Search API
According to an analysis recently published by the U.S. Centers for Disease Control and Prevention (CDC), fewer infants were admitted to hospitals struggling to breathe from Respiratory Syncytial Virus (RSV) last season.
This positive trend was announced in the MMWR (74(16);273–281) on May 8, 2025, following the approval of RSV vaccines and antibody treatments for the 2024-2025 season.
Data from the RSV-Associated Hospitalization Surveillance Network (RSV-NET) and New Vaccine Surveillance Network (NVSN) showed infants aged 0–7 months (eligible for protection with maternal vaccination or nirsevimab), 2024–25 RSV-associated hospitalization rates were lower compared with the 2018–20 pooled rates.
The most significant estimated rate reduction was observed among infants aged 0–2 months (RSV-NET: 52%, 95% CI = 49%–56%; NVSN: 45%, 95% CI = 32%–57%) and during peak hospitalization periods (December–February).
In a sensitivity analysis of the NVSN surveillance site in Houston, Texas, where the RSV season began before vaccine and antibody treatments were available, the effect was even more pronounced, with a 71% drop in infant hospitalizations.
The findings suggest the importance of protecting infants born during the RSV season through either maternal vaccination during pregnancy or nirsevimab (Beyfortus) administration in the first week of life, ideally during the hospitalization for birth.

The latest provisional data published by the UK Health Security Agency (UKHSA) show an increase in travel-associated malaria cases in England, Wales, and Northern Ireland, exceeding the levels seen in recent years.
As of June 10, 2025, the UKHSA data indicate that imported malaria cases remain at concerning levels in the UK, despite a slight decrease in diagnoses, from 2,106 in 2023 to 1,812 in 2024.
Most cases were reported during peak summer travel months between July and October.
The World Health Organization's latest malaria report estimated 263 million malaria cases and 597,000 related fatalities worldwide in 2023.
The WHO African Region bears the heaviest burden of malaria disease, with Burkina Faso, Cameroon, Ethiopia, the Democratic Republic of the Congo, Ghana, Mali, Mozambique, Niger, Nigeria, Sudan, Uganda, and Tanzania being among the countries most affected.
Dr Diana Ayoola Mabayoje, co-founder of African Diaspora Malaria Initiative (ADMI), commented in a media release, 'Our focus is on community engagement and outreach, and we will be targeting African communities in London ahead of summer travel.'
Malaria is a potentially fatal disease, but it is almost entirely preventable when antimalarial tablets are taken correctly.
Furthermore, two malaria vaccines are now offered in specific countries, such as in Africa.

In a demonstration of continental unity, twenty African Union Member States affected by cholera outbreaks in 2025 have issued a call to action.
Following the Africa Centres for Disease Control and Prevention (Africa CDC) lead, H.E. Mahmoud Ali Youssouf, Chairperson of the African Union Commission, called for bold leadership and systemic change on June 2, 2025, "The people of Africa are watching. They expect bold, coordinated, and unwavering leadership that puts their health and dignity first."
As of May 2025, the Africa CDC reported approximately 130,000 cholera cases and 2,700 deaths, representing 60% of global reported cases and an alarming 93.5% of cholera-related deaths.
The Africa CDC emphasized the systemic drivers of the crisis—limited WASH infrastructure, insecurity, weak coordination, and vaccine shortages:
"Africa needs 54 million doses of oral cholera vaccine annually, but receives barely half. This gap is unacceptable."
"Urgent action is needed to scale up local production and secure (vaccine) supply."
Globally, over 244,000 cholera cases and 2,800 related fatalities have been reported in 2025.
The U.S. Food and Drug Administration, the World Health Organization, and the U.K. National Health Service recommend oral cholera vaccines (OCVs) for travelers visiting countries experiencing outbreaks.
The WHO says that all OCVs require two vaccine doses for complete protection for up to three years.
In the United States, OCVs are expected to be available at travel clinics and pharmacies in June 2025.

Dengue fever is endemic in most countries in Southeast Asia and continues to pose a significant public health threat in tropical regions, including Malaysia, Singapore, and Thailand.
Since a recent Dengue fever outbreak began in Malaysia in late 2022, 13 states and three federal territories have been impacted.
According to the World Health Organization, Malaysia confirmed 122,423 Dengue cases and 117 related fatalities in 2024, a 17% increase from the number recorded in 2023.
