Search API
An innovative bladder cancer intravesical therapy today gained its initial approval outside the United States. Bladder cancer is a challenging malignancy, and for many years, researchers have searched for next-generation treatment options.
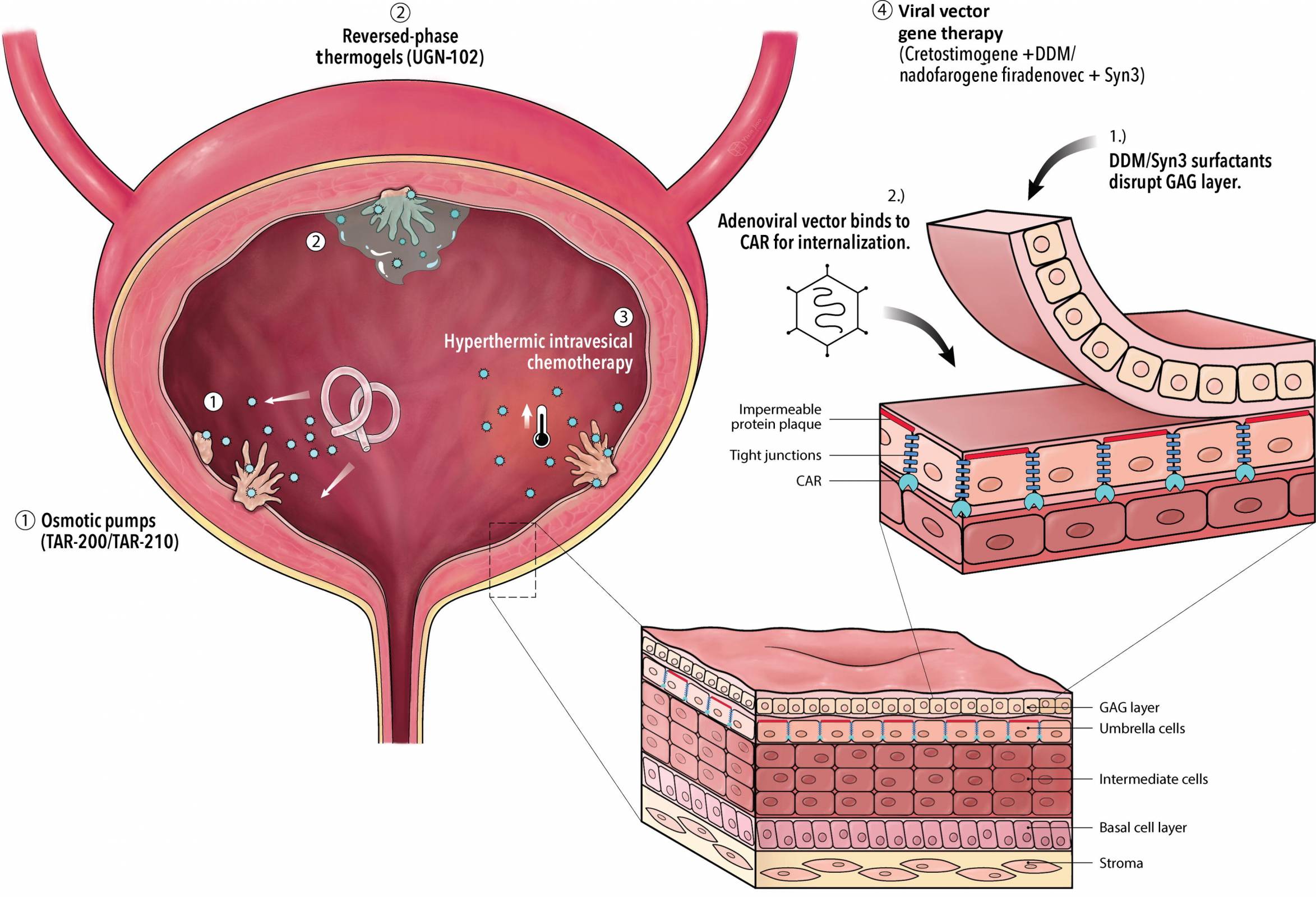
The UK's Medicines and Healthcare products Regulatory Agency (MHRA) has approved nogapendekin alfa inbakicept (Anktiva®) for adults with BCG-unresponsive non-muscle-invasive bladder cancer, where the disease remains confined to the inner lining of the bladder and may include tumors.
