Search API
In the United States, the timing of seasonal respiratory syncytial virus (RSV) outbreaks throughout the country is generally reported between October - April most years.
The 2022–23 season it started later than in 2021–2022 but earlier than prepandemic seasons, suggesting a return toward normal seasonality.
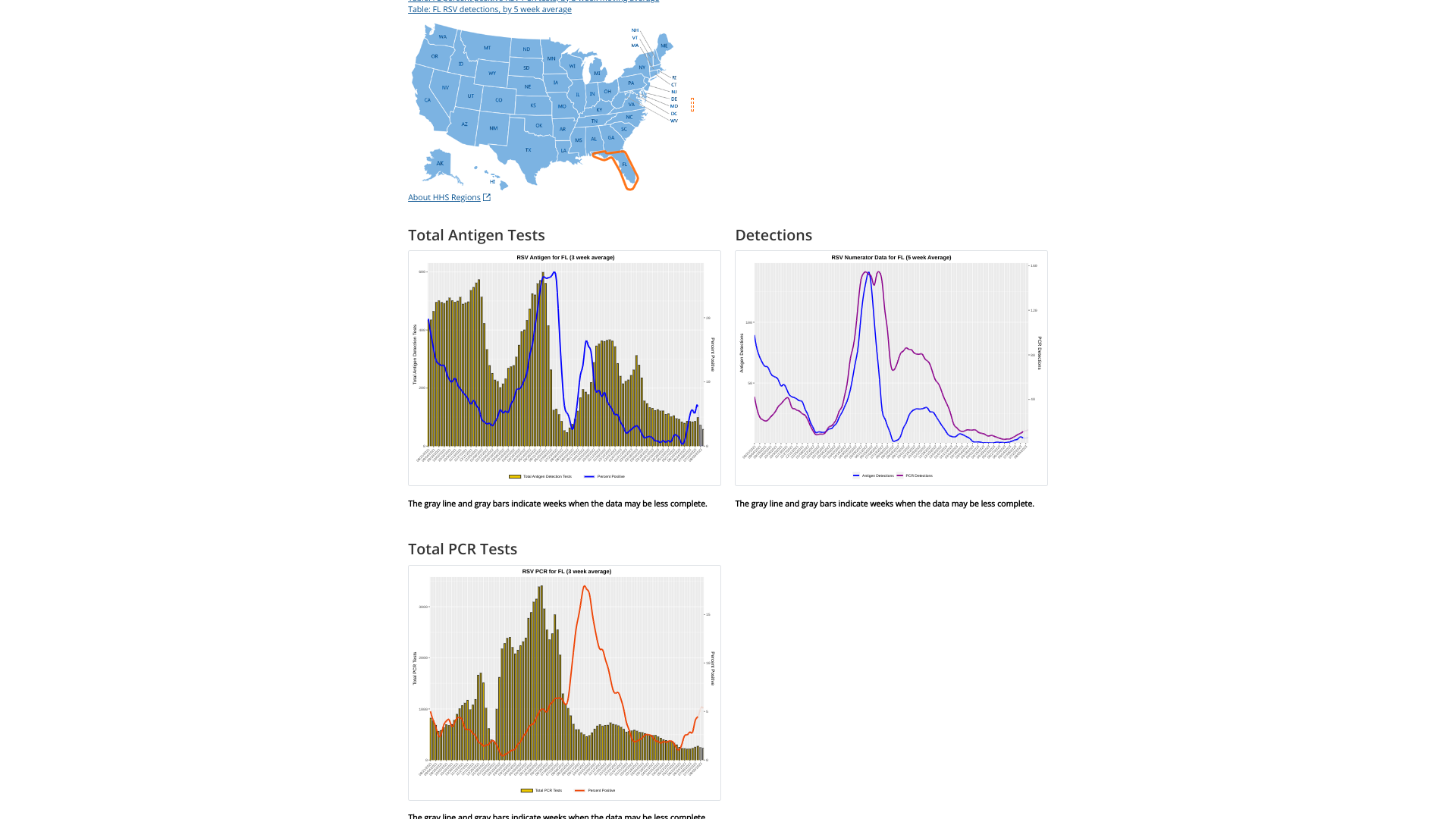
Furthermore, the state of Florida is often a bell-weather state for RSV detections, as it is in 2023.
