Search API
The Louisiana Department of Health today reported a patient who had been hospitalized with the first human case of highly pathogenic avian influenza (HPAI) H5N1 in Louisiana and the United States recently died.
Due to patient confidentiality and respect for the family, this will be the final update about the patient.
The patient was over the age of 65 and was reported to have underlying medical conditions. The patient contracted H5N1 after exposure to a combination of a non-commercial backyard flock and wild birds.
This report was not a case of H5N1 person-to-person spreading.
LDH’s extensive public health investigation has identified no additional H5N1 cases nor evidence of person-to-person transmission. This patient remains the only human case of H5N1 in Louisiana.
HAPI avian influenza (bird flu) is widespread globally and has been causing outbreaks in wild birds, poultry, mammals, and dairy cows for a few years. There have been 67 confirmed human cases of H5N1 bird flu in the United States since 2022.
As of January 7, 2025, the U.S. CDC says the current public health risk for H5N1 to the general public remains low. However, people who work with birds, poultry, or cows or have recreational exposure to them are at higher risk.
Furthermore, the best way to protect yourself and your family from H5N1 is to avoid sources of exposure. That means avoiding direct contact with wild birds and other animals infected with or suspected to be infected with bird flu viruses. Do not touch sick or dead animals or their droppings or bring sick wild animals into your home.
While one avian influenza vaccine has been approved for human use (Audenz™) by the U.S. FDA, and several vaccine candidates are in the U.S. stockpile, they are not CDC-authorized for use, nor are they commercially available in the U.S.

The California Department of Public Health (CDPH) recently announced that pertussis (whooping cough) cases are increasing throughout the state. It urged pregnant women and infant caregivers to receive tetanus, diphtheria, and pertussis vaccinations.
CDPH reported receiving reports of more than 2,000 patients, including 62 infants hospitalized during the last ten months of 2024. One of the hospitalized infants died from the pertussis infection.
This data represents about a 500% increase in cases than in 2023 when 400 pertussis cases were confirmed.
