Search API
The Coalition for Epidemic Preparedness Innovations (CEPI) and Vaxxas today announced an agreement to advance the development of needle-free vaccine-patch delivery technology in a project that could end the need for frozen storage of mRNA vaccines.
CEPI confirmed on January 17, 2023, it would provide up to $4.3 million (AUD6.4 million) for preclinical testing of Vaxxas' platform, a needle-free, high-density microarray patch (HD-MAP) to assess its stability, safety, and immunogenicity.
And to evaluate its potential as a rapid-response technology for heat-stable, dried-formulation mRNA vaccines.
In addition to a Phase I clinical study of a COVID-19 vaccine candidate patch, Vaxxas is performing demonstration work in preparation for clinical evaluation under contract with the U.S. government on pandemic vaccination solutions.
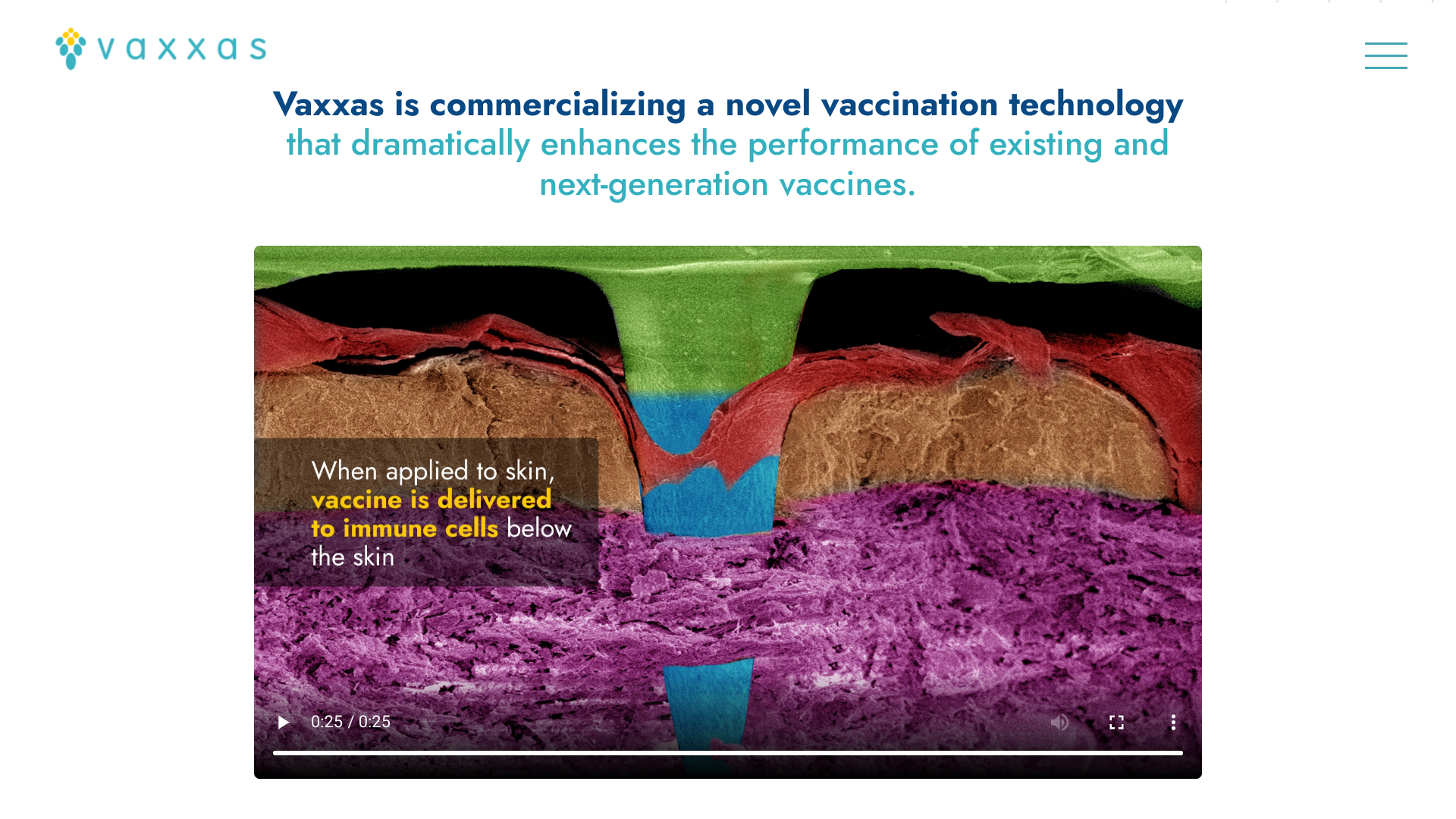
HD-MAPs comprise thousands of microscopic points attached to a small patch.