Without widespread access to a Dengue prevention vaccine, the mosquito-transmitted disease continued to spread in Malaysia in 2025.
Recent data published by the Dengue For Community Portal indicate that Taman Mawar - Sendang was last week's Dengue hotspot, reporting 158 new cases.
Unlike Malaysia, Thailand recently launched a vaccination campaign to eliminate the disease burden on the population and the healthcare system. A large-scale study was launched in April 2025 in Nakhon Phanom province, involving 35,000 children.
Approved by the National Communicable Disease Committee, the clinical trial aims to assess the effectiveness of the QDENGA live-attenuated dengue vaccine, which has already received clearance from Thailand's Food and Drug Administration.
Local media reported on June 10, 2025, that Dr. Panumas Yanawetsakul, Director General of the Department of Disease Control, stated the trial would span three years, after which the vaccine could be added to Thailand's universal healthcare scheme.
If approved, Thailand would join over 30 countries that offer Takeda's QDENGA in 2025.
As of June 2025, QDENGA is not offered in the United States.

When the U.S. Centers for Disease Control and Prevention issued a Global Dengue Travel Advisory at the end of May 2025, the agency included one of the most visited destinations in the South Pacific Ocean, the Republic of Fiji.
According to a Facebook post by the Ministry of Health and Medical Services, Fiji has reported 8,708 cases of dengue in 2025.
As of May 21, 2025, 4,357 cases had been reported in the Western Division of the Fiji Islands.
Additionally, four deaths have been reported verified as attributed to dengue fever, and a red alert remains in effect for residents and visitors.
The dengue serotype causing the current outbreak in the Pacific Ocean is Dengue Virus Type 2, one of four types of virus. Severe disease caused by this mosquito-transmitted virus may be prevented by a second-generation vaccine known commercially as Qdenga.