Dr. Tomás J. Aragón, CDPH Director and State Public Health Officer stated in a press release on January 3, 2025, "Infants are particularly vulnerable (the bacterial disease), and that's why CDPH especially encourages people who are pregnant to receive the Tdap vaccine in their third trimester, which will pass along protective antibodies to the baby."
Throughout the USA, the Centers for Disease Control and Prevention (CDC) note that six times as many pertussis cases were reported in 2024 compared to 2023.
Caused by Bordetella pertussis, people with whooping cough have severe coughing attacks that can last for months, according to the CDC. Infants too young for vaccination are at the most significant risk for hospitalization and death. Early signs of whooping cough are similar to common cold symptoms, such as a runny or stuffy nose and low-grade fever. Babies with whooping cough may have trouble breathing, turn blue, or stop breathing.
The CDC and CDPH recommend speaking with your doctor or pharmacist in 2025 about which vaccines are right for you and your family.

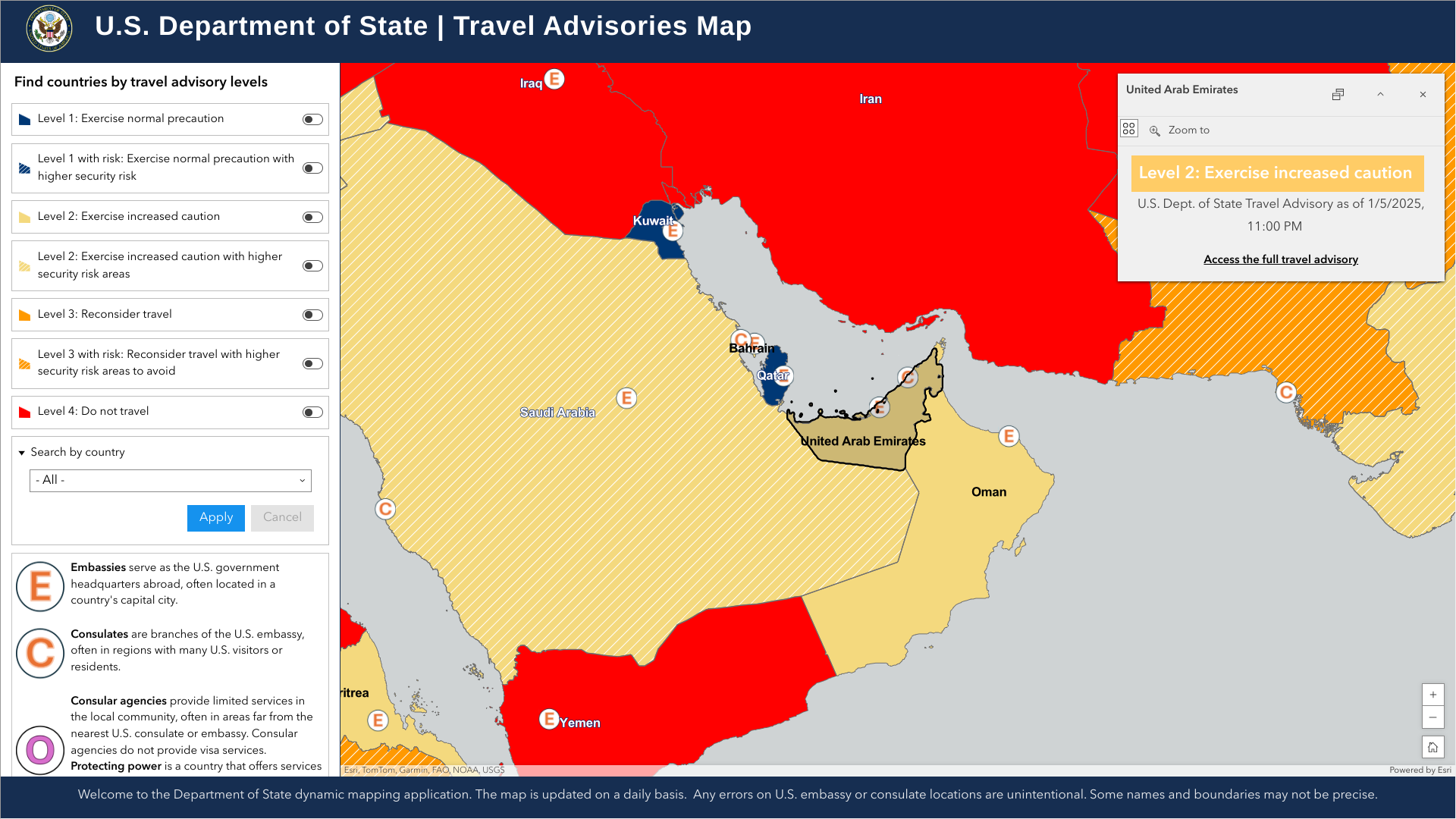
As 2025 kicks off, many international travelers will visit the United Arab Emirates, and according to government agencies, their stays should be healthy and safe.
On January 6, 2025, the U.S. Department of State reissued its Level 2 Travel Advisory, Exercise Caution, saying residents and visitors generally find the UAE a safe and secure environment. Still, regional tensions have aggravated risks for U.S. citizens.
Visitors should also be aware that the UAE has strict rules on social behavior.
Previously, the U.S. Federal Aviation Administration issued an advisory Notice to Air Missions recommending caution for U.S. air carriers and commercial operators operating in the Middle East, including the UAE.
The State Department suggests enrolling in the Smart Traveler Enrollment Program to receive Alerts and make locating you in an emergency easier. For local assistance, the U.S. Embassy in Abu Dhabi is located at Plot 38, Sector W59-02, Street No. 4.
From a health perspective, the U.K. and U.S. CDCs recommend updating travelers with vaccinations and boosters, including measles, mumps, and rubella. The UAE is included in the CDC's current Global Measles outbreak alert.
While there is no risk of yellow fever in the UAE, a vaccination certificate is required upon arrival at the airport.
In the U.S., yellow fever vaccinations are offered at travel pharmacies and clinics. And as of January 2025, several next-generation vaccine candidates are conducting clinical research.