Each of these micro-projections contains a tiny dose of vaccine in a dried formulation. When applied to the skin, the patch delivers the vaccine to the abundant immune cells immediately below the skin surface.
HD-MAP vaccine delivery offers many advantages over more traditional vaccine administration methods.
For example, the dried form of the vaccine is more stable at higher temperatures than vaccines in liquid formulations.
Vaxxas' HD-MAPs have proven safe and tolerable in hundreds of trial participants and have been shown to induce equal or greater immune responses to injected vaccines at lower doses.
Compared with needle and syringe systems, they are also much easier to administer and are likely to have greater acceptability.
Ultimately, HD-MAP patches could enable a future in which vaccine patches could be mailed directly to peoples' homes, workplaces, and schools, avoiding the delay and inconvenience of traditional needle-and-syringe vaccine scheduling and administration.
David Hoey, Vaxxas's CEO, commented in a related press release, "Earning this significant funding from one of the world leaders in vaccine development is a great honor and validates the benefits offered by Vaxxas' HD-MAP vaccine platform in the fight against global epidemic and pandemic threats."
"In addition to providing an opportunity to get life-saving vaccines for infectious diseases that have a disproportionate impact on the most vulnerable populations around the world, the advanced development of HD-MAP delivery of mRNA vaccines could also prove very beneficial for the development of Vaxxas' internal pipeline across several diseases, including Covid-19."
Vaxxas' core technology was initially developed at The University of Queensland in Australia. The private company was established as a start-up in 2011.
On December 5, 2022, Vaxxas announced it completed a financing round that raised US$23 million in new funds.
CEPI is an innovative partnership between public, private, philanthropic, and civil organizations, launched in 2017, to develop vaccines against future epidemics. Its mission is to accelerate the development of vaccines and other biologic countermeasures against epidemic and pandemic threats so they can be accessible to all needy people.
Pfizer Inc. today announced that it has significantly expanded its commitment to An Accord for a Healthier World to offer the full portfolio of medicines and vaccines for which it has global rights on a not-for-profit basis to enable greater health for 1.2 billion people living in 45 lower-income countries.
To better align with disease burden and unmet patient needs in these countries, Pfizer is expanding its offering under the Accord to include off-patent products, bringing the total offering to around 500 products.
Launched in May 2022, the Accord is a transformative initiative focused on reducing health inequities.
The Accord initially included a commitment from Pfizer for access to all its patented medicines and vaccines available in the U.S. or European Union on a not-for-profit basis to 45 lower-income countries.
The Accord portfolio offering now includes chemotherapies and oral cancer treatments that can potentially treat nearly one million new cancer cases in Accord countries each year.
It also includes a wide range of antibiotics that can help to address the rising morbidity, mortality, and costs associated with antimicrobial resistance.
As Pfizer launches new medicines and vaccines, those products will also be included in the Accord portfolio on a not-for-profit basis.
“We launched the Accord to help reduce the glaring health equity gap that exists in our world. Our hope is to empower country governments and co-create solutions with them and other multi-sector partners to break down many of the system-level barriers to better health. In the months since the Accord’s launch, we have heard resoundingly from these leaders that access to a broader and more immediate scope of consistent, high-quality products is needed for meaningful and sustainable transformation. We believe this expansion of our product offering, combined with continued efforts to help address the barriers that limit or prevent access, will help us to achieve and even expedite our vision of a world where all people have access to the medicines and vaccines they need to live longer and healthier lives,” said Pfizer Chairman and CEO Albert Bourla, in a press release on January 17, 2023.
Alongside governments and multi-sector partners, Pfizer is working to co-create scalable solutions that help address systemic barriers to better health focused on finding faster, more efficient pathways for the supply of medicines and vaccines as well as strengthening the resources, capabilities, and platforms that can enable quick and more sustainable access to those medicines.
