to a report from the Suriname Ministry of Health, local transmission of this mosquito-borne virus has resumed after nearly a decade without any reported autochthonous cases.
Local health authorities believe that the actual number of infections is likely much higher—potentially up to three times the confirmed figure, or around 4,000 cases—due to underreporting and typical health-seeking behaviors within the population.
As of now, one death has been officially linked to chikungunya, while a second fatality is under investigation. In February 2026, the U.S. CDC issued a Level 2 Travel Health Notice, advising travelers to take enhanced precautions in light of the chikungunya outbreak in Suriname.
While there is no specific antiviral treatment for chikungunya, approved vaccines are being offered in various countries as of March 24, 2026.
Campania health authorities in Italy have confirmed a significant increase in hepatitis A cases, with 133 confirmed infections reported since January 2026.
As of March 20, 2026, the outbreak, which is centered around Naples and Capri, is primarily linked to the consumption of raw or undercooked bivalve mollusks, such as mussels, clams, and oysters that have been contaminated with the hepatitis A virus.
Additionally, there is evidence suggesting possible secondary person-to-person transmission contributing to this year's outbreak.
Currently, more than 40 to 50 patients are hospitalized at Naples' Cotugno Hospital.
In response to the outbreak, local media reported Naples Mayor Gaetano Manfredi issued an emergency ordinance on March 19-20, banning the sale and consumption of raw seafood in public establishments.
Residents and tourists are urged to thoroughly cook shellfish, practice strict hand hygiene, and avoid raw seafood. Campania, located on Italy's southern coast, is a major tourism hub, attracting approximately 20 million tourists annually.
This situation highlights the recurring food safety challenges faced by international travelers in 2026.
Public health officials recommend vaccination for at-risk groups and emphasize the need for continued vigilance to prevent further spread of the virus.
When departing from the USA for international travel in 2026, hepatitis A vaccination services are offered at travel vaccine clinics.
Argentine health authorities recently announced the country's first locally acquired case of mpox clade Ib. This development in Buenos Aires raises concerns about the potential for community transmission of this variant in South America.
The emergence of locally acquired clade Ib in Argentina underscores the virus's ability to establish transmission chains through sexual networks involving international travelers.
Buenos Aires, a major South American tourism hub, welcomed about 1.5 million foreign visitors last year.
The case, announced in report #9 on March 16, 2026, involves a man living in Buenos Aires who has no recent international travel history but reported sexual contact with multiple partners, including travelers, before the onset of symptoms. The patient is currently receiving outpatient treatment and is recovering. Contact tracing has begun, and there are recommendations for 21 days of follow-up monitoring.
Argentina's Ministry of Health has characterized clade Ib as being more severe and contagious than previous strains. They are urging health authorities to strengthen surveillance, ensure proper isolation until lesions have scabbed over, and provide healthcare workers with appropriate personal protective equipment.
Mpox first gained global attention in May 2022, primarily due to clade IIb of the virus. That outbreak resulted in over 100,000 confirmed cases worldwide across 122 countries, mostly transmitted sexually among men who have sex with men. Clade Ib differs from the milder clade IIb in terms of transmissibility.
In Argentina alone, approximately 1,129 confirmed cases and two deaths were reported between 2022 and early 2023. As of 2026, eight countries in the Americas, including Argentina, have reported a total of 172 mpox cases, with no deaths recorded to date.
As of March 20, 2026, effective mpox vaccines (JYNNEOS) are generally available in most countries.

