Search API
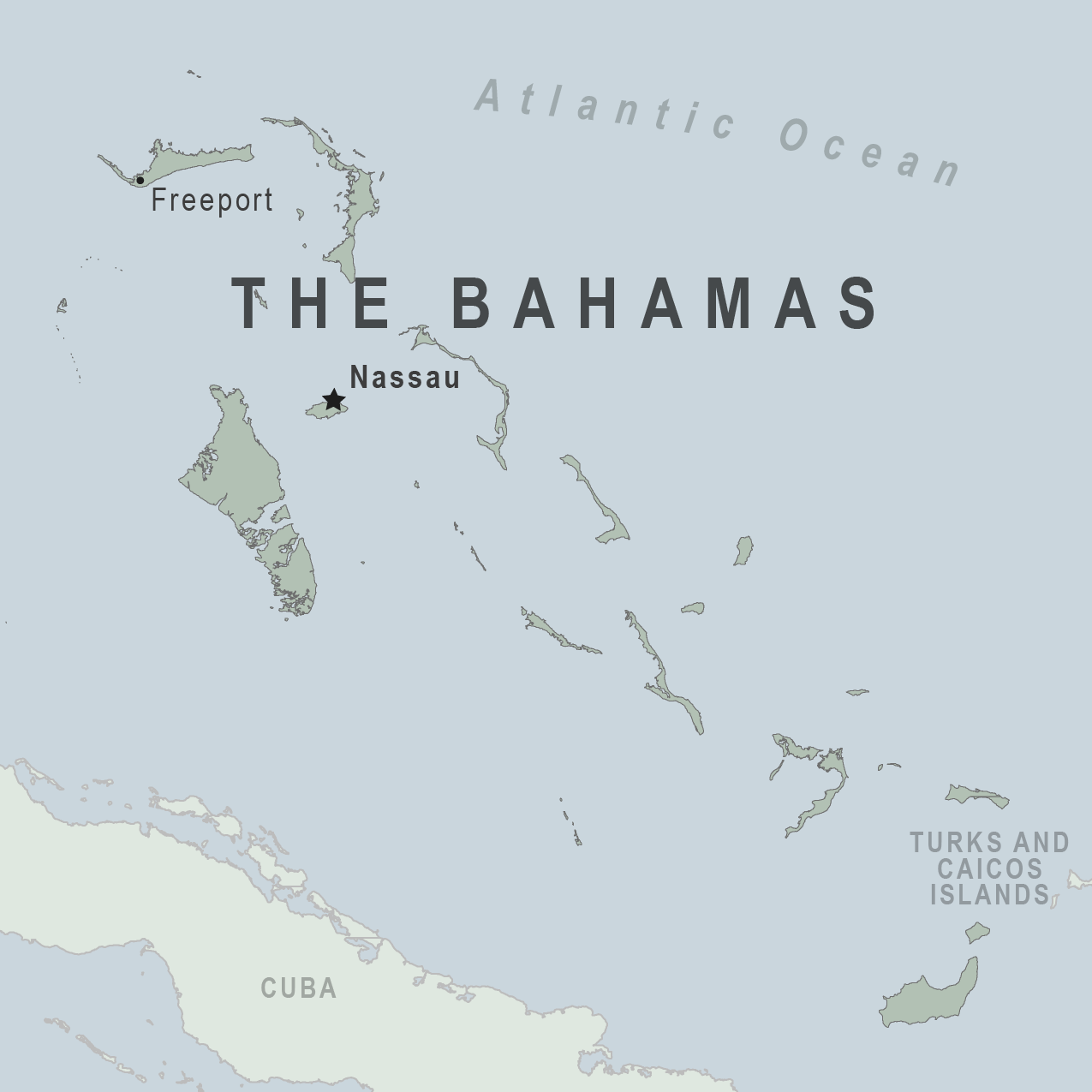
With warm Caribbean waters and beautiful beaches, The Bahamas is a tourist favorite in 2025. Last year, over 11 million people visited the various islands in The Bahamas.
To ensure visitors departing from the United States are appropriately forewarned about risks 60 miles east of Florida, the State Department recently updated its Level 2: Exercise Increased Caution.
As of March 31, 2025, this Advisory stated that crime can occur anywhere in The Bahamas, with frequent reports in Nassau and Freeport on New Providence and Grand Bahama islands. Additionally, visitors should stay alert in the "Over the Hill" area in Nassau, south of Shirley Street.