This includes technical expertise, training, diagnostic capacity, innovative financing, and more.
“The Accord is an important step toward sustainable health security for Rwanda and the broader continent. The expanded portfolio offering and public health system strengthening efforts will further enhance our progress and offer valuable support to key national health initiatives that lead to positive health outcomes,” commented His Excellency Paul Kagame, President of Rwanda.
Further details about An Accord for a Healthier World are available at Pfizer.com/Accord.

The U.S. Embassy in Peru recently confirmed the Government extended a 30-day State of Emergency in selected areas of Peru starting January 15, 2023. The affected areas include the departments of Cusco, Puno, Lima, and the province of Callao.
The province of Andahuaylas in the department of Apurimac, the provinces of Tambopata and Tahuamanu in the department of Madre de Dios, the district of Torata, and the province of Mariscal Nieto in the department of Moquegua are included.
Some national highways are affected, including the Pan-American Highway as well as the Apurimac-Cusco-Arequipa roadway.
For additional details, please see the complete Supreme Decree.
The Embassy suggests U.S. citizens in Peru avoid crowds and demonstrations, comply with instructions from local authorities, and enroll in STEP to receive alerts and messaging from the U.S. Embassy in Lima.
And for in-country assistance, visit the U.S. Embassy in Lima, Peru, at Avenida La Encalada cdra, 17 s/n, Santiago de Surco 15023, Lima , or contact +51-1-618-2000 and [email protected].
From a health perspective, the U.S. CDC says to check the vaccines and medicines list and visit your healthcare provider at least a month before your trip to Peru to get vaccines or medicines you may need.
The U. S. Centers for Disease Control and Prevention (CDC) recently announced it joined the Republic of Uganda in marking the end of the fifth Sudan Ebolavirus outbreak in Uganda.
The last Sudan Ebola outbreak in Uganda was in 2012.
In addition, entry screening and public health monitoring of travelers to the U.S. who have been in Uganda in the last 21 days ended on January 11, 2023.
“I commend the Government of Uganda, local health workers, and global public health partners who worked to end the country’s Ebola outbreak,” said CDC Director Rochelle P. Walensky, M.D., M.P.H., in a media statement.
The CDC confirmed it would continue supporting the Ugandan Ministry of Health in continuing surveillance, infection prevention and control, and response activities to help ensure rapid detection and response to future cases and outbreaks.
Since this outbreak declaration in September 2022, there were 164 cases with a case-fatality ratio was 47%.
Furthermore, three vaccine candidates launched human clinical trials for this type of Ebola in December 2022.

Global interest in developing vaccines against Neisseria gonorrhoeae (NG) has been sparked by the increasing threat of gonococcal antimicrobial resistance and the number of new infections.
And according to the U.S. Centers for Disease Control and Prevention (CDC), over 600,000 cases of gonorrhea were reported in 2020, making it the second most common sexually transmitted infection in the United States.
And the disease is known to be contracted repeatedly without apparently developing protective immunity.
To address this extensive health need, Intravacc today announced favorable preclinical data for Avacc 11®, the prophylactic intranasal gonorrhea candidate vaccine developed in partnership with Therapyx inc.
The results of the candidate, a proprietary outer membrane vesicle (OMV) platform-based gonorrhea vaccine combined with encapsulated IL-12, showed protection against subsequent infection with NG.
In this study, mice were vaccinated via the intranasal route, and the results of this intranasal study were similar to the intravaginal vaccination route. Intranasal immunization resulted in high serum IgG, salivary IgA, and vaginal IgG and IgA anti-gonococcal antibodies when OMVs were administered with IL-12 ms.
The serum IgG and salivary IgA antibodies induced in male mice were similar to the response induced in female mice.
Gamma interferon (IFN-g) production by CD4 T cells from iliac lymph nodes was elevated after vaccination intranasally or intravaginally.
Female mice immunized with OMVs plus IL-12 ms by either route resisted challenge with NG to an equal extent, and resistance generated by intranasal immunization extended to heterologous strains of NG.
These results were published in the peer-reviewed journal MSphere of the American Society of Microbiology.
Dr. Jan Groen, Intravacc's CEO, commented in a press release on January 16, 2023, "Together with our partner Therapyx, we are very pleased with the preclinical data of the intranasal candidate vaccine Avacc 11®."
