Search API
The World Health Organization (WHO) today announced the Access and Allocation Mechanism (AAM) for mpox has allocated an initial 899,000 vaccine doses for nine countries across the African region that are hit hard by the current Clade Ib mpox surge.
By the end of 2024, over 5.85 million vaccine doses are expected to be available to the Mpox Vaccines AAM.
As of November 6, 2024, the most significant number of JYNNEOS vaccine doses, 85% of the allocation, will go to the Democratic Republic of the Congo (DRC) as the most affected country, reporting 80% of laboratory-confirmed mpox cases in Africa in 2024.
Limited vaccination with Bavarian Nordic A/S MVA-BN® (JYNNEOS®) has recently begun in the DRC and Rwanda.
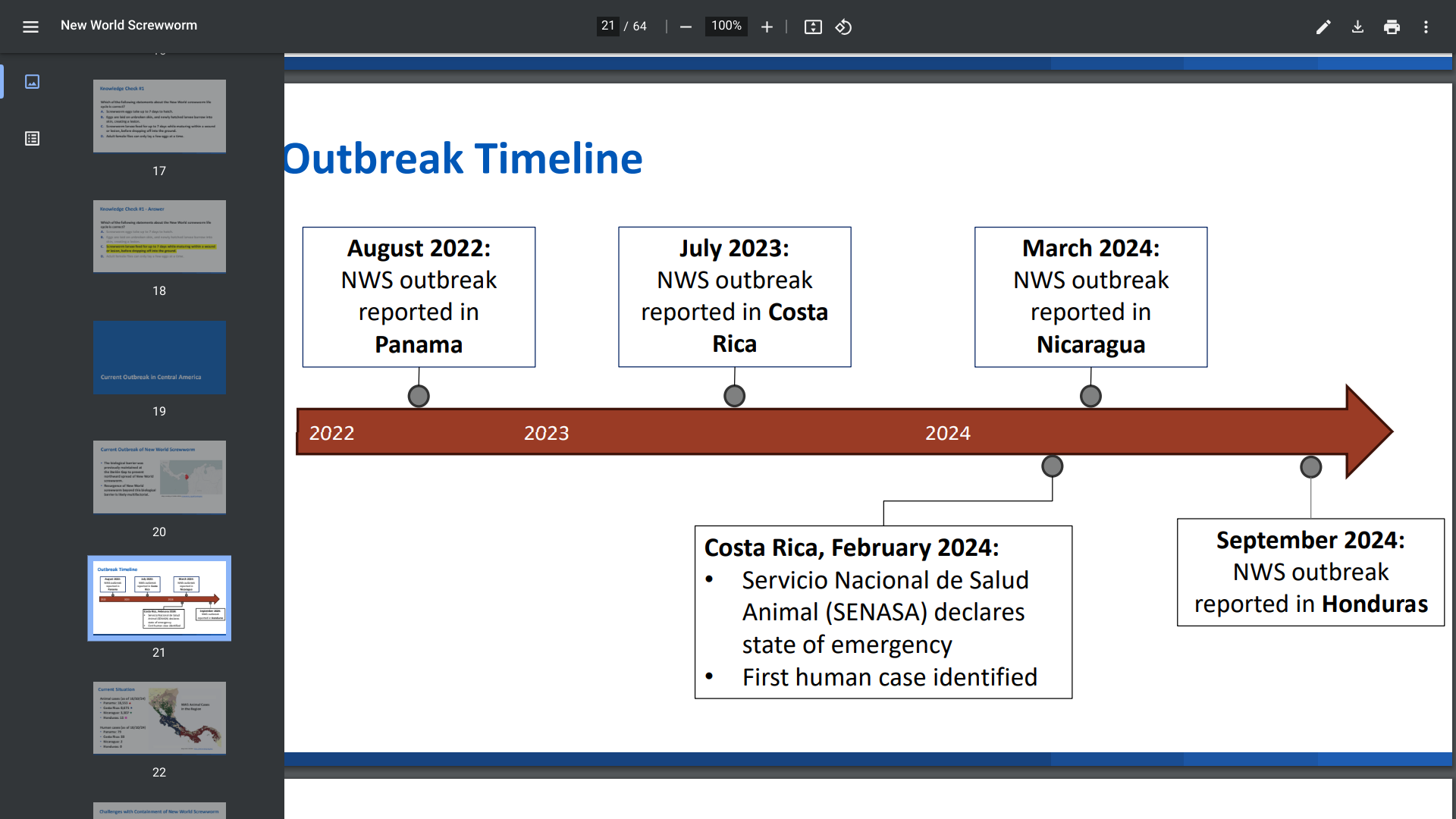
The Mediterranean coast of France is known to many vacationers for its lovely scenery and world-class resorts. In 2023, southern Europe alone accounted for about 500 million arrivals.
However, in 2024, mainland France may experience a record number of mosquito-transmitted diseases, mainly along the Mediterranean.
As of October 30, 2024, Public Health France reported over 4,000 imported cases of dengue, 18 of chikungunya, and 5 of Zika virus in 2024.
Furthermore, the European CDC recently confirmed that 82 locally-acquired dengue cases have been reported in Var (38), Alpes-Maritimes (19 cases), Vaucluse (18), and other departments.
While France has not yet declared dengue endemic, cases in 2024 have eclipsed the entire 2023 mosquito season. In 2023, France only reported 45 autochthonous (local) dengue infections.
From a disease prevention perspective, chikungunya and dengue have approved vaccines available, but Zika vaccine candidates have yet to be authorized in 2024.
While the U.S. CDC does not suggest these vaccines when visiting France, they are generally available in 2024. The CDC recommends that international travelers speak with a vaccine expert at least one month before traveling abroad.

