Search API
The Coalition for Epidemic Preparedness Innovations (CEPI) and the University of Oxford today announced a strategic partnership to accelerate the development of vaccines against 'Disease X,' the threat of unknown pathogens with the potential to cause pandemics.
An outbreak of a future Disease X is inevitable, says CEPI.
Up to $80 million of CEPI funding will support the University of Oxford's team of vaccine scientists to develop prototype vaccines against high-risk viral families, which could be swiftly adapted if a new viral threat is identified.
This would pave the way for developing new vaccines, based on Oxford's ChAdOx technology and other rapid response vaccine platforms, within just 100 days of a virus with pandemic potential emerging.
Dr. Richard Hatchett, CEO of CEPI, commented in a press release on August 29, 2023, "CEPI's strategic partnership with the University of Oxford will make a vital contribution to our work to drive forward the 100 Days Mission."
Earlier in August 2023, CEPI and Gennova Biopharmaceuticals Ltd announced a new $3.6 million funding agreement to advance the development and optimization of a self-amplifying mRNA platform to develop vaccine candidates against Disease X.
In February 2018, Disease X was included in the updated WHO R&D Blueprint list of priority diseases.
Globalization, urbanization, and climate change increase the likelihood and frequency of infectious disease outbreaks. Still, that inevitability does not mean the world is destined to relive the devastating impacts of the recent pandemic.
The scientific advances forged in response to the pandemic have equipped the world with the tools and concepts that would enable us to interrupt outbreaks in the future before they spiral out of control.
Key to the success of the 100 Days Mission is rapid response vaccine technology platforms that can be used to design vaccines in a matter of days.
ChAdOx is one of only a handful of these technologies with proven capability as a platform on which safe and effective vaccines can be quickly developed and manufactured at scale and low cost.
Building on these advances, CEPI is at the forefront of global efforts to develop vaccines against the next Disease X in 100 days. Known as the 100 Days Mission, this goal has been embraced by the G7, G20, and industry leaders.

Valneva SE today announced that Health Canada has completed screening validation of the Company's regulatory application for marketing approval of its single-shot chikungunya vaccine candidate VLA1553 in persons aged 18 years and above and has determined that the New Drug Submission (NDS) application is sufficiently complete to permit a substantive review.
Based on Health Canada's performance standard to process an NDS application, the Company believes the regulatory review could be completed by mid-2024.
VLA1553 is currently the only chikungunya vaccine candidate worldwide for which regulatory review processes are underway. It could become the first licensed chikungunya vaccine to address this unmet medical need if approved.
Juan Carlos Jaramillo, MD, Chief Medical Officer of Valneva, commented in a press release on August 29, 2023, "Chikungunya represents a major threat for people traveling to or living in areas where chikungunya virus and the mosquitos that transmit it are present, including popular destinations for U.S. and Canadian travelers."
"This threat continues to grow as shown by the recent epidemiological alert issued by the Pan American Health Organization."
"No vaccine or specific treatments are currently available for this debilitating disease, and we will continue to work diligently to make VLA1553 available in different territories as quickly as possible."
Chikungunya is a mosquito-borne viral disease caused by the chikungunya virus (CHIKV), a Togaviridae virus transmitted by Aedes mosquitoes. Infection leads to symptomatic disease in up to 97% of humans after four to seven days following the mosquito bite.
While mortality with CHIKV is low, morbidity is high.
Clinical symptoms include acute onset of fever, debilitating joint and muscle pain, headache, nausea, rash, and chronic arthralgia. Chikungunya virus often causes sudden large outbreaks with high attack rates, affecting one-third to three-quarters of the population in areas where the virus is circulating.
As of July 2022, more than three million cases have been reported in the Americas, and the economic impact is considered significant.
There are no preventive vaccines or effective treatments available, and, as such, chikungunya is considered to be a major public health threat.
This vaccine was granted PRIority MEdicine designation by the European Medicines Agency in 2020 and also received FDA Fast Track, Breakthrough Therapy, and Priority Review designations in 2018, 2021, and 2023, respectively.
Other Chikungunya vaccine candidates are also seeking approval.
During the summer of 2023, the European CDC reported autochthonous / non-travel-associated dengue cases in Europe, from France and Italy.
Following investigations carried out by Bouches-du-Rhône, two new cases of autochthonous Dengue were identified in Gardanne. There are now 4 cases in France this year.
In the French Antilles, Martinique and Guadeloupe have entered an epidemic phase, as mentioned by Santé Publique France in an August 2023 press release.
Dengue cases have also been reported in Réunion and French Guiana this year.