Nogapendekin alfa inbakicept (Anktiva) mixed with BCG is administered via a liquid that is diluted and then delivered into the bladder through a catheter inserted into the urethra.
The BCG (Bacillus Calmette-Guérin) vaccine, which has been deployed for approximately 100 years to reduce tuberculosis, has become a standard immunotherapy for early-stage bladder cancer. It is delivered directly into the bladder to stimulate an immune response.
Anktiva's mechanism of action involves the direct, specific stimulation of CD8+ T cells and Natural Killer cells through beta-gamma T-cell receptor binding, thereby generating memory T cells while avoiding stimulation of T-regulatory cells.
As of July 4, 2025, this medicine was approved through the International Recognition Procedure. The approval was granted to Serum Life Science Europe GmbH.
On May 27, 2025, ImmunityBio announced a collaboration to introduce the Cancer BioShield platform, along with Anktiva, to Saudi Arabia and the broader Middle East.
In the United States, ImmunityBio, Inc.'s BioShield platform, powered by Anktiva, was approved by the U.S. FDA for similar indications in April 2024. It is now commercially available at over 60 cancer centers in the U.S.
On July 1, 2025, ScienceDirect published a systematic review highlighting an array of novel intravesical therapies that demonstrate efficacy in bladder cancer patients.
A new study, published this week in JAMA Network Open and conducted by researchers from the University of Kentucky, analyzed data from the Cancer Statistics Incidence Analytic Database.
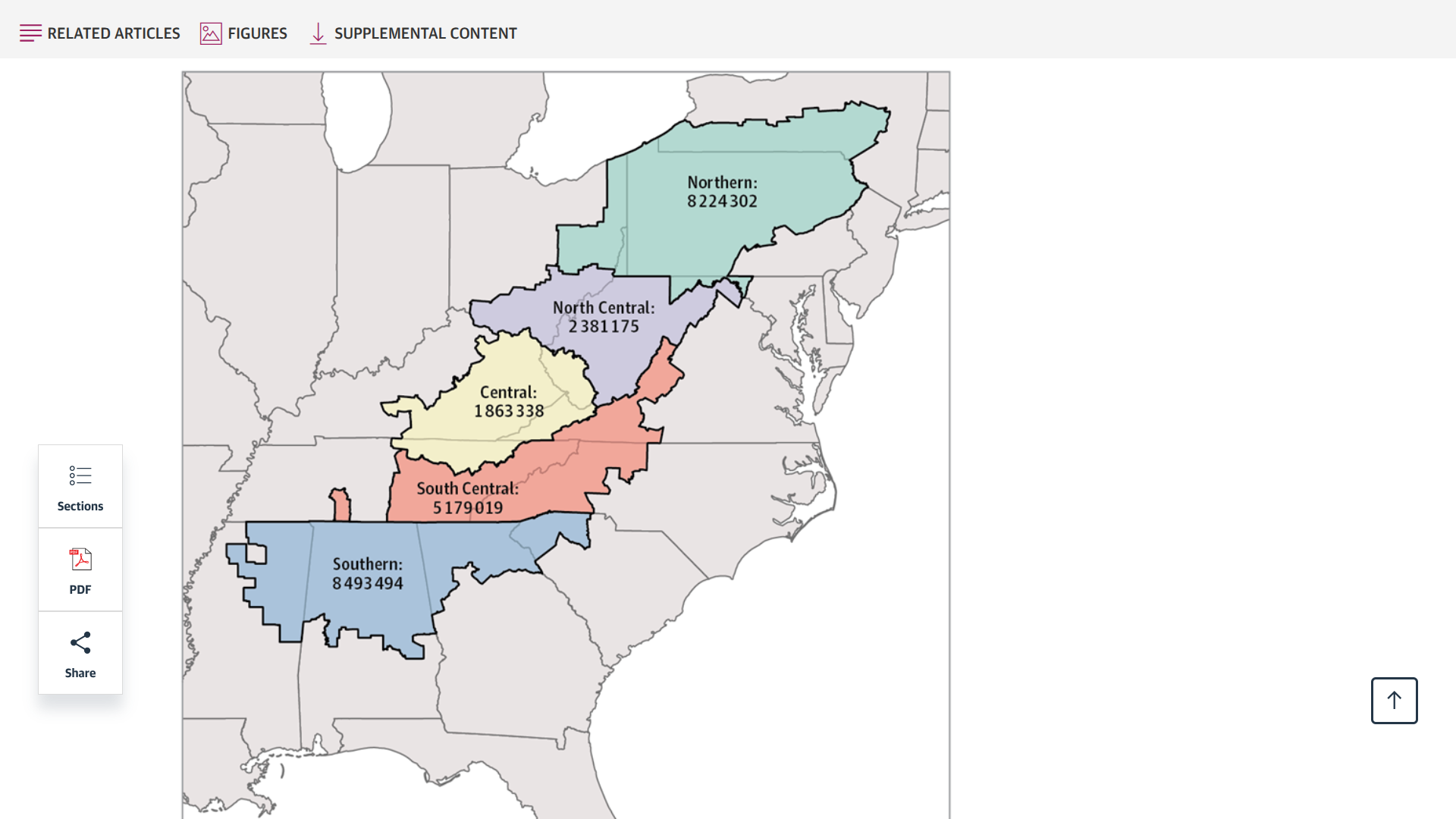
Released on June 30, 2025, this study's findings underscore the significant burden of cancer linked to human papillomavirus (HPV) among residents of Appalachia, who have a 16% higher rate of HPV-related cancers.
Furthermore, regional-specific disparities were seen for HPV-associated male and female oropharyngeal cancers, female anal cancer, vulvar cancer, cervical cancer, and penile cancer.
Incidence was highest among individuals living in the North Central and Central regions.