And stay alert in vacation rental properties, especially where private security is not present.
Furthermore, boating is not well-regulated, as sexual assaults, injuries, and deaths have occurred. Moreover, never swim alone and beware of jet skis or other watercraft operating close to shore.
Due to these risks, U.S. government employees are prohibited from using jet skis and personal watercraft rented from independent operators on New Providence and the Paradise Islands.
And be aware of your surroundings and stay alert in public restrooms, as sexual assaults have occurred on and near downtown Nassau beaches.
For local assistance, the U.S. Embassy Nassau is located at 42 Queen Street, Nassau, The Bahamas. You can enroll in the Smart Traveler Enrollment Program to receive digital messages from the Embassy and make locating you in an emergency easier.
From a health risk perspective, the U.S. CDC recommends various routine and travel vaccinations before visiting the Bahamas in 2025. Unlike its Caribbean neighbors, no mosquito-transmitted, vaccine-preventable Chikungunya or Dengue cases were reported in 2025 or 2024.
Travel vaccines are offered at various clinics and pharmacies in the U.S.
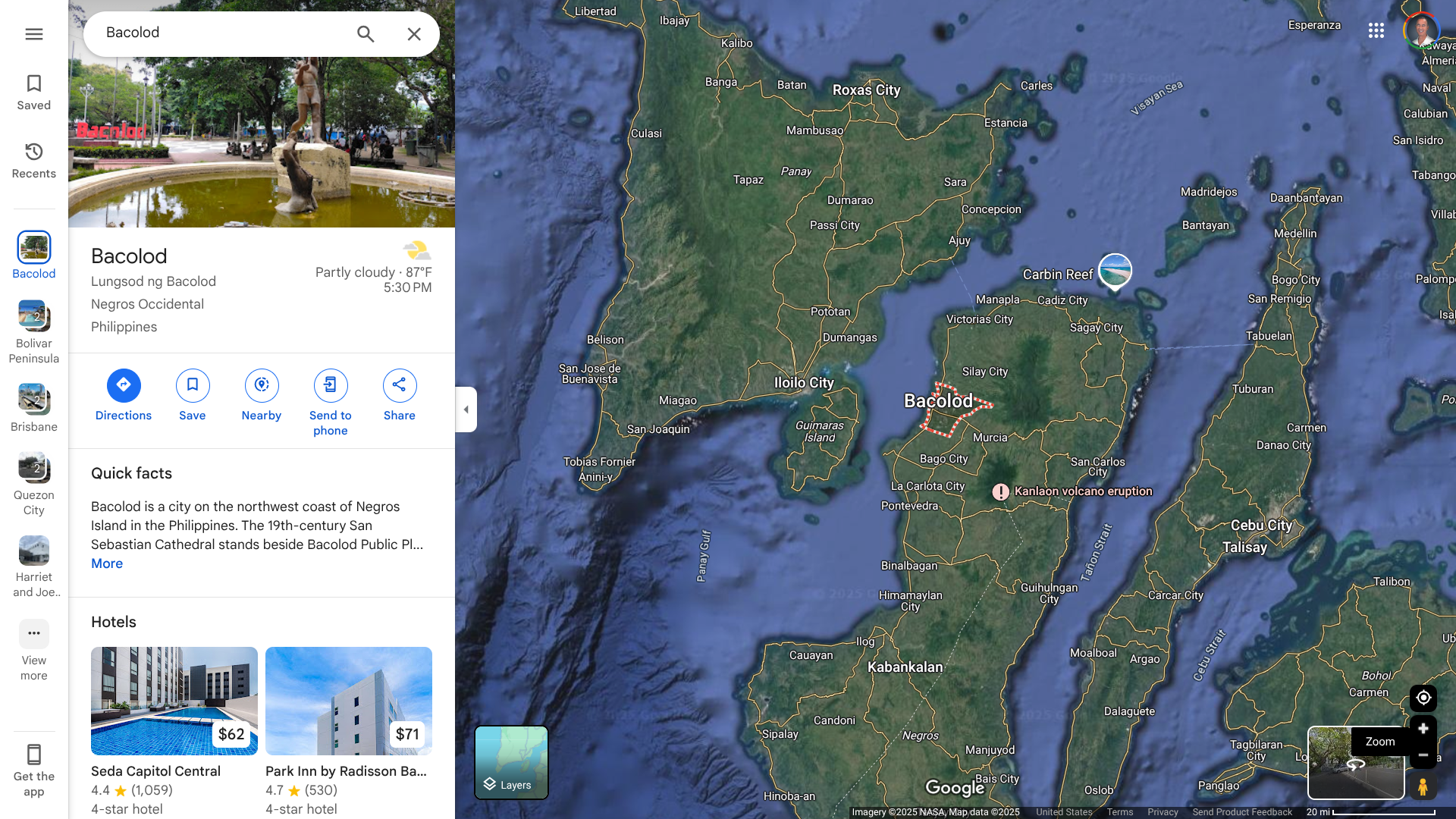
In the Philippines, the Bacolod City Health Office (CHO) recently issued an advisory regarding increasing cases of rotavirus and typhoid.
