Search API
During the year since Malawi confirmed its first case of wild poliovirus patient in 30 years, more than 33 million children across five African countries have been vaccinated against the virus.
About 80 million polio vaccine doses have been administered since mid-2022.
A total of nine wild poliovirus cases have been reported, with one in Malawi and eight in neighboring Mozambique, since the declaration of an outbreak in February 2022 in Malawi.
The last confirmed case to date was in August 2022 in Mozambique.
The wild poliovirus in Malawi and Mozambique originated from Pakistan, one of the two last endemic countries.
"Southern Africa countries have made huge efforts to bolster polio detection, curb the spread of the virus and ensure that children live without the risk of infection and lifelong paralysis," said Dr. Matshidiso Moeti, World Health Organization (WHO) Regional Director for Africa, in a press release on February 17, 2023.
"We continue to support the polio control efforts across the region so that every child receives the protection they need."
And at least five more are planned for 2023 in the five countries.
Polio is highly infectious and affects unimmunized or under-immunized children.
There is no cure for polio, and it can only be prevented by immunization.
Children worldwide remain at risk of wild polio type 1 as long as the virus is not eradicated in the remaining areas where it is still circulating.
There are WHO-approved polio vaccines offered throughout Africa.
These African countries were identified in the U.S. CDC's Global Polio Alert - Level 2, Practice Enhanced Precautions alert issued in January 2023.

The peer-review JAMA Network published an Original Investigation focused on the most appropriate monoclonal antibody (mAb) for preventing respiratory syncytial virus (RSV) in children.
This analysis was essential since RSV is a leading cause of respiratory disease in children and the primary cause of hospitalization for viral respiratory infections.
Published on February 17, 2023, this systematic review and network meta-analysis comparing the efficacy and safety of 4 mAbs for preventing RSV infection in children found associations with the improvement of clinically meaningful outcomes and no significant associations with RSV-related adverse effects and mortality.
These findings suggest that motavizumab, nirsevimab, and palivizumab are associated with reduced rates of RSV infections and hospitalizations.
Similar results were observed in the rate of supplemental oxygen use.
While RSV vaccine candidates are completing late-stage clinical studies, mAbs targeting RSV prevention have been U.S. Food and Drug Administration (FDA) for years.
For example, the U.S. CDC CDCHAN-00479 stated on November 4, 2022, that eligible high-risk children should receive the mAbs palivizumab (Synagis) treatment according to AAP guidelines to prevent RSV-associated hospitalizations.
Recently, the Biologics License Application for nirsevimab (Beyfortus®) was accepted for review by the FDA on January 5, 2023. The FDA's Prescription Drug User Fee Act date for its decision is in the third quarter of 2023.
If approved, nirsevimab would become available in the U.S. for the 2023/2024 RSV season.
Nirsevimab (MEDI8897) was developed in partnership between AstraZeneca and Sanofi.

Although the initial response was led by a sense of urgency and crisis management, the Florida State Surgeon General announced on February 15, 2023, he believes it is critical that, as public health professionals, responses are adapted to the present to chart a future guided by data.
As such, the State Surgeon General notified the healthcare sector and public on February 15, 2023, of a substantial increase in Vaccine Adverse Event Reporting System reports issued from Florida after the COVID-19 vaccine rollout.
To further evaluate this, Surgeon General Dr. Ladap wrote a letter to the U.S. Food and Drug Administration and the Centers for Disease Control and Prevention, illustrating the risk factors associated with the mRNA COVID-19 vaccines and emphasizing the need for additional transparency.