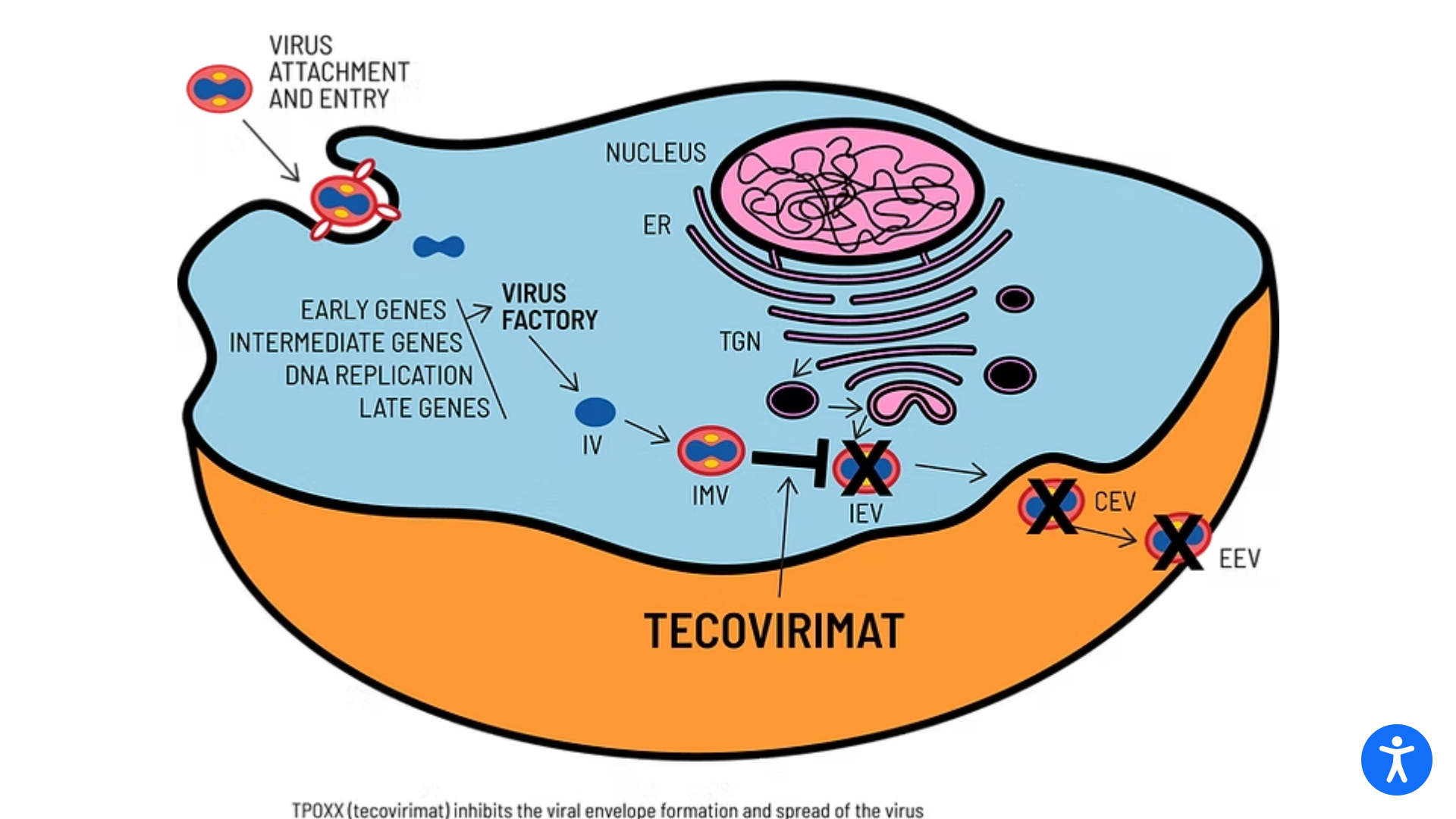
SIGA Technologies announced that its antiviral treatment TEPOXX (tecovirimat 200 mg capsules, TPOXX) had received regulatory approval in Japan for the treatment of smallpox, mpox, cowpox, as well as complications following smallpox vaccination in adults and pediatric patients weighing at least 13 kg. TEPOXX is the first antiviral therapy approved for the treatment of orthopoxviruses.
As of January 2, 2025, SIGA has delivered an order of TEPOXX to help build Japan’s strategic national stockpile.
TEPOXX is a highly targeted small-molecule antiviral that inhibits the VP37 protein found on the surface of all orthopoxviruses. By preventing the virus from exiting infected cells, TEPOXX slows the spread of the infection, enabling the immune system to clear the virus.