On 18 and 20 August 2023, unrelated to travel, two confirmed cases of Dengue were reported by the Lombardy and the Lazio Regions in Italy. As of July 2023, 47 confirmed dengue cases were associated with overseas travel.
As of late August 2023, the U.S. CDC continues alerting international travelers of their health risks when visiting dengue outbreak areas. The CDC says that dengue is spread to people through the bite of an infected Aedes mosquito.
CSL Seqirus today announced it was selected by the Biomedical Advanced Research and Development Authority (BARDA) to deliver one bulk lot of H5N8 A/Astrakhan antigen to the U.S. government.
This acquisition of a bulk lot will increase BARDA's stockpile of vaccines to support rapid response in an associated influenza pandemic.
CSL Seqirus has been working with BARDA in a longstanding partnership for over a decade, including numerous R&D and manufacturing activities and awards supporting BARDA's pandemic preparedness objectives.
Confirmed on August 28, 2023, this is the third award CSL Seqirus has received from BARDA in the last two years related to the ongoing outbreak of HPAI in the United States.
As of August 2023, the Pan American Health Organization reported H5N1 viruses (clade 2.3.4.4b) had been detected in 15 countries in Latin America, the Caribbean, the United States, and Canada over the past 18 months.
This award follows the February 2022 award to produce an H5N8 A/Astrakhan virus vaccine seed and the subsequent October 2022 announcement of the selection of CSL Seqirus to deliver an H5N8 A/Astrakhan virus vaccine candidate for assessment in a Phase 2 clinical study.
While the likelihood of sustained human-to-human transmission of bird flu is "low," according to the U.S. Centers for Disease Control and Prevention (CDC) and World Health Organization, there have been reported a small number of human cases of avian influenza A(H5), including one in the U.S. in April 2022, a case in Ecuador in January 20235 and Chile in March 2023.
"While human cases are rare, sporadic, and isolated, consistent detection of bird and mammalian cases demands vigilance," commented Marc Lacey, Executive Director, Pandemic Response Solutions, CSL Seqirus, in a press release.
"Ongoing surveillance and preparedness efforts are critical to minimize the public health risk."
CSL Seqirus used its cell-based influenza vaccine technology, as utilized for FDA-approved AUDENZ™ (Influenza A(H5N1) Monovalent Vaccine, Adjuvanted), to manufacture the H5N8 A/Astrakhan bulk vaccine at the company's Holly Springs, North Carolina, facility, which was built in partnership with BARDA.
CSL Seqirus has established and will maintain the required pandemic readiness to deliver 150 million doses of cell-based pandemic influenza vaccine within six months of an influenza pandemic declaration in the U.S.
This $46.3 million project has been supported in whole or in part with federal funds from the Department of Health and Human Services, Administration for Strategic Preparedness and Response; BARDA, under contract number 75A50122D00004.