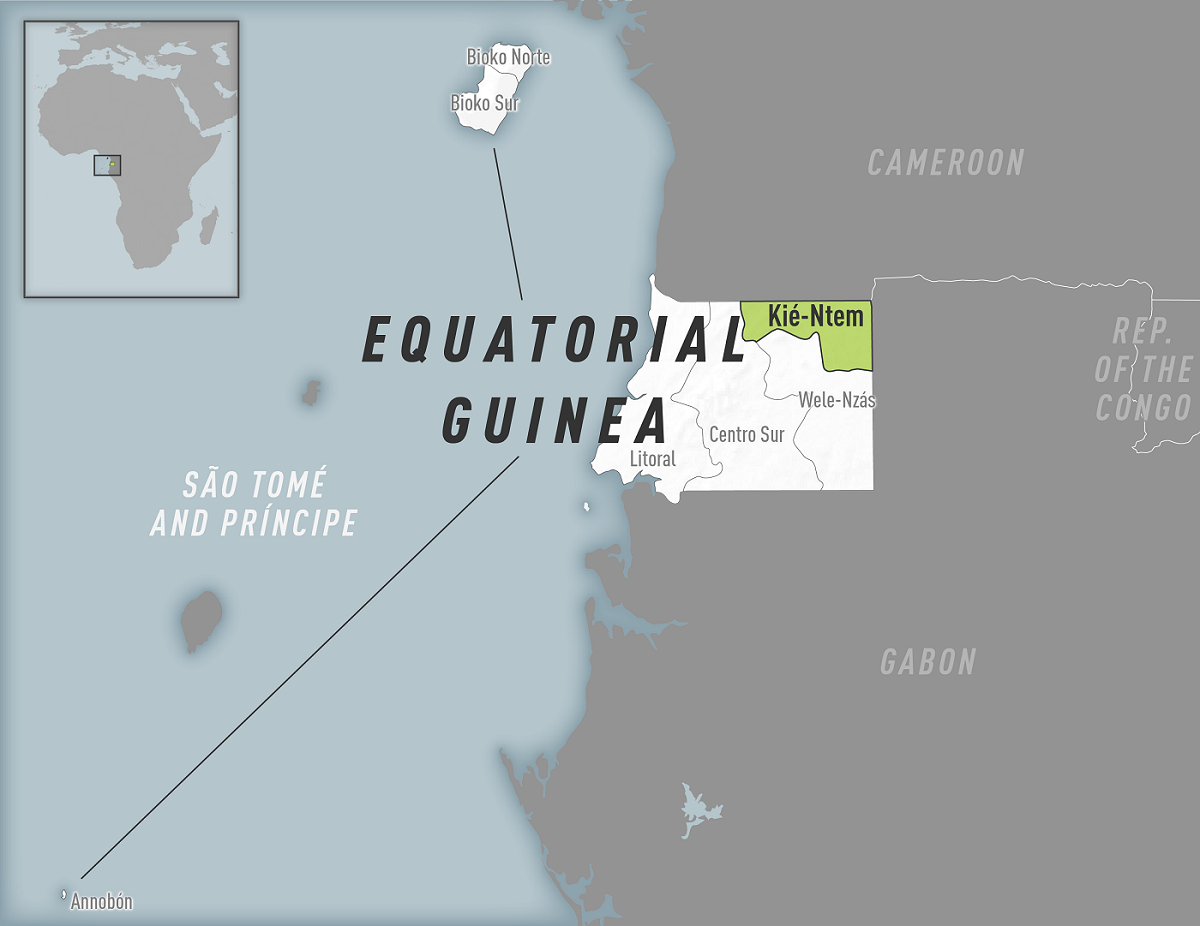
The U.S. Centers for Disease Control and Prevention (CDC) today announced Equatorial Guinea declared an outbreak of Marburg virus disease (MVD) after confirming cases in the Kie Ntem Province.
In response, Equatorial Guinea declared an outbreak of MVD on February 13, 2023.
The CDC stated on February 16, 2023, that MVD is a rare and deadly disease that has sometimes caused outbreaks in several African countries.
Travelers to MVD endemic areas should separate themselves from others and seek medical care immediately if they develop fever, chills, muscle pain, rash, sore throat, diarrhea, weakness, vomiting, stomach pain, or unexplained bleeding or bruising during or after travel (up to 21 days).
And, call ahead before going to a healthcare facility and tell your doctor that you've been to an area reporting MVD cases, says the CDC.
Initially detected in 1967 in Germany, MVD is spread by contact with the blood or body fluids of a person infected with the Marburg virus.
As of February 17, 2023, Angola, DR Congo, Equatorial Guinea, Cameroon, Germany, Ghana, Guinea, Kenya, Serbia, South Africa, and Uganda have previously confirmed MVD cases.
While there are no U.S. FDA-approved Marburg preventive vaccines, several candidates are conducting clinical studies.
NPR reported today Dr. Leana Wen, an emergency physician and professor at the Milken School of Public Health at George Washington University, says there's one more urgent reason a vaccine must be prioritized.
"Healthcare workers are at particular risk, and in Equatorial Guinea and surrounding countries, illness and death of the relatively few doctors and nurses they have would have a tremendous lasting impact on health in their region."
The acting director of the Africa Centers for Disease Control and Prevention (ACDC), Ahmed Ogwell, stated he hopes Mpox vaccines will finally arrive on the continent "in another two weeks, tops" after months of seeking doses.
The AP reported on February 16, 2023, the Mpox vaccines will go first to countries with acute need and the most significant overall burden, such as Congo and Nigeria. Nigeria was listed in the U.S. CDC's Watch - Level 1, Practice Usual Precautions notice issued in November 2021.