The North Central subregion had the highest incidence rates of male oropharyngeal cancer.
In contrast, the Central and North Central subregions had significantly higher rates of vulvar, cervical, and penile cancers than the other subregions.
These researchers wrote, 'This cross-sectional study of HPV-associated cancer incidence found disproportionately high HPV-associated cancer rates among Appalachian residents compared with non-Appalachian residents.'
'These findings highlight the need for targeted efforts to improve HPV vaccine uptake and encourage adherence to evidence-based screening guidelines for HPV-associated cancers in Appalachia.'
Currently, the U.S. CDC recommends HPV vaccination for most adolescents in a two or three-dose regimen. And recommends vaccination for everyone through age 26 if not adequately vaccinated at a younger age.
As of July 3, 2025, Merck's Gardasil 9 vaccine is readily available at health clinics and pharmacies throughout the Appalachian Mountain range and the United States.
Following a significant outbreak in 2024, the first confirmed case of West Nile virus illness was reported on June 24, 2025, in a resident of Brazos County, Texas.
This Texas Department of State Health Services (DSHS) announcement raises concerns for all Texans as virus-carrying mosquitoes remain active into December.
There were 455 cases of West Nile disease in Texas in 2024 and 56 related fatalities.
Over the last five years, Texas has had 929 West Nile cases and 122 deaths.
“Texans should be aware that mosquitoes transmit disease, and some of these illnesses, like West Nile and dengue, can be severe,” said DSHS Commissioner Jennifer A. Shuford, MD, MPH. “But taking steps to prevent mosquito bites and eliminating mosquito breeding areas around homes are proactive measures that can reduce the risk of mosquito-borne illness.”
Infected mosquitoes transmit West Nile virus after biting. Although 80% of people exposed to the virus do not develop symptoms, the remaining 20% will experience symptoms such as fever, nausea, headache, fatigue, and muscle and joint pain.