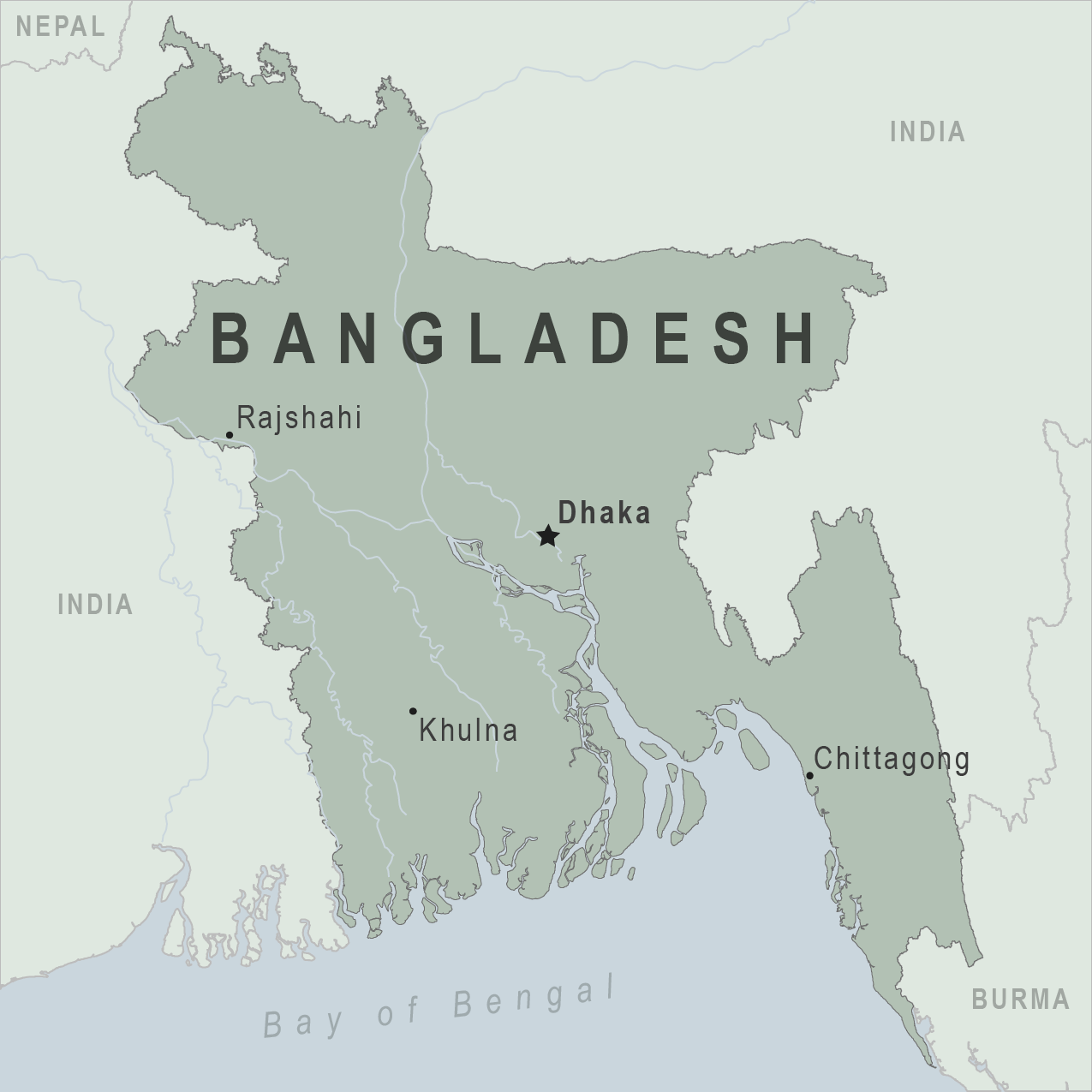
According to media reports, the Dengue outbreak in Bangladesh accelerated in August 2023.
The Dhaka Tribune reported on August 26, 2023, that Bangladesh had its worst day ever for Dengue, with nine deaths and 1,960 hospitalized in a day.
This news increases Bangladesh's total number of dengue-related deaths to 537 in 2023.
Dengue is endemic in Bangladesh, resulting in high morbidity and mortality.
The World Health Organization says Dengue is the fastest-spreading mosquito-borne infectious disease and has emerged as a global public health problem.
As of August 28, 2023, two approved and various Dengue vaccine candidates are in development.
In addition to the ongoing Dengue outbreak, the U.S. CDC suggests various travel vaccines before visiting Bangladesh in 2023.
Valneva SE today reported positive initial Phase 3 safety data in adolescents for its single-dose chikungunya virus (CHIKV) vaccine candidate VLA1553.
On August 28, 2023, Valneva announced the initial safety data generated in the ongoing trial VLA1553-321, Valneva's first clinical trial in an endemic area (Brazil) and with individuals previously infected with CHIKV, showed that VLA1553 was generally safe and well tolerated in adolescents aged 12 to 17 years, regardless of previous CHIKV infection.
In this study, 754 individuals were vaccinated in trial VLA1553-321, and the present analysis includes safety data up to Day 29. An independent DSMB has continuously evaluated safety data during the trial and identified no safety concerns.
Overall, the adverse event profile is consistent with the profile observed in Valneva's pivotal Phase 3 trial in adults, reported in March 2022.
Immunogenicity data for this new trial are expected in November 2023.
Juan Carlos Jaramillo, M.D., Chief Medical Officer of Valneva, commented in a press release, "These new safety data in a younger population and individuals previously infected with the chikungunya virus confirm the safety profile we previously observed in adults and the elderly."
"Chikungunya represents a major threat for people traveling to or living in areas where chikungunya virus is endemic, it is, therefore, our objective to make this vaccine available to all age groups, especially as no vaccine or specific treatments are currently available for this debilitating disease."
Funded by the Coalition for Epidemic Preparedness Innovations and conducted in collaboration with Brazil's Instituto Butantan, the VLA1553-321 adolescent trial is intended to support label extension in this age group following a potential initial regulatory approval in adults from the U.S. Food and Drug Administration. The trial is also expected to support the vaccine's licensure in Brazil, which would be the first potential approval for use in endemic populations.
The present safety analysis will also enable regulatory submission to the European Medicines Agency in 2023.
