Search API
AfricaNews recently reported the United Republic of Tanzania requested medical experts investigate a mysterious "communicable" disease that has already killed five people in the country in March 2023.
Outbreaks are not new in the east African country of Tanzania.
In July 2022, a disease whose symptoms included nosebleeds, fever, headaches, and fatigue, was detected in the Lindi region. A total of 13 patients were detected then.
The World Health Organization later confirmed 20 cases of leptospirosis in two districts in the Lindi Region, including three deaths. The majority of these cases were men who were farmers as of August 8, 2022.
And in 2019, a disease with Ebola-like symptoms killed one woman who had visited Uganda, where an Ebola outbreak was detected.
Tanzania was formed as a sovereign state in 1964 through the combination of Tanganyika and Zanzibar, reports Britannica.
No U.S. CDC health notices are in effect for Tanzania, including Zanzibar, as of March 18, 2023.
The World Health Organization (WHO) today announced that after three decades, authorities in the Republic of Burundi declared an outbreak of circulating vaccine-derived polioviruses - type 2 (CVDPV 2) after confirming eight polioviruses.
Circulating poliovirus type 2 infection can occur when the weakened strain of the virus in the oral polio vaccine spreads among under-immunized populations for long periods.
Additionally, five samples from environmental surveillance of wastewater confirmed the presence of the circulating poliovirus type 2.
Dr. Matshidiso Moeti, WHO Regional Director for Africa, commented in a press release on March 17, 2023, "We are supporting the national efforts to ramp up polio vaccination to ensure that no child is missed and faces no risk of polio's debilitating impact."
Burundi's government declared the detection of the poliovirus a national public health emergency and planned to implement a vaccination campaign for eligible children.
Burundi is a landlocked African country, a historical kingdom, and one of the few countries whose borders were not determined by colonial rulers, reports Britannica.
The WHO says polio is a vaccine-preventable disease.
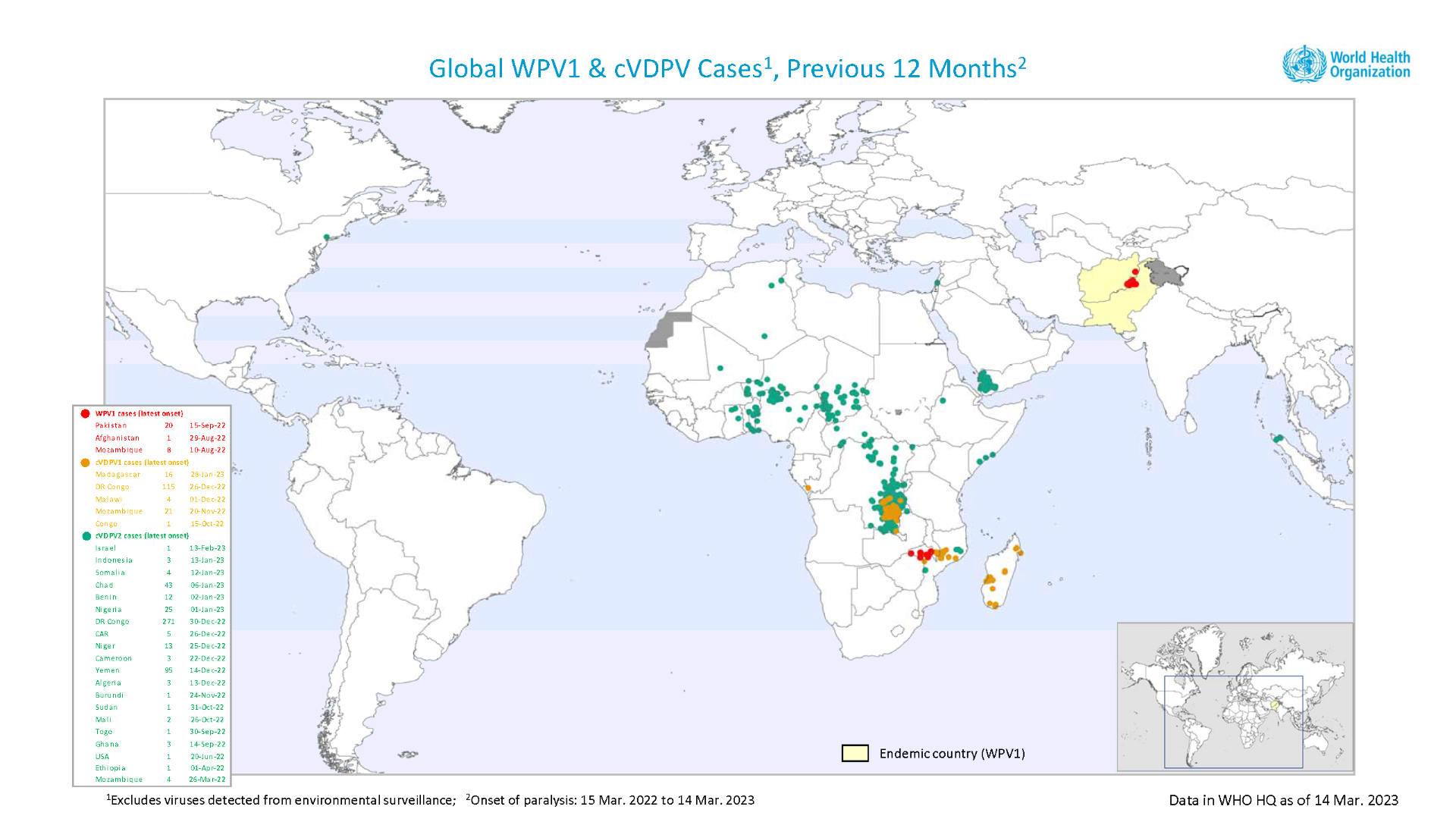
Circulating poliovirus type 2 is the most prevalent form of polio in Africa.
