Search API
LinKinVax today announced the interim results of a double-blind, Phase I clinical trial evaluating CD40.HIVRI.Env, a preventive HIV vaccine candidate.
The ANRS VRI06 study's initial results, published on February 22, 2023, show that the vaccine candidate was safe and induced an early, significant, and sustained immune response.
The vaccine yielded high levels of antibodies directed against the HIV envelope:
- 80-100% at Week 6,
- 100% at Week 26 in all groups (0.3, 1, and 3 mg).
- These levels remained stable or dropped slightly until Week 48.
- Antibodies targeting a specific area of the HIV envelope (V1/V2 region) were also produced.
Furthermore, at Week 26, neutralizing antibodies were detected in 50% of subjects in the 0.3 mg group and 100% of subjects in the other two groups.
The observed counts of CD4+ T cells targeting specifically the HIV envelope protein produced after the vaccination remained stable until Week 48.
Prof. Yves Lévy, Executive Director of the Vaccine Research Institute and CMSO of LinKinVax, commented in a press release, "The immune response profile generated by this vaccine (antibody response and activation of polyfunctional CD4+ T cells) was the one associated with a reduced risk of HIV infection in the RV144 trial."
"However.... the efficacy of this vaccine will only be evaluated in the Phase II/III studies."
LinKinVax's vaccine platform is built around a humanized monoclonal antibody, which is merged with regions of pathogens of interest, targeting the CD40 molecule expressed by dendritic cells, DC, which play a key role in stimulating the immune system.
André-Jacques Auberton-Hervé, Co-founder and CEO of LinKinVax, added, "We are pleased with these very promising immunogenicity results, which demonstrate the robustness of our DC Targeting vaccine platform and confirm its safety."
"This important milestone paves the way for the upcoming Phase II/III clinical studies that we will conduct once the final Phase I results has been obtained."
"These studies will aim to demonstrate the efficacy of the vaccine, the "Everest of vaccine strategies," which has been the elusive goal of HIV research for the past 40 years."
Other HIV vaccine development news is posted at Precision Vaccinations.

Icosavax, Inc. today announced the U.S. Food and Drug Administration (FDA) has granted Fast Track designation for IVX-A12, a bivalent respiratory syncytial virus (RSV) and human metapneumovirus (hMPV) virus-like particle (VLP) vaccine candidate, in older adults 60 years of age and above.
Niranjan Kanesa-thasan, Chief Medical Officer of Icosavax, commented in a press release on February 21, 2023, "IVX-A12 is differentiated as the most advanced vaccine candidate against these two leading causes of pneumonia in older adults."
In October 2022, Icosavax initiated a Phase 1, randomized, observer-blinded, placebo-controlled, multicenter study of IVX-A12, with and without CSL Seqirus' proprietary adjuvant MF59®, in up to 120 healthy older adults aged 60 to 75 years.
Icosavax anticipates announcing topline interim results from this Phase 1 trial in mid-2023, with subjects after that followed through 12 months after vaccination.
The company plans to initiate a Phase 2 trial for IVX-A12 in the second half of 2023.
According to the FDA, Fast Track is a process designed to facilitate the development and expedite the review of investigational drugs to treat serious conditions and fulfill an unmet medical need. An investigational drug that receives Fast Track designation may be eligible for more frequent interactions with the FDA to discuss the candidate's development plan and, if relevant criteria are met, eligibility for Accelerated Approval and Priority Review.
As of February 21, 2023, the FDA has not approved any RSV vaccine candidates conducting late-stage studies. Furthermore, the FDA has approved and is reviewing RSV monoclonal antibody therapies.

Vaxcyte, Inc. announced today that the U.S. Food and Drug Administration (FDA) had cleared the Company's infant Investigational New Drug (IND) application for VAX-24, its lead, 24-valent pneumococcal conjugate vaccine (PCV) candidate designed to prevent invasive pneumococcal disease (IPD).
Vaxcyte stated it plans to initiate the infant Phase 2 study in the second quarter of 2023, with data expected by 2025.
The infant study design will include a primary immunization series consisting of three doses followed by a subsequent booster dose.
"The clearance of the VAX-24 infant IND application marks an important step in expanding the development of our lead, broad-spectrum PCV candidate in this important and vulnerable population," said Grant Pickering, Chief Executive Officer and Co-founder of Vaxcyte, in a press release on February 21, 2023.
"Based on the positive data from our Phase 1/2 proof-of-concept study in adults, we believe VAX-24 has the potential to deliver a best-in-class profile with broader coverage and better immune responses relative to the standard-of-care for both the adult and pediatric populations."
"We remain focused on advancing our VAX-24 clinical programs and look forward to the anticipated initiation of the infant study and announcement of the topline results from our Phase 2 study in adults aged 65 and older in the second quarter of this year."
Despite the effectiveness of current vaccines, IPD, which includes meningitis and bacteremia, remains persistent in the first years of life and is a leading cause of invasive disease in children two years of age and under.
The burden of disease in the pediatric population underscores the need for a broader-spectrum vaccine.
Precision Vaccinations post additional pneumococcal disease vaccine and candidates news.