If licensed, this would be the first CHIKV vaccine candidate approved.

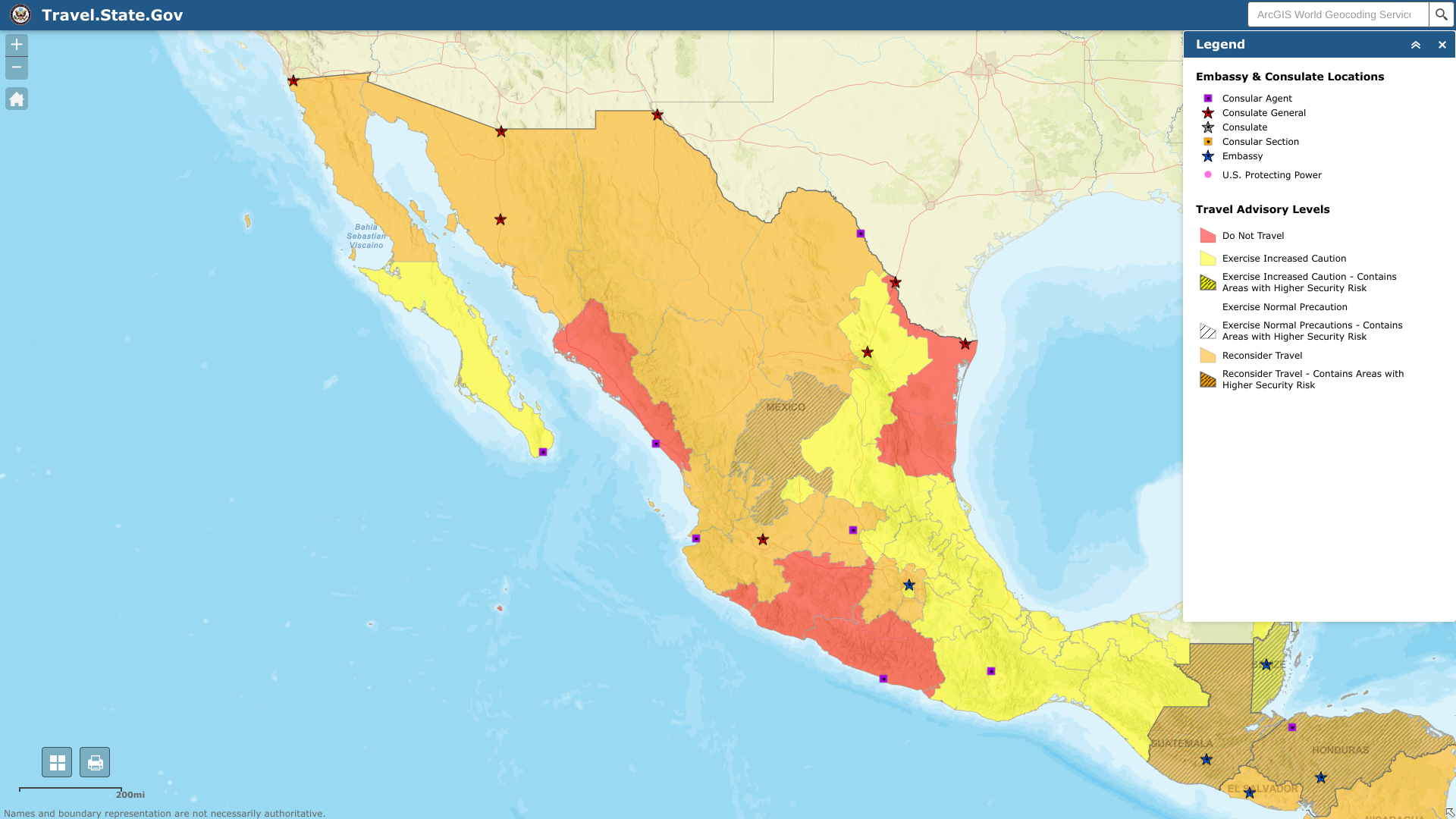
The U.S. Department of State reissued its unclassified travel advisory regarding extensive civil unrest throughout the United States of Mexico.
On August 22, 2023, the State Department reported it has limited ability to provide emergency services to U.S. citizens in many areas of Mexico, as travel by U.S. government employees to certain areas is prohibited or restricted.
In many Mexican states, local emergency services are limited outside the state capital or major cities, including Cancun.
Furthermore, U.S. government employees may not travel between cities after dark, may not hail taxis on the street, and must rely on dispatched vehicles, including app-based services like Uber and regulated taxi stands.
