Search API
When planning a European trip next year, many say the best time to visit Mediterranean Sea beaches is in the spring, when the weather is mild and before the summer crowds arrive.
Another consideration is when mosquitos transmit viruses such as dengue to vacationers.
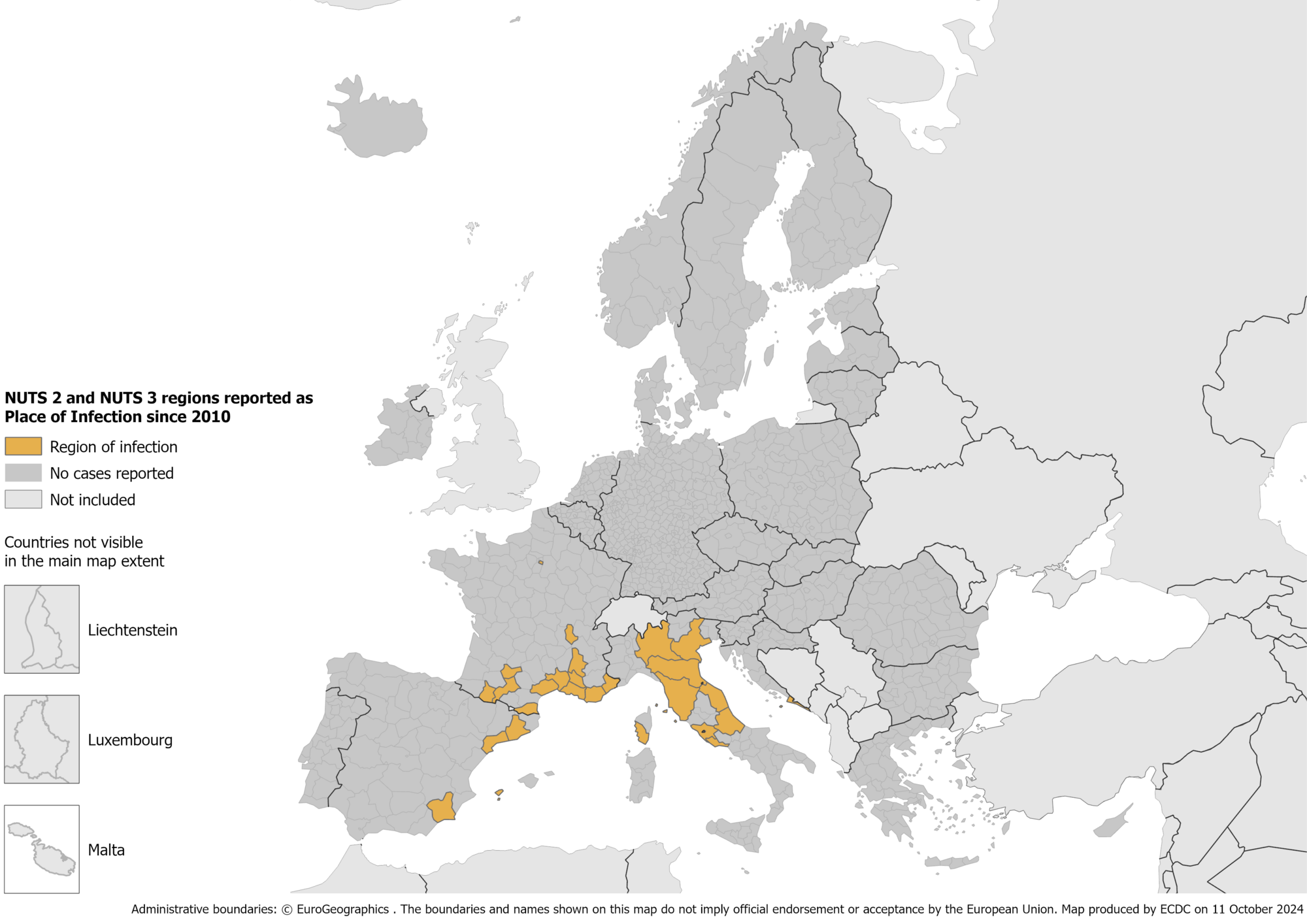
According to the European Centre for Disease Prevention and Control (ECDC), the recent three-month dengue outbreak rate measurably increased from July to September 2024.
As of October 2024, the ECDC reported locally acquired dengue cases in France, Italy, and Spain in 2024.
The Italian National Public Health Authority reported 194 locally acquired dengue cases in 2024. In 2023, 82 locally acquired dengue cases were reported.
In 2024, France reported 80 locally transmitted dengue fever cases and over 3,500 imported cases. In 2023, it reported nine dengue outbreaks that produced 45 autochthonous infections.
Spain reported eight locally acquired dengue cases in the Camp de Tarragona area of the Catalonia region in 2024. Last year, the Catalonia region reported just three local cases.
While unavailable in the United States, a second-generation dengue vaccine has been authorized in Europe since late 2022. Takeda's QDENGA vaccine remains approved in 2024 for use regardless of previous dengue exposure and without pre-vaccination testing.
The ECDC, U.S. CDC, and the WHO encourage travelers to speak with a travel vaccine expert about options to protect themselves from disease one month before departure.
The U.S. Centers for Disease Control and Prevention (CDC) reissued a Global Travel Health Notice on October 15, 2024, regarding Dengue outbreaks reported in 27 countries in the Americas, Africa/Middle East, and Asia/Pacific Islands.
In the Region of the Americas, over 12,076,930 Dengue cases and 7,171 related deaths have been reported this year.
In the United States, the CDC reported that 50 jurisdictions had reported 6,164 Dengue cases as of October 17, 2024, already exceeding reports from 2023.
For example, the Florida Health Department recently reported over 595 travel-associated and 47 locally acquired Dengue cases throughout the state.
The Texas Department of State Health Services says mosquitoes that transmit Dengue fever are found in Texas, with (78) imported cases reported in 23 counties.
In California, the Los Angeles County Department of Public Health reported eight locally acquired Dengue cases in the San Gabriel Valley this year.
While Dengue is a vaccine-preventable disease, no vaccine is currently available in the U.S.