Typhoid is caused by the bacteria salmonella typhi, which spreads through contaminated food and water or close contact with an infected person.
Data showed that from January to March 22, some 27 cases of typhoid, also known as enteric fever, were reported, compared to only 11 cases during the same period in 2024.
Meanwhile, the CHO reported 74 cases of rotavirus, the most common cause of severe diarrhea among children, during the same period in 2025.
The top risk factor for rotavirus is poor sanitation and hygiene, leading to contamination of food and water.
In its advisory, the CHO said prevention includes washing hands with soap and clean water after using the toilet and before eating, as well as drinking only clear water. If unsure, boil water for three minutes or chlorinate water.
“Go to the nearest barangay health station or center, or consult with your physician if experiencing signs and symptoms of typhoid fever,” Dr. Grace Tan, head of the CHO Environment Sanitation Division, said in a press release on April 2, 2025.
Bacolod City, located on the northwest coast of Negros Island in the Philippines, has become a tourist favorite and was visited by over 800,000 people last year.
The U.S. CDC's Travel Health Advisory for the Philippines recommends vaccination for most travelers, especially those staying with friends or relatives or visiting smaller cities or rural areas.
When departing from the United States in 2025, most travel clinics and pharmacies offer vaccines that prevent both typhoid and rotavirus.
Additionally, the CDC highlights vaccines for chikungunya, dengue, and measles when visiting the Philippines.
The Republic of Uganda's Ministry of Health today confirmed it is the 19th country in Africa to introduce an approved malaria vaccine into routine immunisation.
The R21/Matrix-M™ vaccine includes Novavax AB's proprietary saponin-based Matrix-M adjuvant, will initially target 1.1 million children under two years in 105 high- and moderate-transmission districts across Uganda, with plans to expand nationwide.
About 2.278 million doses of malaria vaccines were distributed.
As of April 2, 2025, a malaria vaccination campaign was launched in Apac District, northern Uganda, with support from Gavi, the Vaccine Alliance, and Alliance partners UNICEF, the World Health Organization (WHO), PATH, and CHAI.
The Minister of Health, Dr Jane Ruth Aceng Ocero, commented in a GAVI press release, “The vaccine's introduction marks a significant milestone in our fight against malaria. It is expected to prevent at least 800 cases of severe malaria among children daily and ease the financial burden on families, saving them approximately UGX 15,000 per case that would have been spent on treating severe malaria."
According to the WHO, despite the expenditure of $4 billion per year, malaria deaths and cases have not substantially changed over the last decade. An estimated 263 million malaria cases and 597,000 related fatalities occurred worldwide in 2023.
Recently, Uganda's tourism sector has experienced a significant increase, with thousands of international visitors arriving monthly.
As of April 2025, malaria vaccines are not offered in the United States.

At the end of March 2025, the Republic of Panama's Ministry of Health announced it is strengthening preventive measures in the community of Las Guias in the district of Calobre, due to an increase in malaria cases.
As of March 31, 2025, 66 malaria cases were reported in the province, 6 of which were in Las Guías and La Raya de Calobre.
These malaria infections were confirmed in an area where a sugar harvest occurs in the Ngäbe-Buglé region.
Over 100 years ago, building the Panama Canal generated malaria infections that cost many lives.
In a press release, Alex Sánchez, regional head of Public Health, emphasized that the Vector Control and Epidemiology departments continue to conduct research, case detection, teaching, and preventive operations, providing timely treatment and care.
Furthermore, it is essential to eliminate mosquito breeding sites to prevent all mosquito-transmitted diseases, including Dengue.
During the first three months of 2025, Panama has reported over 4,800 Dengue cases and four related fatalities.