Additionally, the CDC has included Fiji, an archipelago comprising hundreds of islands, in a Global Measles travel alert, as cases are rising in many countries worldwide.
Since approximately 12% of Fiji's visitors come from North America, the CDC recommends that international travelers consult with a travel vaccine expert about their options at least one month before traveling abroad in 2025.

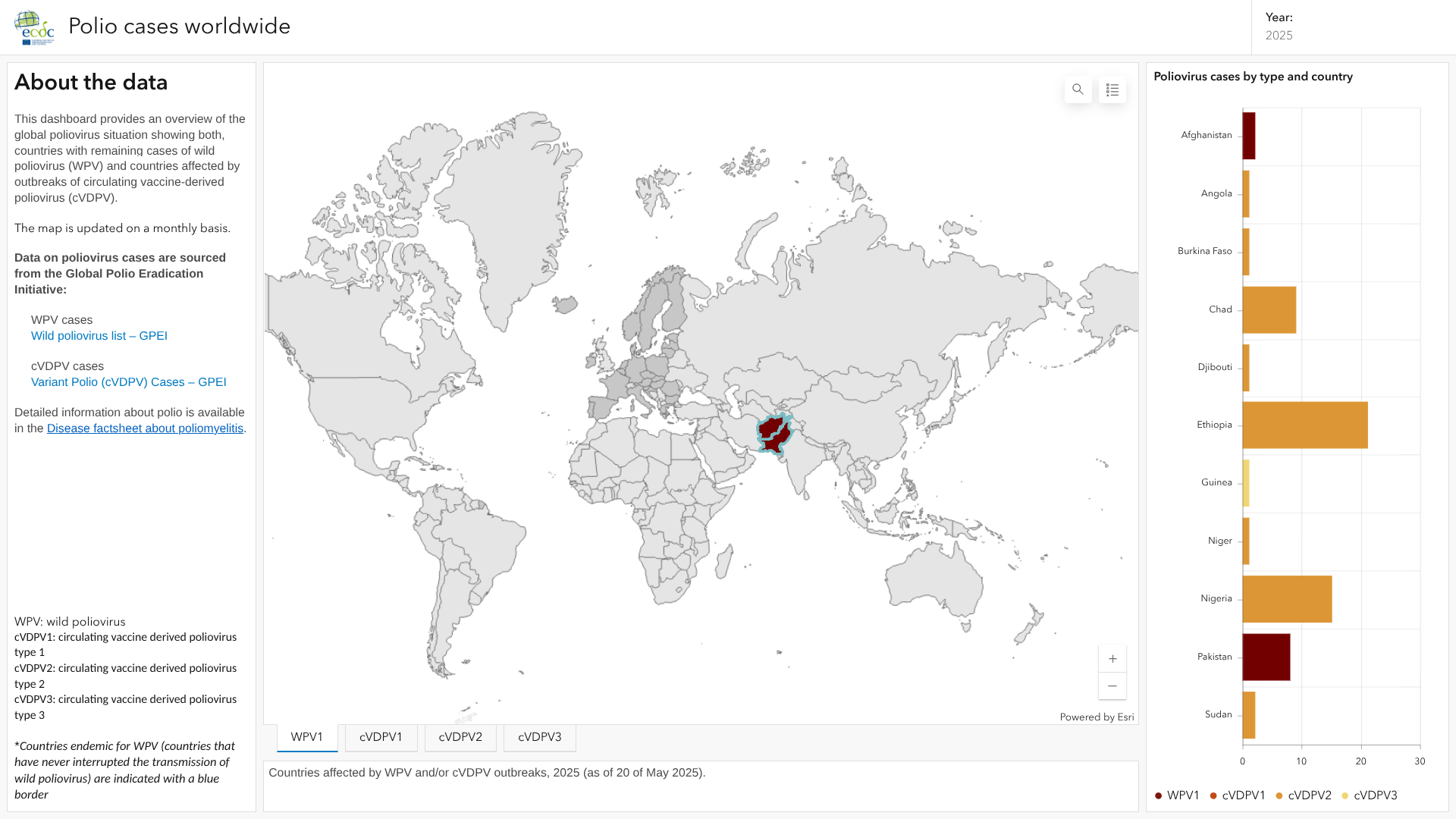
According to an update from the Global Polio Eradication Initiative (GPEI) on June 4, 2025, six countries reported additional polio cases last week.
The Islamic Republic of Pakistan, located in South Asia, reported a case of wild poliovirus type 1 (WPV1) from Gilgit-Baltistan, bringing its total number of cases to 11 in 2025.
Additionally, two WPV1-positive environmental samples were reported from Balochistan and Punjab, indicating the risk of additional cases being reported in 2025.
In 2024, 74 WPV1 cases were reported in Pakistan.
According to the European CDC, the last WPV infection in Europe was in 1998, and the World Health Organization declared the European Region polio-free in 2002.
Five other countries reported circulating vaccine-derived poliovirus type 2 (cVDPV2) cases last week: Chad, Angola, Chad, Ethiopia, Niger, and Yemen.
Additionally, Papua New Guinea reported acute flaccid paralysis cases have been reported across 11 provinces, with 20 cases testing negative for poliovirus and 28 cases still under investigation, as of June 6, 2025.
Since polio is a vaccine-preventable disease, the ECDC published a guide for children and adults that focuses on strengthening the capacities of healthcare providers to better address concerns about vaccination and tackle obstacles to vaccination uptake.
In the United States, Polio vaccination has been part of the routine childhood immunization schedule for decades. Since 2000, the Inactivated polio vaccine (IPV) has been the only polio vaccine offered. In July 2022, a case of polio caused by VDPV2 in an unvaccinated individual from Rockland County, New York.
The U.S. CDC recommends that an IPV booster dose may be advisable when visiting a poliovirus endemic area in 2025.