Varicella zoster virus (VZV), one of 8 herpesviruses known to infect humans, “is a really sneaky virus,” said neurovirologist Maria Nagel, M.D., in an article posted by the JAMA Network on October 18, 2024.
Herpes Zoster, known as shingles, has been a vaccine-preventable disease for several years.
“Our aging population is growing, and it’s really concerning that 1 in 9 people over 65 have Alzheimer’s,” Nagel added. “It’s critical that we figure out what to do about this disease.”
Could the infection increase people's risk for dementia? Conversely, could shingles vaccination protect against dementia? And what about adult vaccines against other infections?
The unedited article - Researchers Are Exploring the Role of Shingles—and a Protective Role of Shingles Vaccine—in Dementia, is posted at this JAMA link.
As of October 19, 2024, various approved shingles vaccines are offered worldwide.

The U.S. Government has begun health screening air travelers who have been to the Republic of Rwanda in the last 21 days. This action is related to the recent Marburg virus outbreak in which 15 people have died.
Virgin Atlantic Airways confirmed that effective October 16, 2024, these Marburg virus-related screenings are a precautionary measure and will take place in Washington Dulles, Chicago O'Hare, and New York JFK.
This means any customer who has recently visited Rwanda will be rerouted through one of these airports to facilitate these mandatory screenings.
The Company says if you have booked directly with Virgin Atlantic and have been to Rwanda, don't hesitate to contact us to discuss your options.
As of October 19, 2024, Marburg vaccine candidates are being evaluated in various clinical trials, but the U.S. FDA has approved no vaccine.

The Federal Republic of Nigeria’s Federal Ministry of Health has been leading critical malaria control interventions, including introducing the R21/Matrix-M™ malaria vaccine as part of the country’s comprehensive strategy to combat the disease.
“The arrival of (800,000) malaria vaccines is a monumental step in our national efforts to reduce malaria morbidity and mortality,” said Professor Muhammad Ali Pate, Coordinating Minister of Health and Social Welfare, in a press release on October 17, 2024.
This announcement is critical to Nigerians.
According to the 2023 World Malaria Report, nearly 200,000 deaths from malaria occurred in Nigeria. In some regions, such as Kebbi State, this malaria prevalence rate is as high as 49%.
The malaria vaccine, which requires four doses, will be administered to children under one year of age as part of Nigeria’s Routine Immunization schedule. The first phase of the rollout will begin in Kebbi and Bayelsa States in November 2024.
On October 2, 2023, the World Health Organization recommended R21/Matrix-M to prevent malaria in children. Since then, it has been offered in various African countries.
The U.S. CDC says malaria vaccines reduce uncomplicated malaria by ~40%, severe malaria by ~30%, and all-cause mortality by 13%.
Neither approved malaria vaccine is available in the United States, but the CDC recommends that travelers going to Nigeria take prescription medicine to prevent malaria.
Additionally, the CDC has issued travel advisories for Nigeria's yellow fever, measles, polio, and diphtheria outbreaks.

When Johnson & Johnson (J&J) announced third-quarter earnings this week, its growth exceeded Wall Street’s expectations.
However, based on J&J's announcement on October 4, 2024, its future results will not include a Dengue product.
J&J confirmed the discontinuance of the Phase 2 field study to evaluate the efficacy of the investigational antiviral candidate mosnodenvir in preventing Dengue virus in adults. The decision to discontinue this study is part of a strategic reprioritization of the Company’s Communicable Diseases research and development portfolio.
This unfortunate news will bring essential focus to second-generation dengue vaccines and innovative vaccine candidates conducting late-stage clinical research in 2024, such as Butantan Institute's Butantan-DV tetravalent dengue vaccine.
As of October 18, 2024, Dengue's global outbreak continues unabated.