Last year, the Pan American Health Organization reported Panama confirmed 37,231 Dengue cases and 52 fatalities.
There is also an outbreak of Oropouche in Espírito Santo, Brazil, and Darién Province.
Like Dengue and Malaria, Oropouche is spread primarily through the bites of infected midges and mosquitoes.
While the U.S. CDC has issued Travel Health Advisories for Panama, as of April 2, 2025, it has not authorized the use of preventive vaccines against Dengue and Malaria.
A second-generation Dengue vaccine, QDENGA®, is offered in various countries in the Americas.
Moreover, malaria vaccines have also been approved for use in Africa.
According to cruise ship news in early 2025, passing through the Panama Canal does not pose a significant health risk for passengers!

As the respiratory syncytial virus (RSV) season diminishes in 2025, most European adults are gaining access to an innovative vaccine.
Pfizer Inc. today announced that the European Commission (EC) has issued a decision amending the marketing authorization for ABRYSVO®, the company’s bivalent RSV prefusion F vaccine, to extend the indication to include prevention of lower respiratory tract disease (LRTD) caused by RSV in individuals 18 through 59 years of age.
This action by the EC expands the previous authorization for individuals aged 60 and older, and ABRYSVO now offers the broadest RSV vaccine indication in the EU.
“We are thrilled that ABRYSVO is now approved in the EU to help prevent RSV in adults aged 18 and older, which causes approximately 158,000 adult hospital admissions annually from RSV disease, a common respiratory virus with symptoms that can be severe or even life-threatening,” said Alexandre de Germay, Chief International Commercial Officer, Executive Vice President, Pfizer, in a press release on April 1, 2025.
In Europe, RSV is a common respiratory virus that causes mild, cold-like symptoms. However, in infants under six months of age, people over 65, and people with a compromised immune system, RSV can cause severe illness and death.
According to the ECDC on April 2, 2025, RSV activity peaked in the EU/EEA in week 52, 2024, and has since decreased, but has remained elevated, with considerable variation between countries.
Before visiting Europe, travel clinics and pharmacies in the United States offer RSV vaccination services.

The U.S. Centers for Disease Control and Prevention (CDC) today confirmed an ongoing outbreak of clade I mpox in Central and Eastern Africa. Previous data indicates that about 15 million people visit central Africa each year.
As of April 1, 2025, the CDC updated its Level 2 Travel Health Advisory saying, 'There is ongoing person-to-person transmission of mpox in Burundi, Central African Republic, Democratic Republic of the Congo (DRC), Kenya, the Republic of the Congo, Rwanda, Tanzania, Uganda, and Zambia.'
Person-to-person transmission has occurred through various means during this outbreak.
There are two types of Monkeypox virus. Historically, clade I has been associated with a higher percentage of people with mpox developing severe illness or dying, compared to clade II. The global outbreak of clade II began in May 2022.
The CDC writes, 'Mpox vaccination is recommended for people who anticipate the following sexual activities during travel to countries with ongoing person-to-person transmission of clade I mpox.'
In the United States, the Bavarian Nordic JYNNEOS® (MVA-BN®, IMVAMUNE®, IMVANEX®) two-dose vaccine is commercially offered at various travel clinics and pharmacies in April 2025.
Those eligible for mpox vaccination should get two doses of JYNNEOS at least 28 days apart, before visiting an mpox outbreak area.

As the global outbreak of the Chikungunya virus continues to expand in India, Brazil, and La Réunion, European adolescents can now receive an effective vaccine that protects them from this mosquito-transmitted disease.
On April 1, 2025, Valneva SE announced that the European Commission (EC) has granted marketing authorization in Europe for Valneva’s single-dose vaccine, IXCHIQ®, for the prevention of disease caused by the chikungunya virus in individuals 12 years of age and older.
With this approval, IXCHIQ® will become available for adolescents in the EU, Norway, Liechtenstein, and Iceland.
In addition to the EC's approval in adolescents and adults in the EU, it has been approved in the United States, Canada, and the United Kingdom to prevent diseases caused by the chikungunya virus in individuals 18 and older. Similar label extension applications for adolescents were also submitted in the U.S., Canada, and the U.K.
Dr. Richard Hatchett, Chief Executive Officer of the Coalition for Epidemic Preparedness Innovations, commented in a media release, “Cases of chikungunya are increasing around the world, making populations of all ages vulnerable to the disease’s long-term debilitating effects, such as prolonged joint pain and inflammation."