TPOXX is approved in the U.S. and Canada for the treatment of smallpox. In the European Union and the United Kingdom, marketed as Tecovirimat-SIGA, it is approved for treating smallpox, mpox, and cowpox and to treat complications following smallpox vaccination.
The Japanese approval is based on data from 15 clinical trials of oral TEPOXX in over 800 healthy volunteers, including a pivotal repeat-dose phase 1 pharmacokinetics (PK) trial involving 20 healthy volunteers conducted in Japan. TEPOXX has also been studied in NHPs infected with the variola virus, which causes smallpox, where TEPOXX demonstrated improved survival and reduced lesions.
In 2024, the U.S. NIH announced unsatisfactory results regarding mpox usage.
In 2024, the unfortunate leader in extensively drug-resistant (XDR) Shigellosis outbreaks was the state of California, with 4,365 of the 20,621 cases reported nationwide.
A Brief Report published by AJIC on December 9, 2024, describes a mutation of Shigella sonnei, a strain of Shigella bacteria resistant to five of the antibiotic classes most commonly prescribed for such infections.
This report describes a distinct genetic mutation that made the bacteria resistant to another class of antibiotics, the cephalosporins.
These researchers say this XDR Shigella strain appears unique to LA country.
"These cases highlight the rapid expansion of extensively drug-resistant Shigella in the United States and the urgent need for appropriate detection and management,' wrote these UCLA and Quest researchers.
Since first detected in California in 2017, XDR Shigella has been increasing in prevalence. By 2022, 3.2% of Shigella isolates were XDR. From January through May 2024, 12% (118/978) of California Shigella isolates were reported as XDR. Shigella sonnei isolates accounted for the most significant percentage (78%) of cases.