Less than one percent of those exposed will suffer from West Nile neuroinvasive disease, which affects the nervous system and can cause disorientation, neck stiffness, tremors, paralysis, convulsions, and even death.
DSHS urges anyone experiencing West Nile symptoms to contact their health care provider and mention any exposure to mosquitoes.
As of July 3, 2025, no vaccine for West Nile virus exists.

Florida health officials recently reported additional cases of mosquito-transmitted and travel-related chikungunya, dengue, and malaria in 2025.
After a slow start, these serious diseases have been concentrated in southeast Florida, in the great Miami area, which welcomes millions of international visitors each year.
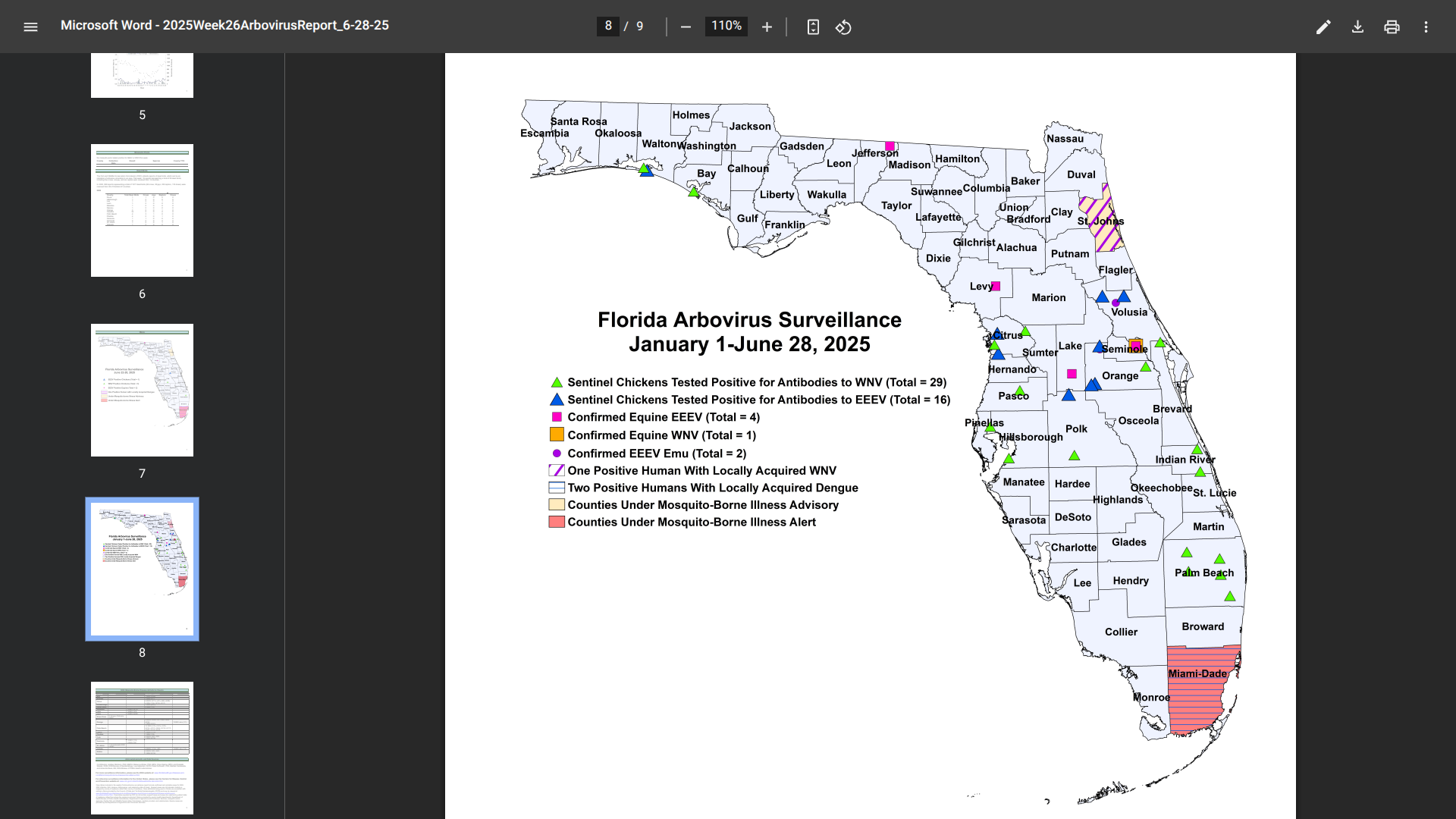
Currently, Miami-Dade County remains under a mosquito-borne illness alert.