Florida’s RSV season is longer than the rest of the nation and has distinct regional patterns. For this reason, the state is segmented into five RSV regions, each with its own season.
The Florida Department of Health recently announced RSV activity during week #33 had increasing detection and admission rates.
As of August 19, 2023, two RSV outbreaks were confirmed in Martin County, located north of Jupiter. Last week, one RSV outbreak occurred in Jacksonville, Florida.
In the U.S., RSV infections cause thousands of hospitalizations among young children and adults aged ≥65. The U.S. Centers for Disease Control and Prevention (CDC) RSV detection 5-week moving average graphs for each state are displayed at this link.
This RSV season, the U.S. government has approved RSV vaccines and monoclonal antibody therapeutics available in specific clinics and pharmacies as of August 22, 2023.
The CDC says people should speak with a healthcare provider if these new therapeutics are appropriate for their health.
Novavax, Inc. today announced that its updated protein-based XBB COVID vaccine candidate induced neutralizing antibody responses to the EG.5.1 and XBB.1.16.6 subvariants in small pre-clinical studies.
As of August 22, 2023, SARS-CoV-2 coronavirus XBB sublineage variants are overwhelmingly responsible for the majority of current COVID-19 cases in the U.S. and European Union.
"Our data have shown that Novavax's protein-based COVID vaccine induces broadly neutralizing responses against XBB subvariants, including EG.5.1 and XBB.1.16.6," commented Filip Dubovsky, President of Research and Development, Novavax, in a press release.
Non-clinical data previously showed that Novavax's COVID vaccine candidate induced functional immune responses for XBB.1.5, XBB.1.16, and XBB.2.3 variants, indicating a broad response that could potentially be applicable for forward-drift variants.
Novavax is submitting applications for its XBB.1.5 COVID vaccine candidate to regulatory authorities globally.
Novavax COVID-19 vaccine brands (Nuvaxovid, CovoVax, NVX-CoV2373, TAK-019) have been authorized in about 40 markets.
The Novavax COVID-19 Vaccine, Adjuvanted, has not been approved or licensed by the U.S. FDA but is authorized for emergency use. Novavax vaccines are available in specific clinics and pharmacies in the U.S.

The U.S. Food and Drug Administration (FDA) today approved ABRYSVO™, the first vaccine approved for use in pregnant women to prevent lower respiratory tract disease (LRTD) and severe LRTD caused by respiratory syncytial virus (RSV) in infants from birth through six months of age.
Abrysvo is approved for use at 32 through 36 weeks gestational age of pregnancy, says the FDA.
Previously, the FDA approved Pfizer Inc.'s Abrysvo in May 2023 to prevent LRTD caused by RSV in individuals 60 and older.
"RSV is a common cause of illness in children, and infants are among those at highest risk for severe disease, which can lead to hospitalization," said Peter Marks, M.D., Ph.D., director of the FDA's Center for Biologics Evaluation and Research, in a press release on August 21, 2023.
"This approval provides an option for healthcare providers and pregnant individuals to protect infants from this potentially life-threatening disease."
Recently, the FDA approved Beyfortus™ for infants born during or entering their first RSV season and for children up to 24 months of age who remain vulnerable to severe RSV disease through their second RSV season.
On July 17, 2023, Beyfortus became the first extended half-life monoclonal antibody offering passive immunization to prevent LRTI caused by RSV.
RSV is a highly contagious virus that causes respiratory infections in individuals of all age groups, causing frequent outbreaks. Most individuals can be expected to be infected with RSV by the time they reach two years of age, says the FDA.