The U.S. CDC estimates that Shigellosis cases occur annually, making it the third most common bacterial enteric disease. As of week #52, ending December 28, 2024, the CDC confirmed 20,621 Shigella cases, led by California and New York (2,990).
In 2023, the CDC reported 17,176 cases in the U.S.
From a prevention perspective, Valneva SE and LimmaTech Biologics AG are co-developing the Shigella4V (S4V), a tetravalent bioconjugate vaccine candidate against Shigellosis. On November 13, 2024, the companies launched a Phase 2b clinical trial for Shigella4V.
Previously, the U.S. FDA granted Shigella4V Fast Track designation.

The Federative Republic of Brazil recently announced it broke its record for international tourists last year with over 6.621 million travelers, exceeding results from 2018.
This data, posted on January 3, 2025, indicates that Brazil is approaching its goal of attracting 8.1 million international tourists in the next three years.
However, health agencies such as the U.S. CDC suggest that tourists speak with travel vaccine experts before visiting Brazil in 2025 to learn how to prevent mosquito-transmitted diseases, such as chikungunya.
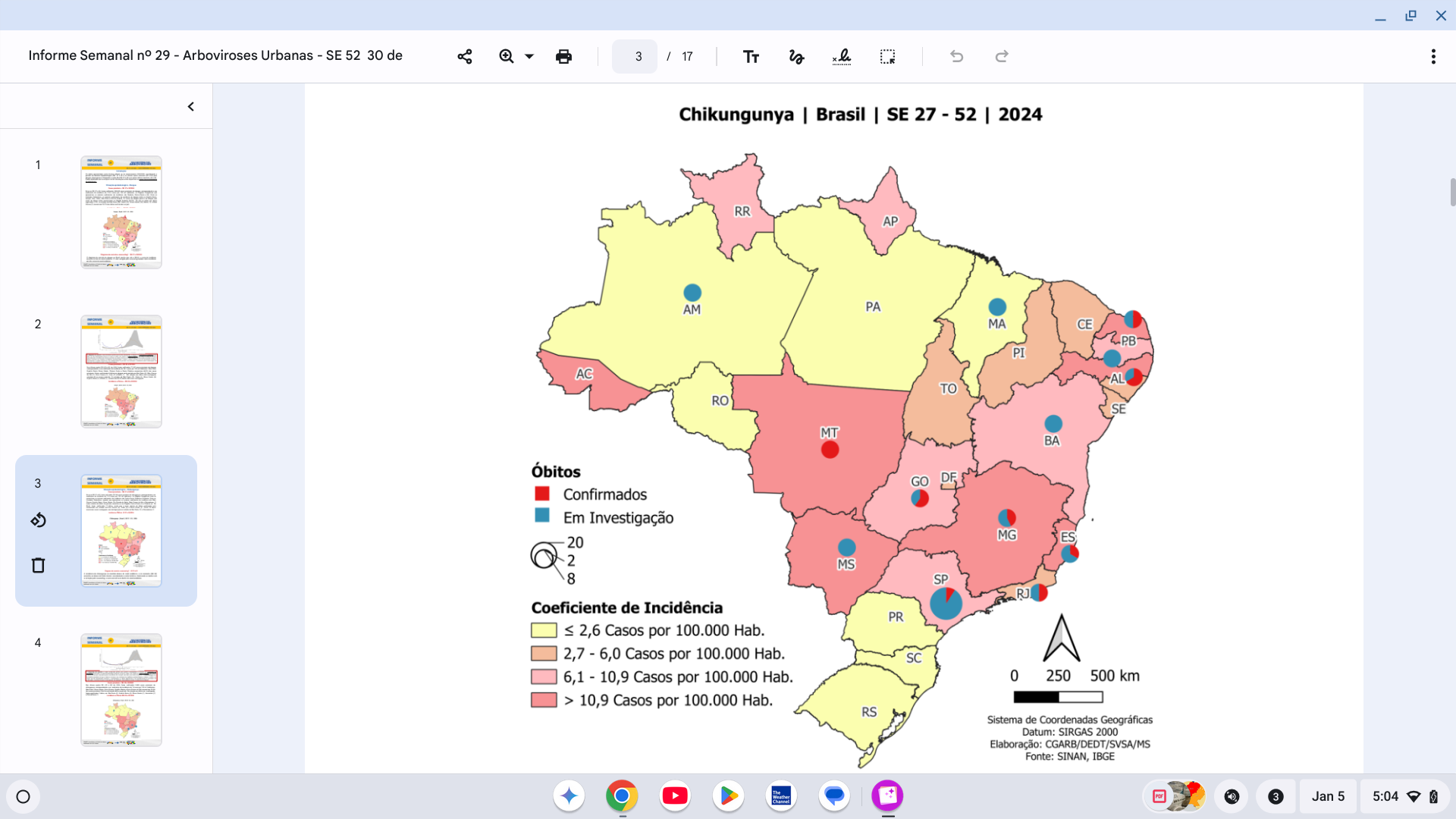
According to Brazil's Secretariat for Health and Environmental Surveillance, chikungunya outbreaks have been reported in Brazil since 2014. As of the end of 2024, the PAHO reported over 420,139 chikungunya cases and 236 related deaths throughout Brazil.
Chikungunya 'Indian Ocean Lineage' clusters with the highest risk were initially located in Brazil's northeast, followed by the central-west and coastal areas of São Paulo and Rio de Janeiro (2018–2021), and then increased in the northeast (2019–2021).
On October 28, 2024, a study reported that in 2023, during the epidemic in Minas Gerais, there was a mortality rate of 35.1/100,000 inhabitants.
When departing from the United States, the first U.S. FDA-approved chikungunya vaccine, Valneva SE's IXCHIQ®, is commercially available in 2025. IXCHIQ's approval in Brazil is pending.