As of June 28, 2025, Florida Health's Weekly Arbovirus Report confirmed a second, indigenous case of dengue in Miami-Dade County.
The most recent case has been identified as serotype DENV-4, while the early case was DENV-3.
In 2024, a total of 91 cases of locally acquired dengue have been reported across ten Florida counties.
Additionally, there have been four cases of chikungunya and 20 cases of malaria related to international travelers this year.
While there are no travel advisories issued for Florida's southeast coast, Canada and the UK both advise speaking with a travel vaccine expert before visiting at-risk areas in 2025.
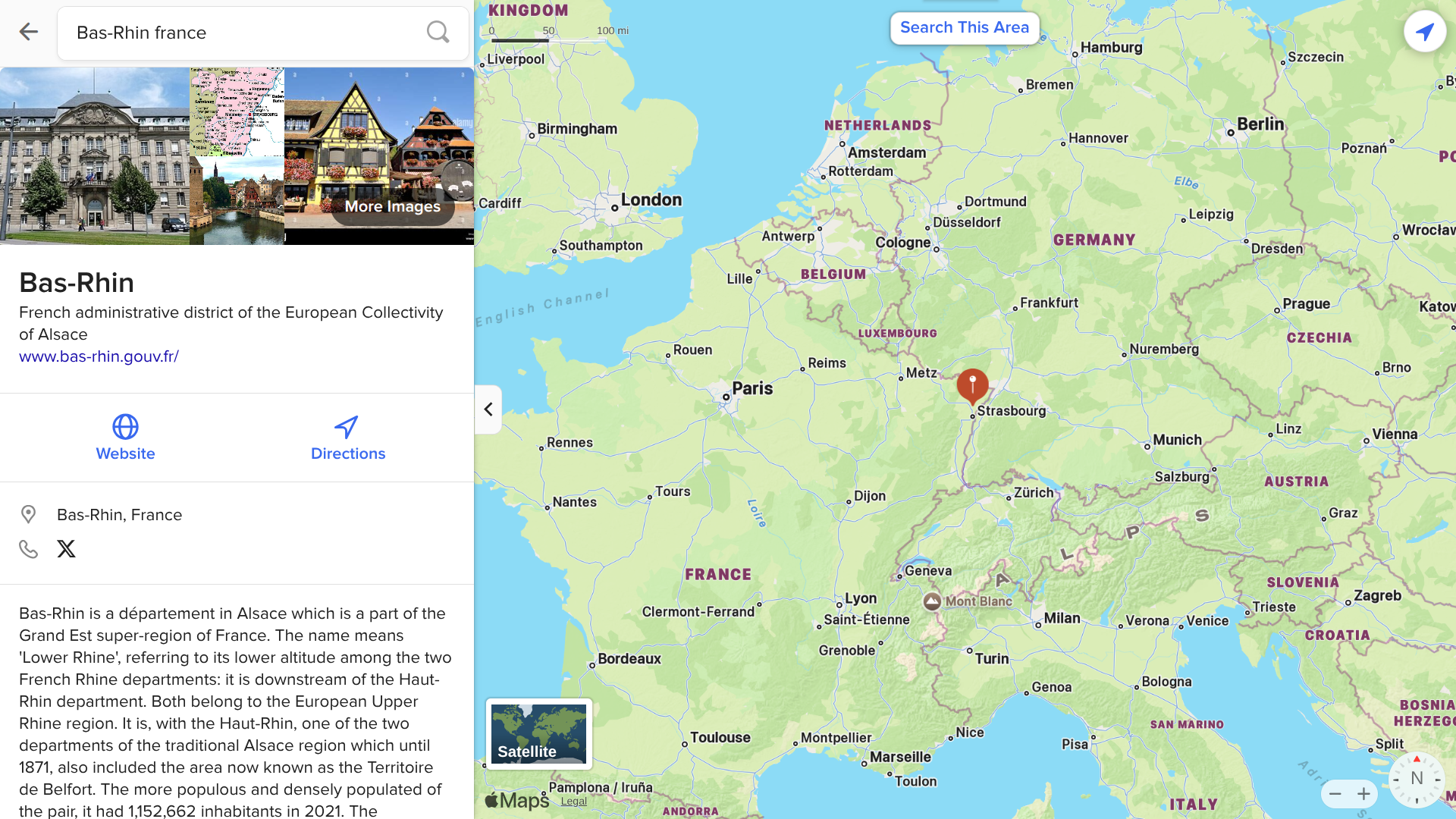
The regional health agency in Grand Est, France, reported its first locally acquired case of chikungunya in Bas-Rhin.