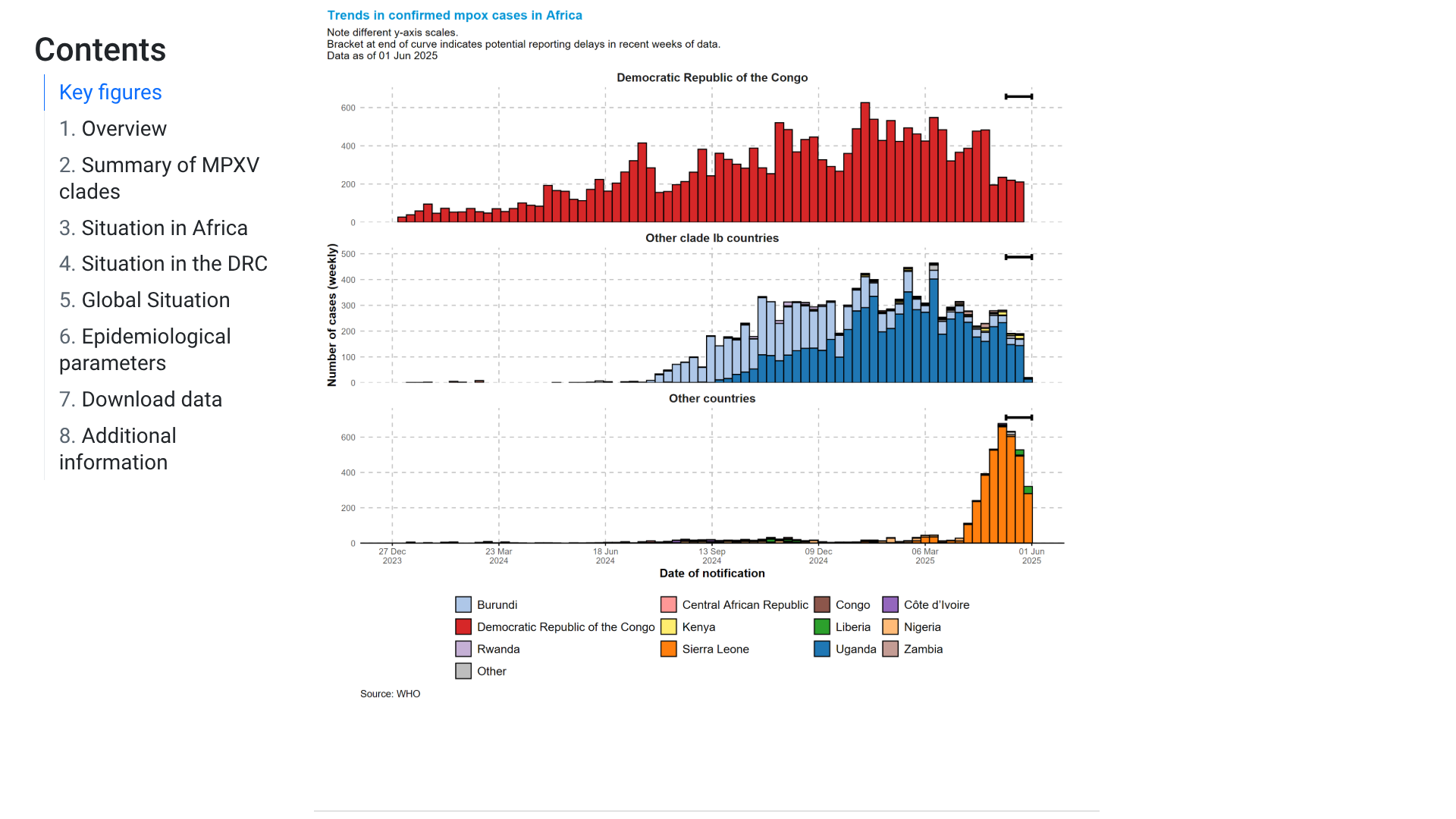
The World Health Organization (WHO) Director-General, Dr Tedros Adhanom Ghebreyesus, today announced that the mpox upsurge continues to meet the criteria of a public health emergency of international concern (PHEIC).
As of June 9, 2025, this PHEIC has been declared, based on the continuing rise in the number of cases, including a recent increase in West Africa, and likely ongoing undetected monkeypox virus (MPXV) transmission in some countries beyond the African continent.
The Director-General also concurred with and issued the Committee's revised temporary recommendations to Member States experiencing mpox outbreaks.
Regarding preventive vaccinations, the WHO advises preparing for and implementing targeted use of vaccines for "Phase 1- Stop the outbreak" through the identification of disease hotspots and targeting those groups at high risk of mpox exposure to interrupt sustained community transmission.
As of early June 2025, the U.S. CDC states that JYNNEOS is a two-dose vaccine developed to protect against mpox and smallpox. People need to receive both doses of the vaccine for optimal protection against mpox.
In the United States, JYNNEOS® is commercially offered at health clinics and pharmacies, with insurance options available.
Furthermore, to be most effective, mpox vaccination should be included as part of broader prevention activities and routine sexual health care, such as HIV or gonorrhea.
'Whether or not you've been vaccinated, continue to reduce your risk of getting mpox,' writes the CDC.