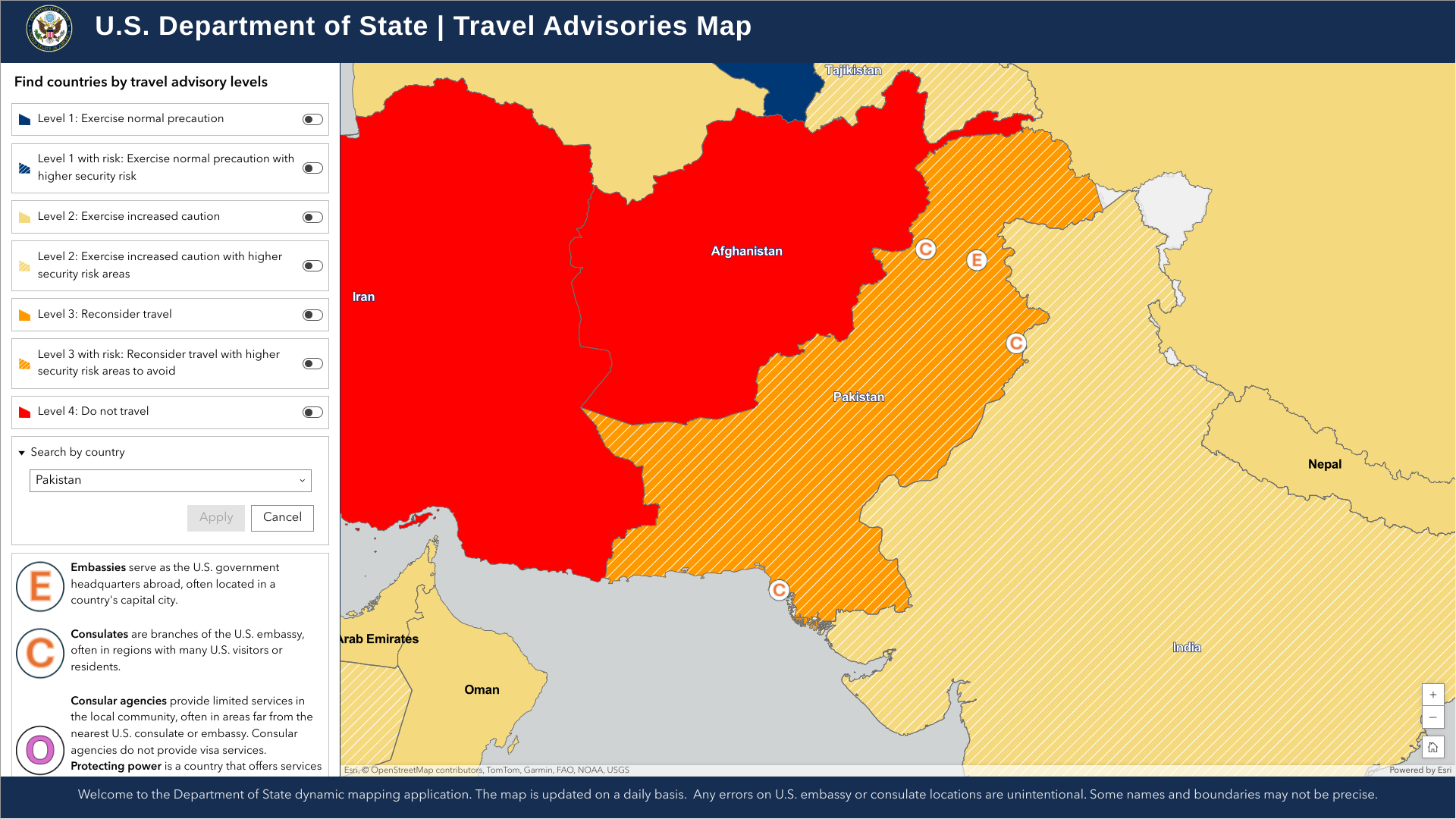
The Global Polio Eradication Initiative recently reported that the Republic of Pakistan has confirmed three cases of wild poliovirus type 1 (WPV1) infections in the Sindh, Balochistan, and Khyber Pakhtunkhwa provinces.
As of the end of 2024, the total number of WPV1 cases in Pakistan has reached 67.
Furthermore, 52 WPV1-positive environmental samples were reported last week, which indicates that additional polio cases will be confirmed in 2025.
The South Asian nation of around 240 million people has been declared as one of two countries (Afghanistan) where polio is endemic.
As VOA reported last year, a representative of Pakistan's Ministry of Health stated that polio eradication is the country's top priority. "Despite the recent resurgence, a unified plan aims to stop polio transmission by 2025."
According to the World Health Organization (WHO), countries affected by poliovirus circulation are subject to temporary recommendations issued by the Emergency Committee of the International Health Regulations on Poliovirus.
The WHO and the U.S. CDC recommend that all travelers to 36 polio-affected areas be fully vaccinated against polio. Residents and visitors of infected regions who have been there for more than four weeks should receive an additional oral or inactivated polio vaccine (IPV) within four weeks to twelve months of travel.
Globally, the type 2 novel oral polio (nOPV2) vaccine has been administered over 1 billion times in the past few years.
In the United States, the IPV is offered at travel clinics and pharmacies in 2025.
Seperately, the U.S. Department of State says, 'Reconsider travel to Pakistan as some areas have increased civil unrest risk.'