More than 400 cases were reported in 14 countries in 2022.
International travelers were alerted to this and other polio outbreaks when the U.S. Centers for Disease Control and Prevention reissued its Global Polio travel alert in March 2023.
The Janssen Pharmaceutical Companies of Johnson & Johnson recently announced an early-stage antiviral candidate (JNJ-1802) provides strong protection against dengue in non-human primates and mice.
The new data indicate JNJ-1802 is effective against all four dengue serotypes in mouse models and provides strong protection against two tested serotypes (DENV-1 and -2) in non-human primates.
The first-in-class antiviral, which was shown to be safe and well tolerated in a first-in-human clinical study, is progressing into Phase 2 clinical studies for the prevention and treatment of dengue.
"The unprecedented rise in dengue outbreaks throughout the past years offers a glimpse of what lies ahead ....." said Ruxandra Draghia-Akli, M.D., Ph.D., Global Head, Global Public Health R&D at Janssen Research & Development, LLC, in a press release on March 15, 2023.
"We know an antiviral will be critical to addressing the unmet needs today and tomorrow."
The peer-review journal Nature published this study: Blocking NS3–NS4B interaction inhibits dengue virus in non-human primates. These findings support the further clinical development of JNJ-1802, a first-in-class antiviral agent against dengue, which is now progressing in clinical studies to prevent and treat dengue.

Measles outbreaks increased by about 80% during 2022, and disease experts warned of increasing outbreaks in 2023.
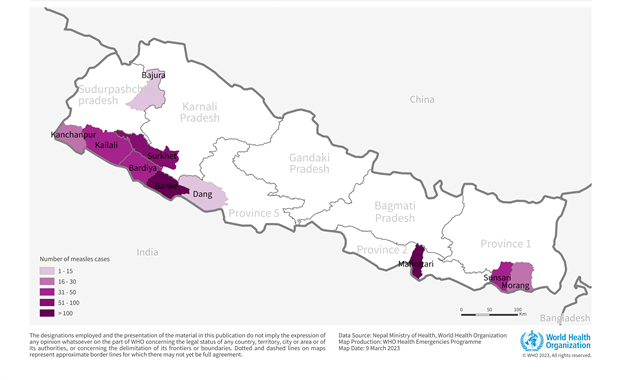
For example, the World Health Organization (WHO) recently announced an outbreak of measles was confirmed in Nepalgunj sub-metropolitan city (SMC) in Banke district, Nepal, following a cluster of fever and rash cases.
Following the confirmation and through active case search as of March 14, 2023, the index case was identified, with the onset of symptoms on November 24, 2022. Between November 24, 2022, and March 10, 2023, 690 measles cases, including one associated death (case fatality ratio: 0.14 %), were reported from seven districts in western Nepal and three in eastern Nepal (Terai ecological region).