People become infected with the mpox virus through contact with the bodily fluids of infected animals or humans.
Mpox occurs throughout Central and West Africa, often near tropical rainforests. The first human case of Mpox was identified in the Democratic Republic of Congo in 1970.
In November 2022, the government of the Republic of Korea, through the Korean Disease Control and Prevention Agency, announced it would donate the first batch of the Mpox vaccine to Africa through the ACDC.
In the U.S., over 1.1 million doses of the Bavarian Nordic JYNNEOS® (MVA-BN) vaccine have been distributed since May 2022.
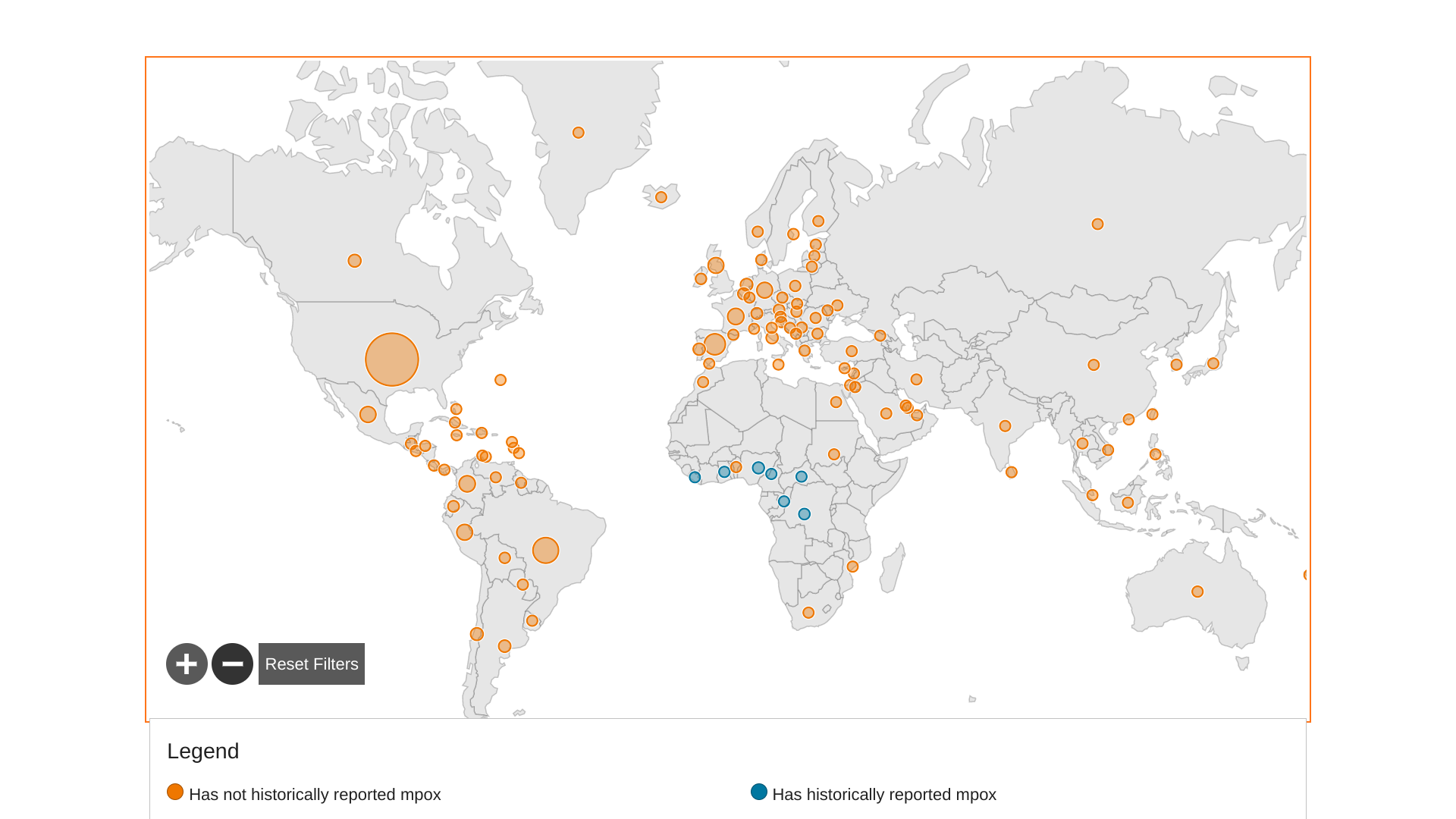
Since May 5, 2022, about 110 countries have reported 85,800 Mpox patients and 88 related fatalities.