"This intranasal gonococcal vaccine is more suitable to fight gonorrhea infections, which are becoming increasingly resistant to antibiotic treatments."
In October 2022, Intravacc was awarded a $14.6 Million U.S. NIH/NIAID contract to develop this intranasal candidate gonorrhea vaccine further.
For the development of vaccines, Intravacc has designed and developed a platform based on outer membrane vesicles, spherical particles with intrinsic immune-stimulating properties.
The OMVs can be designed with immunogenic peptides and/or proteins that stimulate effective adaptive immunity.
The OMV carrier has been optimized to induce a more effective immune response against these newly introduced antigens.
Intravacc has also developed genetic tools to increase the yield of the OMVs, reduce toxicity and achieve the desired antigenic composition.
Intravacc's OMV platform is scalable and allows rapid and efficient modification of the antigen composition, either through genetic modification of the bacterial host or by associating antigens with stored OMVs.
As of January 2023, there is no effective gonorrhea vaccine available in the U.S.

Since influenza is an airborne virus, efforts to avoid catching the flu depend on your proximity to an infectious (sneezing) person. While this logic is simple to say, what reports can a person trust to make virus avoidance decisions in 2023?
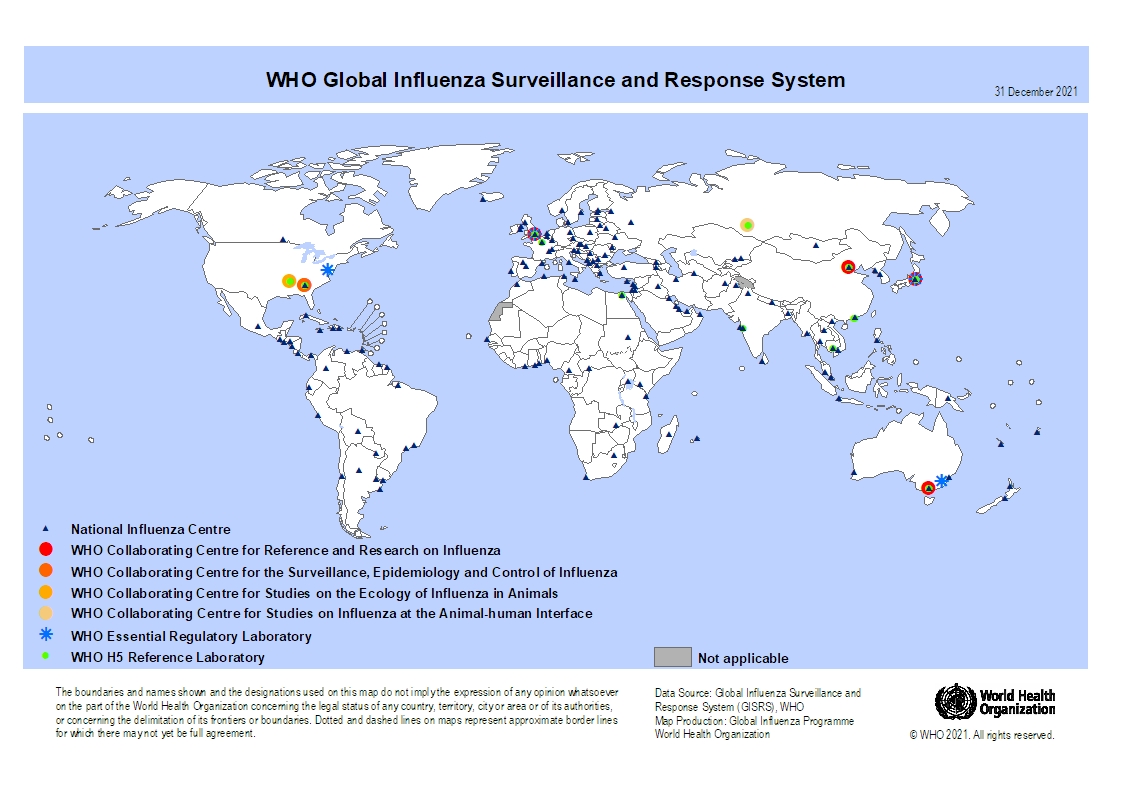
According to the World Health Organization (WHO) Influenza Update N° 436, most countries of North America indicate influenza activity decreased while others were stable or continued to increase.