The majority of the cases (n=591; 86%) have been reported in children aged less than 15 years.
While measles is endemic in Nepal and is reported every year, the magnitude and extent of the current outbreak are unusually high compared to the previous years.
Only sporadic isolated measles cases had occurred since 2004 when a significant outbreak of over 12,000 cases was reported.
The WHO says the risk of the spread of measles is assessed as high at the national level and moderate at the regional level.
Measles is a vaccine-preventable disease, with various vaccines offered at clinics and community pharmacies in the U.S. during 2023.
The U.S. Centers for Disease Control and Prevention (CDC) Technical Report issued on March 17, 2023, stated that despite the geographic spread of highly pathogenic avian influenza (HPAI) A(H5N1) viruses, only a small number of 'bird flu' cases in people have been identified.
The CDC stated all reported human cases since 2022 have been associated with poultry exposures.
And no bird flu cases of human-to-human transmission have been identified, says the CDC.
However, because of the potential for influenza viruses to evolve rapidly, continued sporadic human infections are anticipated.
To date, more than 6,300 people in 52 jurisdictions have been monitored since 2022, and only one human case in Colorado has been identified.
However, there has been an upsurge in various mammals dying from bird flu in Canada and the U.S. during 2023.
The CDC presented some good news regarding bird flu vaccines.
An H5 candidate vaccine virus (CVV) produced by CDC is nearly identical or, in many samples, identical to the hemagglutinin (HA) protein of recently detected clade 2.3.4.4b HPAI A(H5N1) viruses in birds and mammals and could be used to produce a vaccine for people.
This H5 CVV is available and has been shared with vaccine manufacturers.
One manufacturer, CSL Seqirus Inc., has already produced a U.S. FDA-approved vaccine, Audenz™.
Audenz is a monovalent, adjuvanted, cell-based inactivated influenza (H5N1) subunit vaccine designed to protect people from bird flu.

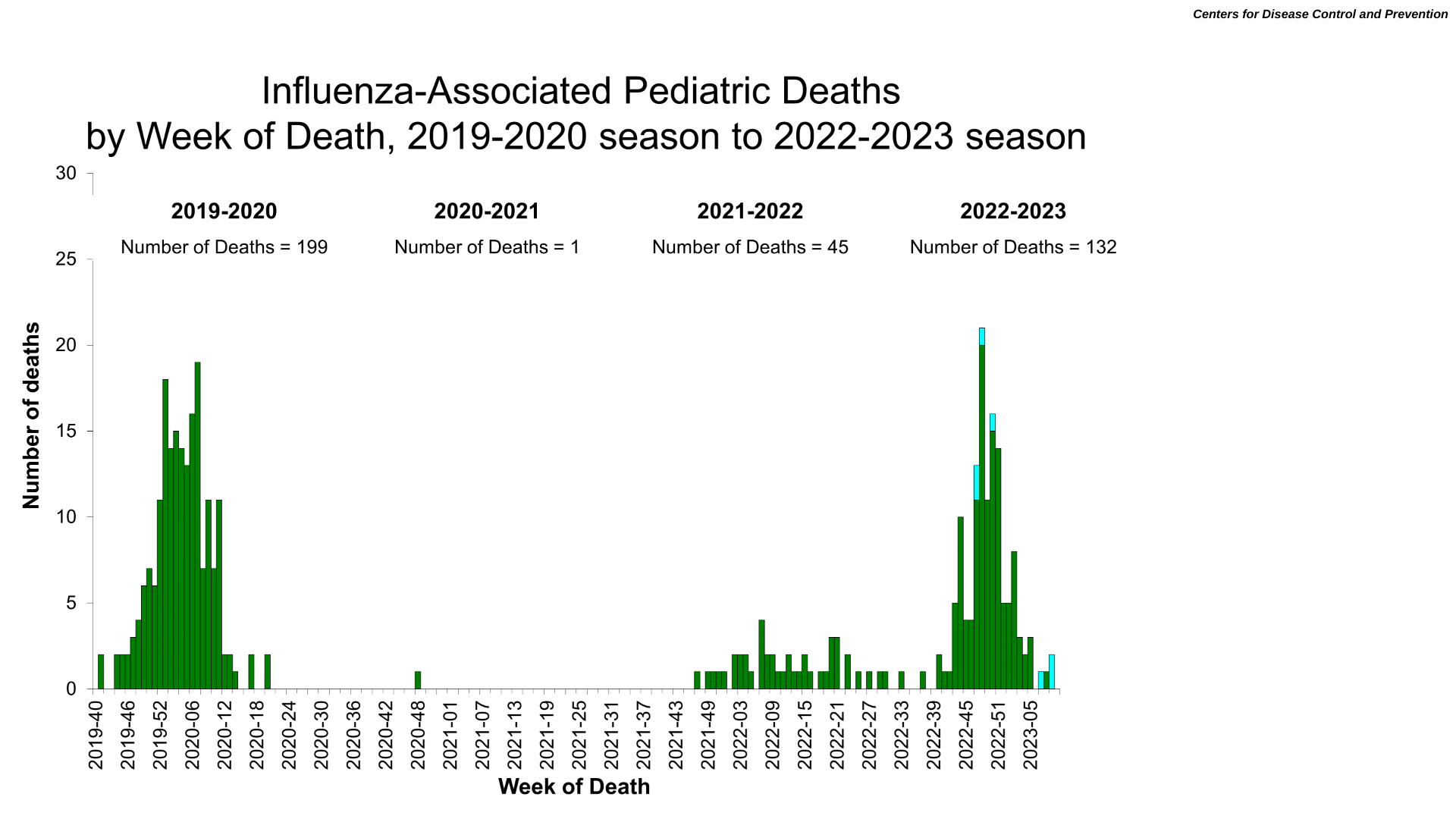
The U.S. Centers for Disease Control and Prevention (CDC) today reported two insightful data points. First, as of March 17, 2023, the CDC's national outpatient respiratory illness data is now below baseline, as are six of 10 HHS regions.
This data indicates the 2022-2023 flu season in the U.S. is coming to an early close.
However, as of week #10 data, seven additional influenza-associated pediatric deaths occurred.
The deaths occurred between week #47 of 2022 and week #9 of 2023.
All seven deaths were associated with influenza A viruses. Four of the influenza A viruses had subtyping performed; one was an A(H1N1) virus, and three were A(H3) viruses.