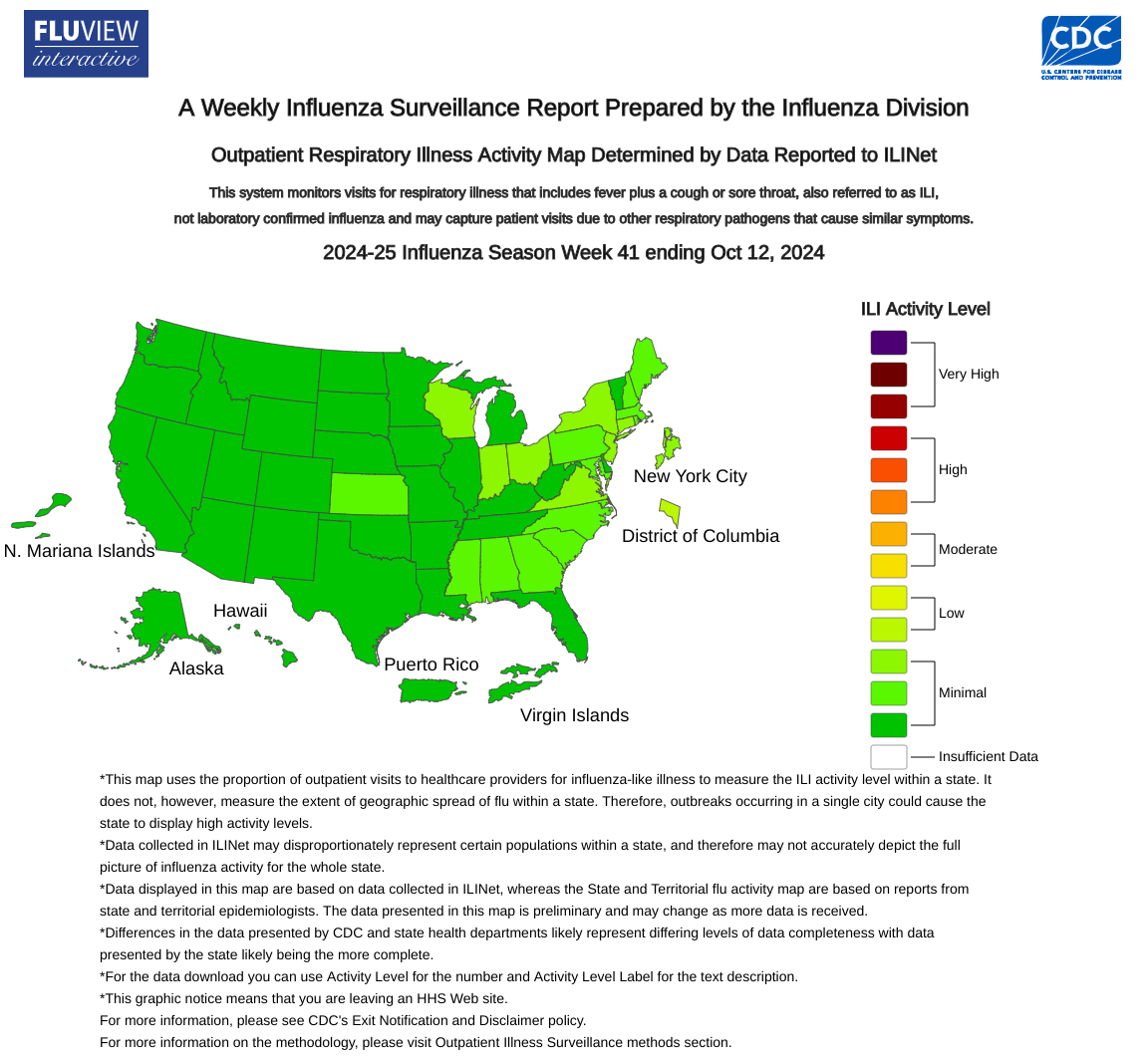
As the 2024-2025 flu season ramps up in the United States, new data reaffirms the protections offered by various influenza vaccines.
The results of the first pragmatic randomized study to evaluate the relative vaccine effectiveness of adjuvanted inactivated influenza vaccine (aIIV) compared to high-dose inactivated influenza vaccine (HD-IIV) for influenza prevention were published today.
The first season's results demonstrated that adjuvanted and high-dose influenza vaccines did not differ in effectiveness against Polymerase Chain Reaction-confirmed influenza with relative vaccine effectiveness of aIIV vs. HD-IIV of 1.5% (95% confidence interval: -8.4 to 10.5) among adults 65 years of age and older.
In addition, there was no difference in effectiveness between aIIV and HD-IIV for preventing hospitalization or emergency department visits for confirmed influenza and hospitalization for all-cause community-acquired pneumonia.
The data was presented during an oral presentation this week at the IDWeek 2024 conference.
The second season of the study is ongoing. Results may differ depending on the match between the vaccine and circulating influenza strains and other factors.
This study was funded by CSL Seqirus, which provided all the adjuvanted influenza vaccine doses to members of a large health system.
As of October 18, 2024, over 92 million flu shots have been distributed in the U.S. this season.