U.S. government employees should avoid traveling alone, especially in remote areas. U.S. government employees may not drive from the U.S.-Mexico border to or from the interior parts of Mexico, except daytime travel within Baja California and between Nogales and Hermosillo on Mexican Federal Highway 15D, and between Nuevo Laredo and Monterrey on Highway 85D.
From a health perspective, several disease outbreaks have been reported in 2023, and the U.S. CDC suggests various pre-trip vaccinations.
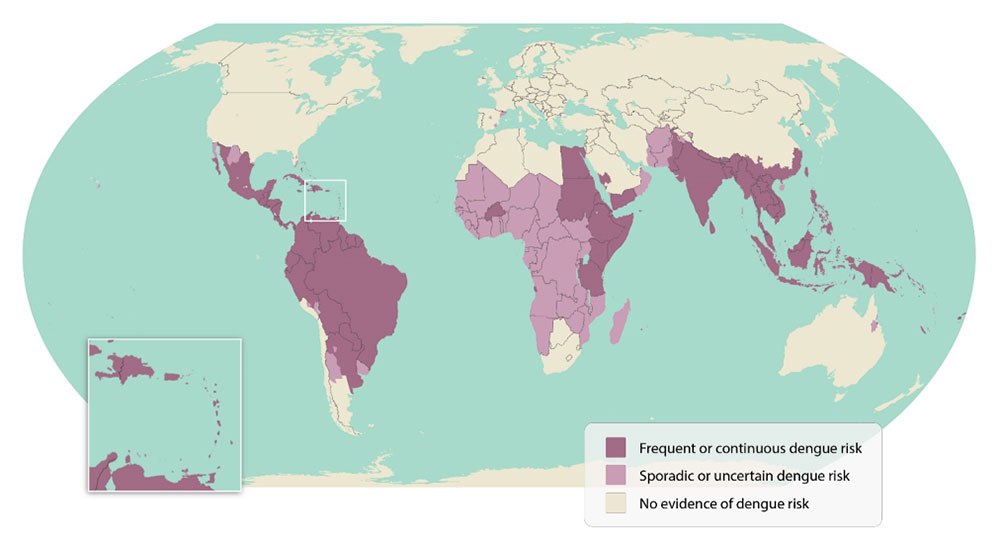
Since dengue outbreaks cause about 400 million infections yearly, the Pan American Health Organization (PAHO) and other agencies continue issuing health alerts for this deadly disease.
In the Americas, the total number of dengue cases reported was 2,997,097 through July 2023. Additionally, 1,302 deaths were reported in the Region.
To limit these deaths, two dengue vaccines have been approved and deployed in 2023.
According to a new study published in the journal Vaccines on August 22, 2023, the effectiveness of these vaccines varies.
Dengvaxia® has shown an efficacy of 60.8 % (95 % CI, 52.0–68.0) against symptomatic, virologically confirmed dengue (VCD) caused by any of dengue's four serotypes more than 28 days after the third dose in Asia (2 to 14-year children), and Latin America (9 to 16-year children) [15], respectively.
While QDENGA® has shown around 80.2 % (95 % CI, 73.3–85.3) efficacy.
And during a phase 1 clinical trial, the efficacy against VCD at 28 days after a single dose of a U.S. NIH-developed vaccine candidate was 79.6 %.
This study assessed the safety and immune response regarding nAbs induced by the SII Dengue vaccine in healthy adults in Australia.
The study showed the vaccine was safe and highly immunogenic in adults, primarily seronegative at baseline.
In the vaccine group, 59.0 % of participants showed DENV vaccine viremia post-vaccination.
This vaccine (Dengusiil) was formulated at Serum Institute of India Pvt. Ltd. (SII) to contain DENV 1, DENV 3, and DENV 4 serotypes at not less than 2.5 log10 PFUs and DENV 2 at not less than 3 log10 PFUs per single dose of 0.5 mL.
SII executive director Rajeev Dhere recently informed the TOI, "The Phase-I trial was conducted in Australia as we needed participants who had not been exposed to dengue previously."
"India has a significant proportion of people with dengue antibodies, so it was essential to test the vaccine's safety and effectiveness on individuals who were not already immune to the disease."
As of August 27, 2023, Dengvaxia is approved in the U.S., but availability requires pre-admission testing.
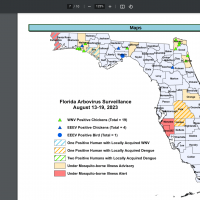
During 2023, the state of Florida and Puerto Rico have reported locally acquired and travel-related dengue cases.