As of July 1, 2025, epidemiological investigations indicated that this locally acquired chikungunya case had frequented the municipalities of Lipsheim and Fegersheim during their infectious period.
Unlike previous local cases, this area is located in France's northeast.
In June 2025, Occitane (Hérault and Gard), Provence-Alpes-Côte d'Azur (Var and Bouches-du-Rhône), Auvergne-Rhône-Alpes (Drôme), and the island of Corse (Corse-du-Sud) reported local chikungunya cases.
And in the southern Indian Ocean, France's departments have reported significant chikungunya outbreaks in 2025.
According to the World Health Organization and the United States, chikungunya is a vaccine-preventable disease, with two vaccines commercially available as of July 2025.
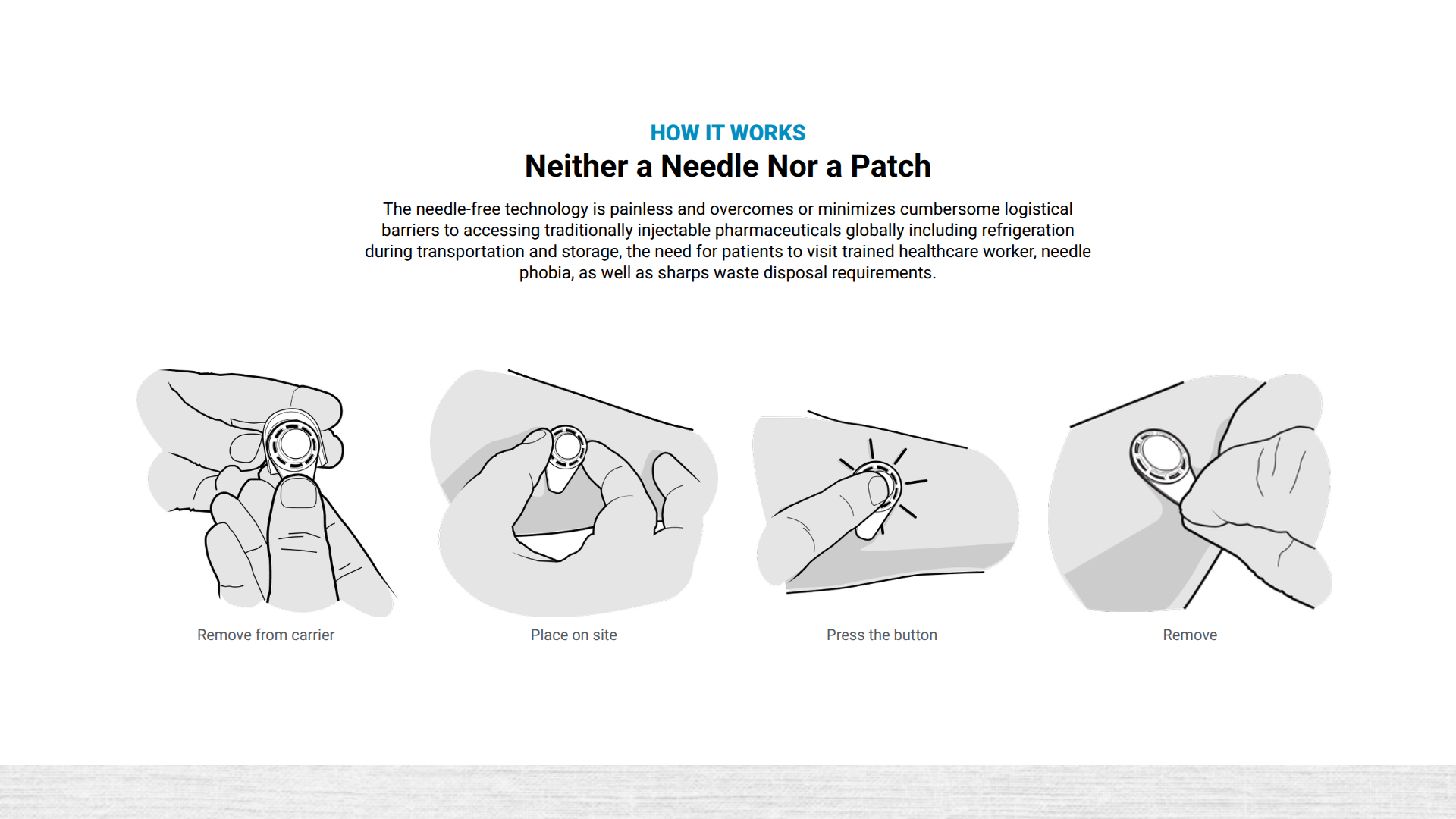
Emory University and Micron Biomedical recently announced the first clinical trial for a novel rotavirus vaccine, CC24.
This vaccine candidate is delivered using dissolvable microarray technology and is currently enrolling participants in the Phase 1 study, which launched in mid-June 2025.
Notably, this trial represents the first clinical evaluation of a drug or vaccine administered via patch or microarray, sponsored by the U.S. Centers for Disease Control and Prevention (CDC).
"CDC recognizes the potential of this groundbreaking clinical trial, which will test the safety of both our novel vaccine, CC24, in adults and the vaccine's delivery with 'patch' technology," says Dr. Demetre Daskalakis, Director of CDC's National Center for Immunization and Respiratory Diseases, in a press release.
"The trial marks significant progress in vaccine technology innovation and is a critical step toward saving more children from rotavirus illness and death."
Rotavirus infection is a leading cause of diarrheal deaths among children, particularly in low and middle-income countries where existing oral vaccines are often less effective. CC24 is a uniquely inactivated rotavirus vaccine developed by the CDC to provide an alternative to the oral administration of rotavirus vaccines.
Micron Biomedical's unique needle-free technology rapidly delivers vaccines and therapeutics via dissolvable microarray compounds that are painlessly pressed into the upper layers of the skin.
As of July 2, 2025, U.S.-FDA-approved rotavirus vaccines are available.