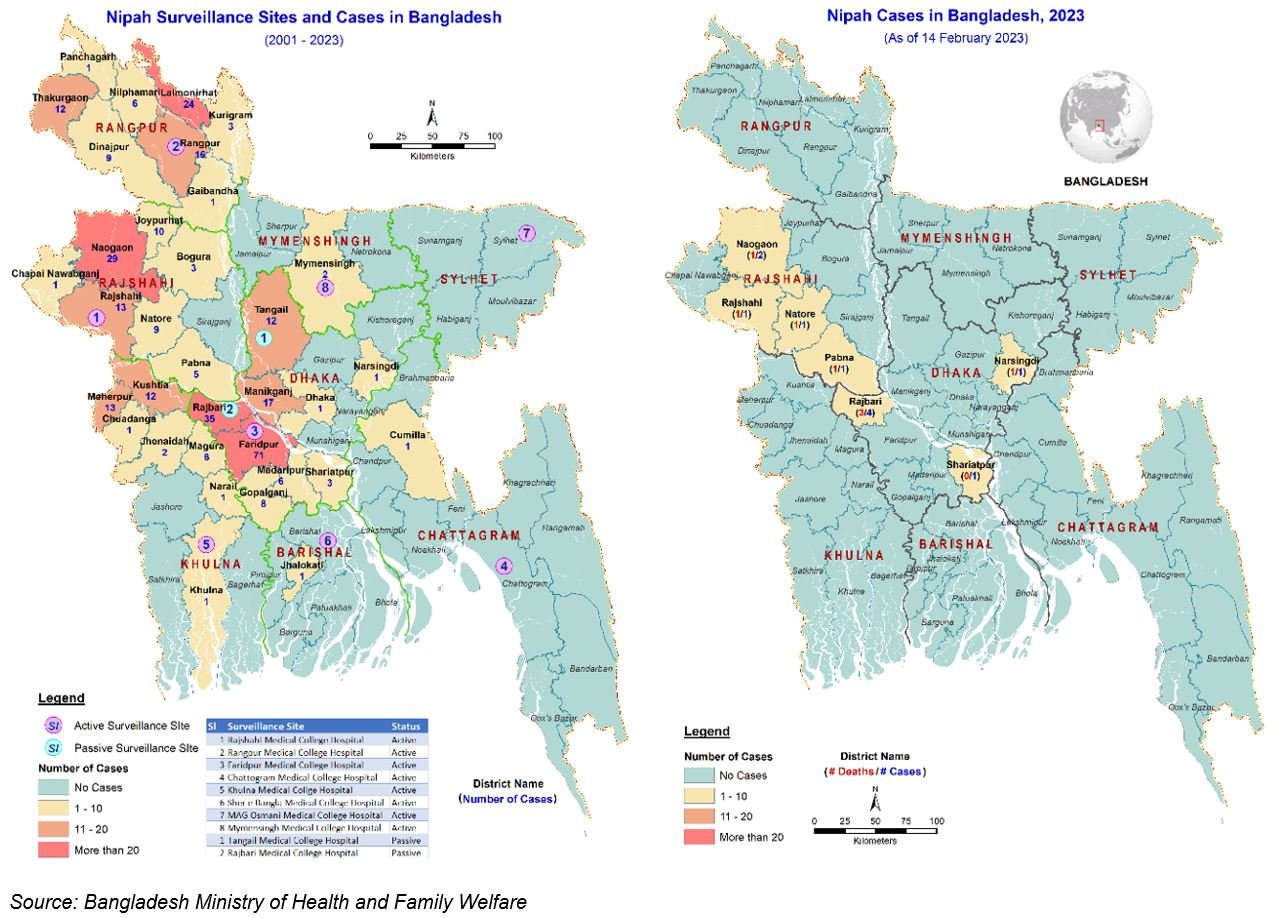
The World Health Organization (WHO) reported last week Nipah virus outbreaks have been seasonal in Bangladesh, with cases usually occurring annually between December and May.
Nipah virus is a paramyxovirus whose wildlife reservoir is the fruit bat.
However, since early January 2023 and as of February 13, 2023, the WHO reported 11 Nipah cases had been confirmed, including eight deaths (Case Fatality Rate (CFR) 73%) reported across two divisions in Bangladesh.
A multisectoral response has been implemented by the Ministry of Health and Family Welfare, Bangladesh, including strengthened surveillance activities, case management, infection prevention and control, and implementation of risk communication campaigns.
As of February 20, 2023, the WHO assesses the risk as high at the national level, moderate at the regional level, and low at the global level.
The U.S. CDC previously published a Dengue travel alert for Bangladesh, but not for this Nipha virus outbreak.
No Nipah Virus vaccines are approved, but candidate vaccines are conducting studies, such as PHV02.
Pakistan's Polio Eradication Programme recently stated it is focused on eliminating polio in 2023 by continuing to conduct extensive vaccination programs.
Pakistan launched its first nationwide anti-polio campaign in 2023 to immunize children under the age of five against the crippling disease.
Pakistan has repeatedly come close to eradicating polio, but long-running propaganda in conservative rural areas that the vaccines cause sterility in children has set back the mission, reported VOA in January 2023.
This goal is within reach as only 20 wild poliovirus type 1 cases were reported in the Khyber Pakhtunkhwa province in 2022.
Polio is a crippling and potentially deadly disease that affects the nervous system of children.
For a summary of polio-related activities in Pakistan for the last two weeks, take a look at the latest National Emergency Operations Centre newsletter.
Pakistan is included in the U.S. CDC's Alert - Level 2, Practice Enhanced Precautions polio travel advisory issued in 2023.