"EC’s marketing authorization for use of IXCHIQ® in adolescents in the EU is an important stepping stone that could help accelerate the approval of the vaccine in this age group in other regions, including areas where the disease is endemic.”
In the United States, IXCHIQ is commercially available at most travel clinics and pharmacies and is recommended by the U.S. CDC for people visiting Chikungunya endemic areas in 2025.

With clade I mpox outbreaks occurring in hard-to-reach areas in Central and Eastern Africa, a new version of an effective vaccine has been approved.
Bavarian Nordic A/S announced that the U.S. Food and Drug Administration (FDA) has approved the freeze-dried formulation of the JYNNEOS® vaccine to prevent smallpox and mpox disease in adults 18 and older.
Announced on March 31, 2025, this FDA approval will enable additional flexibility for stockpiling against a smallpox event or mpox outbreak.
The Company recently informed the African CDC that it can manufacture about ten million vaccine doses in addition to current orders by the end of 2025.
The current liquid-frozen formulation of JYNNEOS, approved by the FDA in September 2019, has specific cold-chain requirements, while the freeze-dried formulation provides advantages in terms of transportation, storage conditions, and shelf life, all of which are essential factors for long-term stockpiling.
“Today’s FDA approval represents a significant milestone in our development of this next generation of JYNNEOS and in our collaborative efforts with the U.S. government to strengthen public health security,” said Paul Chaplin, President and CEO of Bavarian Nordic, in a press release.
“As a long-term supplier of JYNNEOS to the U.S. biological preparedness, we are committed to supporting the government’s efforts to protect its citizens against current and future public health threats.”
To alert international travelers of this continued health risk, the U.S. CDC maintains Level 2 Travel Health Advisories.
JYNNEOS, a two-dose vaccine based on a live, attenuated vaccinia virus, is commercially available in pharmacies and clinics in the United States.
Internationally, JYNNEOS is known as MVA-BN®, IMVAMUNE®, and IMVANEX®.

Following the launch of the Global Polio Eradication Initiative in 1988, the number of paralytic poliomyelitis cases was reduced by about 99% globally, with wild-type PV1 remaining endemic in only Afghanistan and Pakistan.
However, the World Health Organization recently reconfirmed that the spread of the poliovirus remained a Public Health Emergency of International Concern. For example, the U.S. CDC identified polio outbreaks and poliovirus detections in 39 countries in 2025.
To address these concerns, a recent study highlighted the benefits of an innovative polio vaccine candidate.
According to a study published by NPJ Vaccines on March 31, 2025, the success of the poliovirus (PV) vaccines has enabled the near-eradication of wild PV. Their continued post-eradication use poses concerns due to the 'potential for virus escape during vaccine manufacture.'
While the current generation of PV vaccines has achieved great success, the continued use of oral PV has facilitated the continued appearance of circulating vaccine-derived PV (cVDPV), which now outnumbers wild PV cases yearly.
These researchers wrote, 'Recombinant virus-like particles (VLPs) that lack the viral genome remove this risk.'
They demonstrate the production of PV VLPs for all three serotypes by controlled fermentation using Pichia pastoris.
The cryo-EM structure of a new PV2 mutant, SC5a, was determined compared to PV2-SC6 b VLPs described previously, and the immunogenicity of PV2-SC5a VLPs was investigated.
Finally, a trivalent immunogenicity trial using bioreactor-derived VLPs of all three serotypes in the presence of Alhydrogel adjuvant showed that these VLPs outperform the current IPV vaccine in the standard vaccine potency assay, offering the potential for dose-sparing.
Overall, 'these results provide further evidence that yeast-produced VLPs have the potential to be a next-generation polio vaccine in a post-eradication world.'
'The most important pre-requisite of any next-generation PV vaccine is that it elicits the same long-lasting immunity against disease as the current vaccine.'
Over the past few years, PT Biofarma has produced the type 2 novel oral polio (nOPV2) vaccine, derived from the live, infectious virus and 'triple-locked.' The nOPV2 vaccine is reported to be more genetically stable than previous oral polio vaccines, with a lower risk of reversion to neurovirulence.
It has been administered over 1.1 billion times, mainly in Africa.
Since 2000, the IPV has been the standard polio vaccine offered in the United States, and booster doses are suggested when visiting poliovirus outbreak areas.