GSK today announced that AREXVY has been approved in Canada to prevent lower respiratory tract disease caused by respiratory syncytial virus (RSV) in adults 50-59 years of age at increased risk for RSV disease.
In Canada, the vaccine was previously approved for use in adults aged 60 and older and is strongly recommended by the National Advisory Committee on Immunization (NACI) for all adults 75 years and older and those aged 60 and above in nursing homes and other chronic care facilities. NACI also recommends that adults aged 60-74 may consider RSV vaccination in consultation with a health care provider.
Michelle Horn, Interim Country Medical Director, GSK, said in a press release on November 5, 2024, “The natural age-related decline in immune function we all experience, which can increase our vulnerability to viruses like RSV, becomes more evident the older we get. Not surprisingly, the incidence of RSV-associated hospitalizations in adults starts to increase at the age of 50. For adults with underlying medical conditions, RSV can worsen these conditions and lead to serious consequences. We are proud to be the first to provide a vaccine to help protect Canadians aged 50-59 at increased risk of lower respiratory tract disease caused by RSV.”

As part of the Immunisation Agenda 2030, a World Health Organization (WHO) study published today in eBioMedicine named 17 pathogens that regularly cause diseases in communities as top priorities for new vaccine development.
In five out of six WHO regions, annual child deaths and contribution to antimicrobial resistance were the most heavily weighted criteria.
“Too often global decisions on new vaccines have been solely driven by return on investment, rather than by the number of lives that could be saved in the most vulnerable communities,” said Dr Kate O’Brien, Director of the Immunization, Vaccines and Biologicals Department at WHO, in a press release on November 5, 2024.
Pathogens where vaccines are approaching regulatory approval, policy recommendation, or introduction
- Dengue virus
- Group B Streptococcus
- Extra-intestinal pathogenic E. coli
- Mycobacterium tuberculosis (TB)
- RSV
Pathogens where vaccine research is needed
- Group A streptococcus
- Hepatitis C virus
- HIV-1
- Klebsiella pneumoniae
Pathogens where vaccines need to be further developed
- Cytomegalovirus
- Influenza virus (broadly protective vaccine)
- Leishmania species
- Non-typhoidal Salmonella
- Norovirus
- Plasmodium falciparum (malaria)
- Shigella species
- Staphylococcus aureus
This global prioritization exercise for endemic pathogens complements the WHO R&D blueprint for epidemics, identifying priority pathogens that could cause future epidemics or pandemics.

The U.K. Health Security Agency (UKSHA) announced today that two cases of Clade Ib mpox are currently under specialist care at Guy's and St Thomas' NHS Foundation Trust in London, England.
These are household contacts of the first mpox case reported last week.
As of November 4, 2024, this news brings the total number of confirmed mpox cases to three in London. These mpox cases are unrelated to the May 2022 Clade II outbreak.
In a press release, Professor Susan Hopkins, Chief Medical Adviser at UKHSA, said, "Mpox is very infectious in households with close contact, so it is not unexpected to see further cases within the same household."
"The overall risk to the U.K. population remains low. We are working with partners to ensure all contacts of the cases are identified and contacted to reduce the risk of further spread."
All contacts will be offered testing and vaccination as needed and advised on any necessary further care if they have symptoms or test positive.
In the U.K., the Bavarian Nordic A/S IMVAMUNE® (JYNNEOS®, MVA-BN®) mpox / smallpox vaccine is available in 2024.

Starting early next year, the Pan American Health Organization (PAHO) will provide countries of the Americas with access to the Pfizer-produced vaccine against respiratory syncytial virus (RSV).
Only the Pfizer vaccine has been approved by the World Health Organization to prevent RSV-related diseases in infants.
The PAHO stated on November 1, 2024, that around 13 million children are born in the region each year, and if the RSV vaccine is offered to pregnant women, they could benefit from this measure.
The PAHO reported in October 2024 that the new RSV season had started slowly in the Americas.
In the United States, the U.S. FDA-approved single-dose, extended half-life monoclonal antibody Beyfortus™ offers passive immunization to prevent lower respiratory tract infections caused by the RSV to newborns and infants experiencing their first or second RSV season.
In September 2024, the WHO's advisory group recommended that all countries introduce passive immunization to prevent severe RSV disease in young infants.

New World screwworm (NWS) infestations have continued to be reported in the Region of the Americas this year, and the U.S. government is concerned that NWS maggots may return after five decades.
According to a notice issued by the U.S. Department of Agriculture on November 1, 2024, NWS is endemic in Cuba, Haiti, the Dominican Republic, and countries in South America.
By October 26, 2024, Panama had reported 20,417 positive NWS cases, Costa Rica had 7,718, and Nicaragua had 4,384 cases in 2024.
In 2023, more than 6,500 NWS cases were detected in Panama.
The USDA says it eradicated NWS from the United States in 1966 using the sterile insect technique, in which sterile flies are released in the wild. Further, the USDA says there is a constant risk of NWS reintroduction in the United States.
In mid-October 2024, the U.S. CDC Clinician Outreach and Communication Activity call highlighted concerns about a potential northward spread of NWSs.
The cornerstone of a collaboration between the Panama-United States Commission for the Eradication and Prevention of Screwworms is releasing sterile male screwworm flies in the Darian Province of eastern Panama to create a biological barrier. This effort is scheduled to continue for an extended period.
As of November 4, 2024, there are no NWS vaccines available.