The countries with the highest cumulative notification rates in Europe are Spain, Portugal, and Luxembourg. Additional Mpox outbreak news is posted at MpoxToday.
While the World Health Organization has maintained its multi-country outbreak notice, the U.S. recently discontinued its Mpox alert.
The continual spreading of the Highly Pathogenic Avian Influenza (HAPI) H5N1 influenza A virus across the globe has prompted many questions without clear answers.
These questions accelerated recently when seven people, including various mammals, became infected over the past year.
Amesh Adalja, M.D. with MedPageToday, published an opinion article on February 16, 2023, focused on whether an influenza pandemic caused by this version of HAP is imminent.
'This is an important question that must be asked of all novel influenza viruses. However, the answers regarding influenza are not that simple and require placing this strain into a larger context of avian influenza more generally,' wrote Dr. Adalja. The full unedited article is posted at this link.
Avian influenza A (bird flu) viruses are common and widespread in birds.
Should a human-to-human HAPI outbreak occur, the U.S. Food and Drug Administration has already approved one type of 'bird flu vaccine' that could be effective.
While most people have already received their annual flu shot, this type of vaccine would not be very effective against HAPI viruses.
The World Health Organization (WHO) Influenza Update N° 438 recently reported influenza activity decreased globally.
In the countries of North America, most indicators of influenza activity fell to levels similar to or below levels typically observed this time of year.
As of February 9, 2023, Influenza A(H3N2) was the predominant virus detected in the U.S., whereas A(H1N1)pdm09 and A(H3N2) were co-circulated in Canada in the most recent reporting week.
In Europe, overall influenza activity continued to decrease, but influenza positivity from sentinel sites remained above the epidemic threshold at the regional level.
Separately, a leading mRNA-based flu shot candidate for adults announced encouraging interim results from its pivotal Phase 3 safety and immunogenicity trial of mRNA-1010 (P301).
As of February 4, 2023, the U.S. CDC confirmed about 172.76 million influenza vaccines had been distributed for the 2022-2023 flu season in the U.S.

Moderna Inc. today announced one of five influenza vaccine candidates in its portfolio published interim results from its pivotal Phase 3 safety and immunogenicity trial.
mRNA-1010 (P301) is an mRNA-based seasonal influenza (flu) vaccine candidate for adults.
mRNA-1010 encodes for hemagglutinin (HA) glycoproteins of the four influenza strains recommended by the World Health Organization (WHO) to prevent influenza.
Interim results indicate that mRNA-1010 achieved superiority on seroconversion rates for A/H3N2 and A/H1N1 and superiority on geometric mean titer ratios for A/H3N2 and non-inferiority on geometric mean titer ratios for A/H1N1.
Non-inferiority was not met for either endpoint for the influenza B/Victoria- and B/Yamagata-lineage strains.
"Today's results represent an important step forward in developing mRNA-based influenza vaccines to address the substantial burden of disease caused by influenza."
"We are encouraged by the safety and tolerability profile and the strong immunogenicity results against Influenza A viruses which cause the overwhelming majority of flu-related disease in older adults."
"We now look forward to the efficacy results from the ongoing pivotal Phase 3 efficacy study being conducted in parallel," said Stephen Hoge, M.D., Moderna's President, in a press release on February 16, 2023.
"While we did not achieve non-inferiority for the Influenza B strains, which are more frequent in younger populations, we have already updated the vaccine that we believe could improve immune responses against Influenza B and will seek to quickly confirm those improvements in an upcoming clinical study thanks to the agility of our mRNA platform."
The first per-protocol interim analysis of efficacy is now expected to be reviewed by an independent Data and Safety Monitoring Board (DSMB) before the end of the first quarter of 2023.
Based on these results, the DSMB will notify the Company whether the primary efficacy endpoint has been met or whether the study should continue accruing further cases toward the final analysis.
Moderna is advancing a portfolio of five influenza vaccine candidates that include additional HA antigens for broader coverage of circulating influenza A strains (mRNA-1011 and mRNA-1012) and candidates that incorporate both HA and neuraminidase (NA) antigens to target multiple proteins involved in the influenza virus lifecycle to reduce the potential of viral antigenic escape (mRNA-1020 and mRNA-1030).
Moderna is also developing combination vaccine candidates, including vaccine candidates against influenza and SARS-CoV-2, influenza and RSV, and influenza, SARS-CoV-2, and RSV.
The goal of Moderna's combination vaccine candidates is to protect against multiple respiratory pathogens in a single vaccine.
Moderna's unedited press release is available at this link.
Other flu shot news is posted at PrecisionVaccinations.