This CDC update means there have been 132 influenza-associated pediatric deaths this flu season, the most significant amount since 2019.
The CDC continues to remind everyone that flu shots remain available at most health clinics and pharmacies in the U.S.
GSK plc recently announced positive results from a phase 3 clinical trial evaluating its MenABCWY combination vaccine candidate, administered as two doses given six months apart in healthy individuals aged 10-25 years.
GSK's MenABCWY vaccine candidate combines the antigenic components of its licensed meningococcal vaccines, Bexsero (MenB) and Menveo (MenACWY).
All primary endpoints were met, including the non-inferiority of the vaccine candidate for all five Neisseria meningitides serogroups (A, B, C, W, and Y) compared to licensed meningococcal vaccines Bexsero and Menveo in terms of an immune response.
In addition, the vaccine candidate was well tolerated, with a safety profile consistent with Bexsero and Menveo.
Tony Wood, Chief Scientific Officer at GSK, commented in a press release on march 14, 2023, "These statistically significant phase III data are a very encouraging step toward reducing the incidence of meningococcal disease."
"In the U.S., routine use of a 5-in-1 meningococcal vaccine with a two-dose regimen in adolescents at 16 to 18 years of age, just before this disease's incidence peak, could drive significant public health impact."
Invasive meningococcal disease (IMD), a significant cause of meningitis and septicemia, is an uncommon but serious illness that can cause life-threatening complications or even death, typically amongst previously healthy children and adolescents.
Five Neisseria meningitides serogroups (A, B, C, W, and Y) account for nearly all IMD cases worldwide.
Among those contracting meningococcal diseases, one in ten will die, sometimes in as little as 24 hours, despite treatment.
As yet, no licensed combination vaccine offers protection against these serogroups in a single vaccine.
Currently, in the U.S., two separate vaccines needing four injections are required to protect against all five serogroups.
This immunization regimen and low awareness of the disease can lead to sub-optimal immunization coverage rates, particularly for MenB, with an estimated coverage of only about 31% of adolescents in the U.S.
GSK works closely with regulators to review the complete phase III data set, including the supplemental Biologics License Application for Bexsero.
This clinical trial was the confirmatory trial for Bexsero and the phase III trial for MenABCWY.
Detailed results from this phase III trial will be presented in a peer-reviewed publication and at upcoming scientific meetings.