The WHO finding was supported by the recent U.S. Centers for Disease Control and Prevention (CDC) report issued on January 13, 2023, that stated seasonal influenza activity continues but is declining in most areas, with specimens testing positive for influenza in clinical laboratories decreasing in all regions.
Elsewhere around the globe, the WHO reported:
In Europe, overall influenza activity continued to increase, with influenza positivity from sentinel sites remaining above the epidemic threshold at the regional level.
In central Asia, influenza activity increased with influenza A(H1N1)pdm09 viruses predominant, followed by influenza B viruses.
In Northern Africa, influenza detections increased among reporting countries with all seasonal subtypes detected. And influenza activity remained low in tropical Africa, with detections of all seasonal influenza subtypes reported. However, activity increased in some countries in Eastern Africa.
In Western Asia, influenza activity decreased overall with all seasonal influenza subtypes detected, though increased activity was reported in some countries.
In East Asia, influenza activity of predominantly influenza A(H3N2) viruses remained low overall among reporting countries but with increases reported in Mongolia and the Republic of Korea.
In the Caribbean and Central American countries, influenza activity of predominantly influenza A(H3N2) viruses decreased overall but remained elevated in Mexico.
In the tropical countries of South America, influenza detections were generally low. However, influenza positivity was at a moderate level in Ecuador.
In the temperate zones of the southern hemisphere, influenza activity decreased in Argentina and Chile to low levels and remained low elsewhere.
In Southern Asia, influenza activity remained low, mainly due to decreased activity reported in the Islamic Republic of Iran.
In South-East Asia, detections of predominantly influenza B remained elevated due to continued detections reported in Malaysia.
Before visiting these areas, the CDC suggests speaking with a doctor, nurse, or pharmacist to ensure you are appropriately protected from influenza.
In the U.S., access to flu shots remains abundant, as over 157 million vaccines have already been distributed and are generally available at most clinics and pharmacies.
As the new year began a few weeks ago, various scientists are focused on developing Human Immunodeficiency Virus (HIV) vaccines. And vaccine development has accelerated in 2023 with candidates utilizing innovative technologies such as mRNA.
Vaccines work by inducing the immune system to make antibodies that can neutralize a particular pathogen.
But doing so for HIV has been challenging because there are countless variants worldwide, wrote the U.S. National Institutes of Health (NIH) on December 13, 2022.
This challenge is why mRNA vaccines may become the solution.
Encouraging news was announced in 2022 when Moderna Inc., a global leader in mRNA vaccines, confirmed it was participating in the NIH's HVTN 302 study that examines the safety and immune responses of BG505 MD39.3 mRNA, BG505 MD39.3 gp151 mRNA, and BG505 MD39.3 gp151 CD4KO vaccines.
Each of Moderna's vaccine candidates are designed to present the spike protein found on the surface of HIV that facilitates entry into human cells and encodes for different but highly related stabilized proteins.
While this early-stage, Phase 1 clinical trial was updated on October 3, 2022, it could be years from achieving U.S. Food and Drug Administration (FDA) approval.
As of January 15, 2023, the FDA had not approved any HIV prevention vaccine for use by people.
Unfortunately, the AIDS epidemic continues to impact people everywhere, specifically in Africa.
About 38 million people worldwide are living with HIV, and about 70% of them live in Africa.
However, over 28 million people were accessing antiretroviral therapy in 2022, a significant increase from 7.8 million in 2010.
And in 2023, these people have expanded treatment options.
Gilead Sciences, Inc. announced on December 22, 2022, that the FDA approved Sunlenca® for treating HIV-1 infections in heavily treatment-experienced adults with multi-drug resistant HIV-1 infection.
And previously, the U.S. FDA-approved Apretude for use by at-risk adults and adolescents weighing at least 35 kilograms for pre-exposure prophylaxis to reduce the risk of sexually acquired HIV on December 20, 2021.
Anthony S. Fauci, M.D., former director at the U.S. National Institute of Allergy and Infectious Diseases, recently commented about HIV vaccine development efforts in an Emerging and Reemerging Infectious Diseases Perspective: It Ain't Over Till It's Over…but It's Never Over.