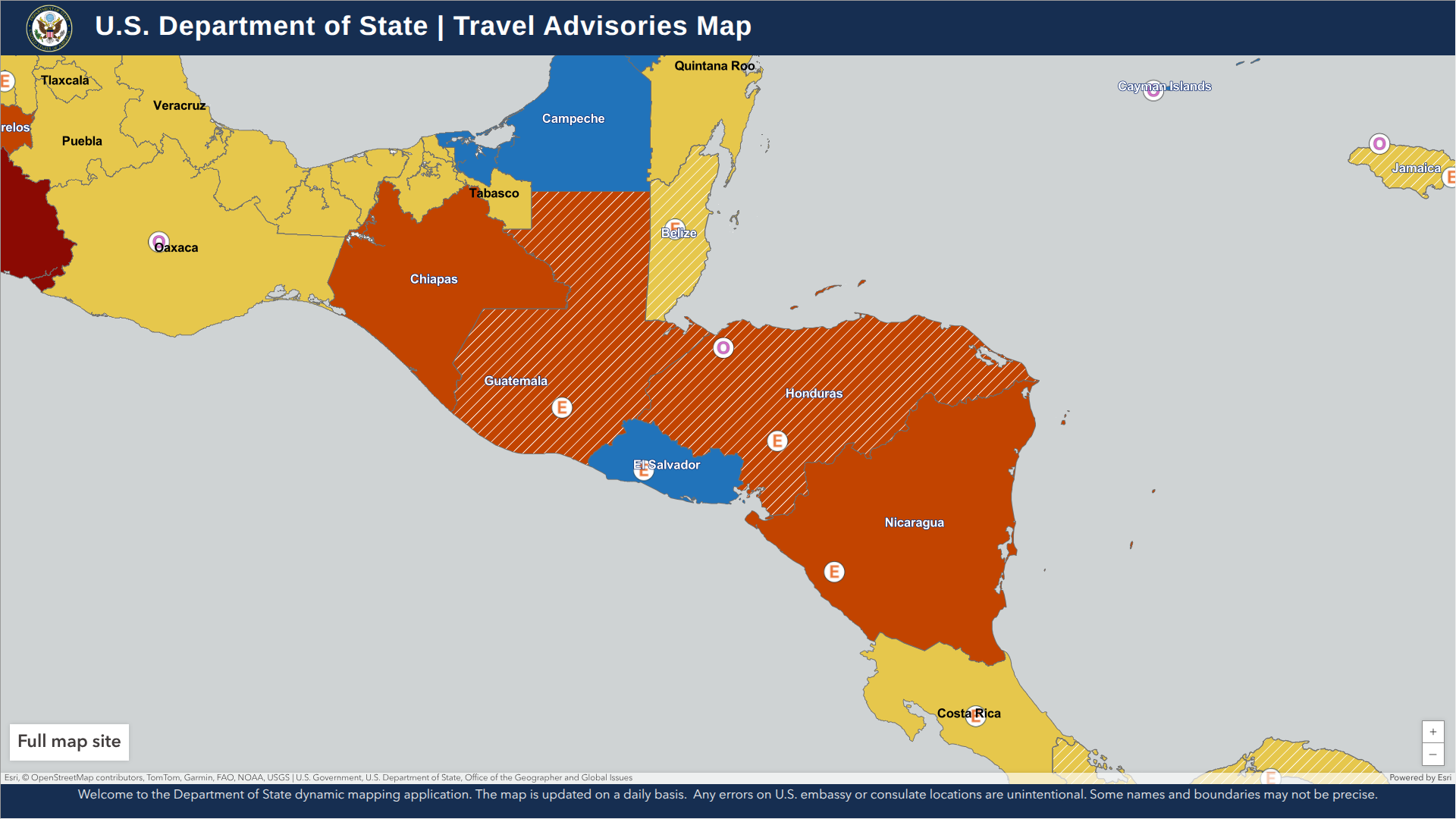
According to the Secretary of State at the Ministry of Foreign Affairs and International Cooperation, Honduran authorities are requiring documentation to show proof of a measles vaccine (MMR) for travelers coming from various countries, including the United States.
As of June 28, 2025, this new requirement applies to all foreign travel to the Republic of Honduras, whether by land, sea, or air.
The U.S. Embassy in Honduras stated that if someone cannot receive the vaccine, they will need to provide proof of a negative measles test result 72 to 80 hours before the flight.
This means that cruise ship passengers visiting this Central American country may need to carry proof of their vaccination.
In addition to measles vaccination, the U.S. CDC recommends being protected against chikungunya, dengue, malaria, typhoid, and Zika when visiting Honduras.
For local assistance during July 2025, the U.S. Embassy Tegucigalpa is available to U.S tourists.
Additionally, the Department of State stated in 2024, "Reconsider travel to Honduras due to civil unrest." The Smart Traveler Enrollment Program is available to travelers interested in receiving digital travel alerts while abroad.