The U.K. Health Security Agency (UKHSA) recently reported surveillance data shows laboratory reports of norovirus are 66% higher than the average at this time of year.
As of February 9, 2023, the most significant increase in norovirus has been seen in those aged 65 years and over.
While high numbers of cases in this age group are expected at this time of year, these levels haven't been seen in over a decade, says the UKHSA.
Norovirus is highly infectious and quickly spread through contact with someone with the infection or contaminated surfaces.
In response to the increase in cases, UKHSA is reminding the public of the actions they can take to reduce the spread of norovirus.
One of the best ways to prevent the spread of norovirus is by practicing good hand hygiene.
Most people will fully recover within 2 to 3 days.
Still, it is essential to drink plenty of fluids to prevent dehydration, especially for the very young, elderly, or those with weakened immune systems, which are more at risk.
The U.S. CDC's Vessel Sanitation Program (VSP) requires cruise ships to log and report the number of passengers and crew who say they have symptoms of gastrointestinal illness. As of mid-February 2023, various norovirus outbreaks have been reported to the CDC.
And the list of Cruise Ship Inspection scoring 100 was updated in January 2023.
As of February 20, 2023, the U.S. Food and Drug Administration had not approved a norovirus vaccine.

Tonix Pharmaceuticals Holding Corp. recently announced that it is obtaining an exclusive license from Columbia University in New York for the development of a portfolio of fully human (TNX-3600) and murine (TNX-4100) monoclonal antibodies (mAbs) for the treatment or prevention of SARS-CoV-2 infection.
The Company believes that murine mAbs, such as TNX-4100, have the potential to generate high-affinity antibodies that recognize different epitopes on the SARS-CoV-2 spike protein.
This is because mice have a different repertoire of antibodies than humans.
The technology for generating antibodies optimizes the selection of appropriate B cells by the timing of immunization, harvesting approach, and screening platform.
Seth Lederman, M.D., Chief Executive Officer of Tonix Pharmaceuticals, commented in a press release on February 13, 2023, "Immunocompromised individuals, including organ transplant recipients, are at increased risk of severe COVID-19 and poor clinical outcomes."
"We believe there is a need for second-generation mAb treatments and prophylactics to protect this population."
Although five mAb products containing seven distinct mAbs received emergency use authorization from the U.S. Food and Drug Administration for either treatment or prevention of COVID-19, none have remained useful or available in the U.S. since January 26, 2023.
Other mAbs news is posted at Coronavirus Today.