In most parts of the U.S., RSV circulation is seasonal, typically starting in Florida and peaking in the winter.
A study published by PLOS Medicine in July 2023 concluded RSV disease burden is high in the nearly 600 million children under five living in 121 low-income and middle-income countries. The peak age of community-based RSV incidence is 4.8 months.

A recent study concluded the high proportion of children too young to be vaccinated among unvaccinated Invasive meningococcal disease (IMD) cases suggests that starting the vaccination earlier may prevent more of these cases.
The JAMA Network published an Original Investigation on a screening cohort study and matched case-control study on August 18, 2023, which found high effectiveness of a 4-component recombinant protein–based (4CMenB) vaccination and more significant reduction in incidence rate ratios (IRR) for early-start vaccination schedules in preventing invasive serogroup B meningococcal disease.
This case-control study represents the most comprehensive multiregional evaluation of the effectiveness of 4CMenB vaccination in the pediatric population of Italy.
Vaccine effectiveness (VE) data obtained from a large group of serogroup B IMD cases with the simultaneous application of 2 independent computational methods (screening and case-control) are unique to the literature.
And VE was firmly greater than 90% in children old enough to receive the first vaccine dose.
Regional differences in the vaccination schedule allowed population-based comparison of outcomes and confirmed the greater efficacy associated with early-start strategies. At the same time, a lack of protection in the very early months of life was apparent even when starting immunization at age two months, prompting the identification of extended prevention strategies.
Current, population-based evidence about VE and reduction in IRRs associated with 4CMenB has been reported in studies conducted in the UK, Australia, Canada, Portugal, and Italy, with heterogeneous methods and across different healthcare settings and age groups.
The estimates of VE for 4CMenB ranged from 59% to 100% in fully vaccinated cohorts.

CARsgen Therapeutics Holdings Limited today announced a collaboration agreement with Moderna Inc. to investigate CARsgen's investigational Claudin18.2 CAR T-cell product candidate (CT041) in combination with Moderna's investigational Claudin18.2 mRNA cancer vaccine.
CT041 (satricabtagene autoleucel) is CARsgen's autologous CAR T-cell product being investigated for potentially treating gastric, pancreatic, and other specified digestive system cancers.
It is currently in multiple ongoing clinical studies in China and North America.
"CT041 is the most advanced solid tumor CAR-T in development (pivotal phase II) and continues to show promise in treating gastric and pancreatic cancers. In our quest to make cancer curable, we are continuously exploring multiple modalities to eradicate tumors. Attacking tumors with CAR T-cell therapy in combination with a cancer vaccine could potentially provide greater clinical benefit to patients." said Dr. Zonghai Li, Founder, Chairman of the Board, Chief Executive Officer, and Chief Scientific Officer of CARsgen Therapeutics Holdings Limited, in a press release on August 21, 2023.
Dr. Li added, "Moderna has clearly established itself as a scientific and commercial leader in mRNA-based vaccines and therapeutics, and we are pleased to partner with Moderna to explore a potential synergism between our innovative therapies."
Moderna is developing an investigational off-the-shelf mRNA cancer vaccine that encodes for the Claudin18.2 protein, a tumor-associated antigen.
The collaboration contemplates conducting preclinical studies and a phase I clinical trial to evaluate CT041 in combination with Moderna's Claudin18.2 mRNA cancer vaccine.
"We are pleased to partner with CARsgen to explore the potential synergy of CAR-T with an investigational mRNA cancer vaccine that encodes for the Claudin18.2 protein. Claudin18.2 is a promising therapeutic target to potentially treat multiple cancer types with high unmet medical need. We continue to deliver on the promise of mRNA science to create a new generation of transformative medicines in oncology," added Dr. Lin Guey, Chief Scientific Officer of External Research Ventures, Moderna.
CARsgen is a biopharmaceutical company with operations in China, and the U.S. focused on innovative CAR T-cell therapies for treating hematologic malignancies and solid tumors.