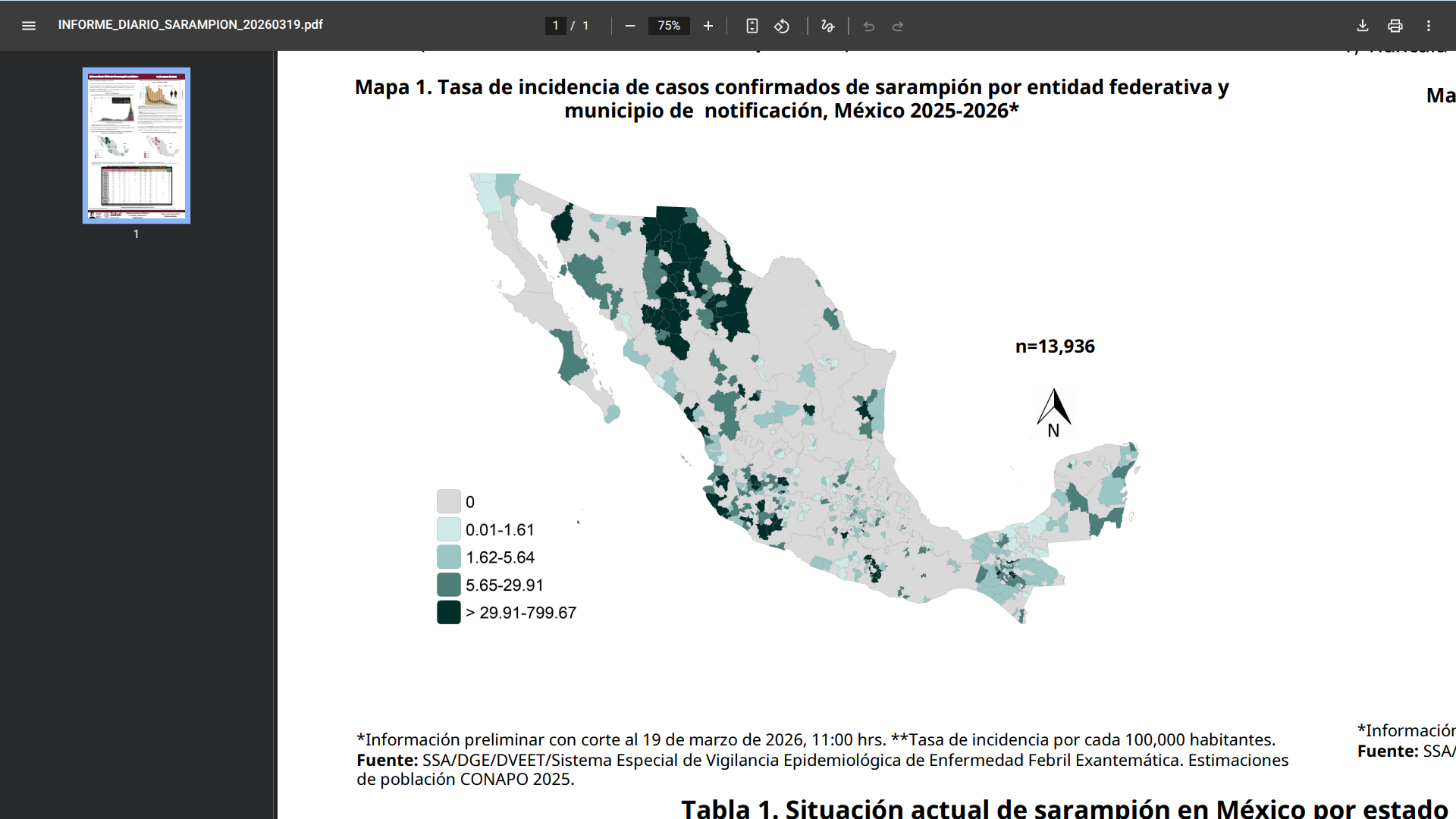
Mexico's Ministry of Health recently reported a significant increase in measles cases in 2026, with more than 7,400 confirmed infections recorded since the beginning of the year.
According to the epidemiological update on March 19, 2026, Mexico has reported 7,403 confirmed measles cases this year alone.
This rise in cases marks a sharp escalation in the ongoing outbreak that began in 2025. When including cases from late 2025, the cumulative total since the outbreak started exceeds 13,855 confirmed measles cases and 32 related fatalities across all 32 states and 437 municipalities.
The state most affected by the outbreak, Jalisco, a western Mexican state, has recorded 4,358 confirmed cases, accounting for the majority of this year's infections.
In response to the outbreak, health authorities have intensified vaccination efforts. From early 2025 through mid-February 2026, over 13.3 million doses of the measles vaccine have been administered nationwide.
Throughout Mexico, vaccination campaigns are ongoing, particularly in high-risk areas with low vaccination coverage, to help curb further spread.
As of March 20, 2026, Mexican officials are urging the public to ensure their vaccinations are up to date, particularly for children and travelers. This recommendation comes as the country prepares for large-scale events in the coming years.
To inform travelers about this ongoing health risk when departing from the United States, the CDC has issued a Level 1 - Practice Usual Precautions, Global Measles Travel Health Notice for Mexico.
In the U.S., measles vaccination services are offered at most travel vaccine clinics and pharmacies.
- 1 of 431
- next ›