Search API
Last year’s flu season began earlier than expected in the United States, and many children were not protected from influenza.
To better understand this health risk, the U.S. Centers for Disease Control and Prevention (CDC) Clinician Outreach and Communication Activity webinar on August 31, 2023, presented an update on 2023-2024 influenza recommendations for non-adults.
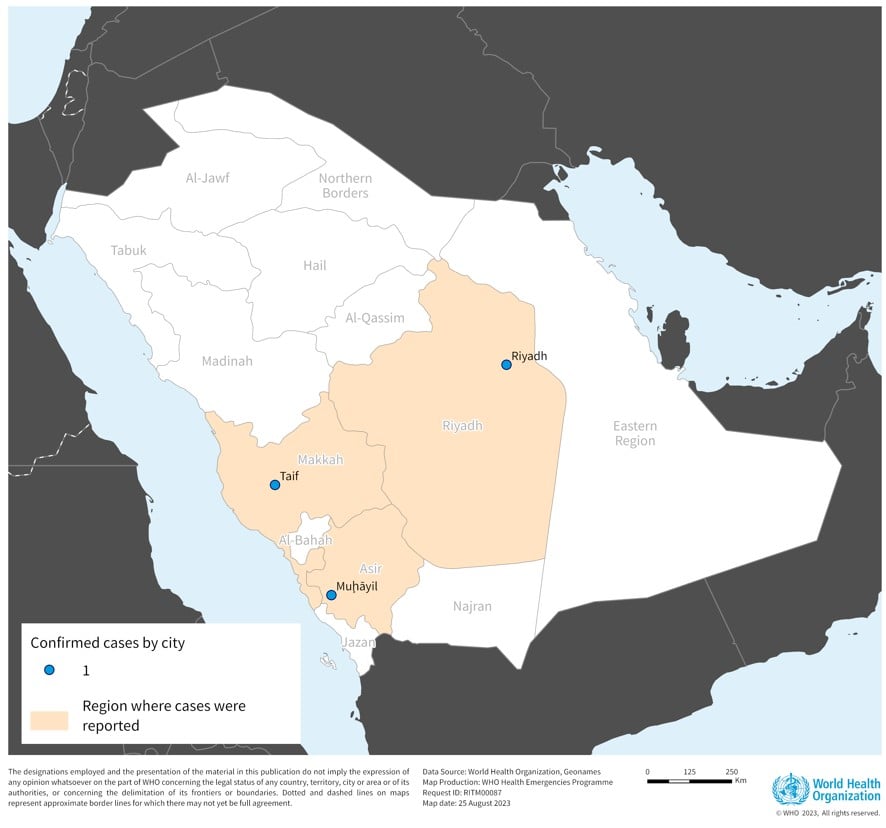
This digital presentation revealed on slide #42 that influenza vaccination coverage in children six months to 17 steadily decreased during 2019-2023.
During 2019-2022, 62.2% of children got an annual flu shot.
In 2022-2023, only 55.1% of children received an annual flu shot.
Furthermore, the CDC reported that influenza-associated pediatric deaths during the 2022-2023 season have reached 172, a three-year high.
The CDC says annual flu shots for most people over six months of age are the most effective way to prevent influenza illness and associated complications.
From a protection perspective, the CDC says over 65% of influenza-vaccinated children were protected from flu-related hospitalization during 2023.
A clarification for this flu season is people with egg allergy may receive any influenza vaccine (egg-based or non-egg-based) that is otherwise appropriate for their age and health status; additional safety measures are no longer recommended.
And multiple vaccines can be administered along with a flu shot.
Additionally, coincident with National Immunization Awareness Month in August, the American Academy of Pediatrics released an updated policy statement and technical report on August 29, 2023, with recommendations for preventing and controlling influenza in children for the 2023-’24 influenza season.
As the summer of 2023 comes to a close, disease-carrying mosquitoes in Florida are not taking a break.
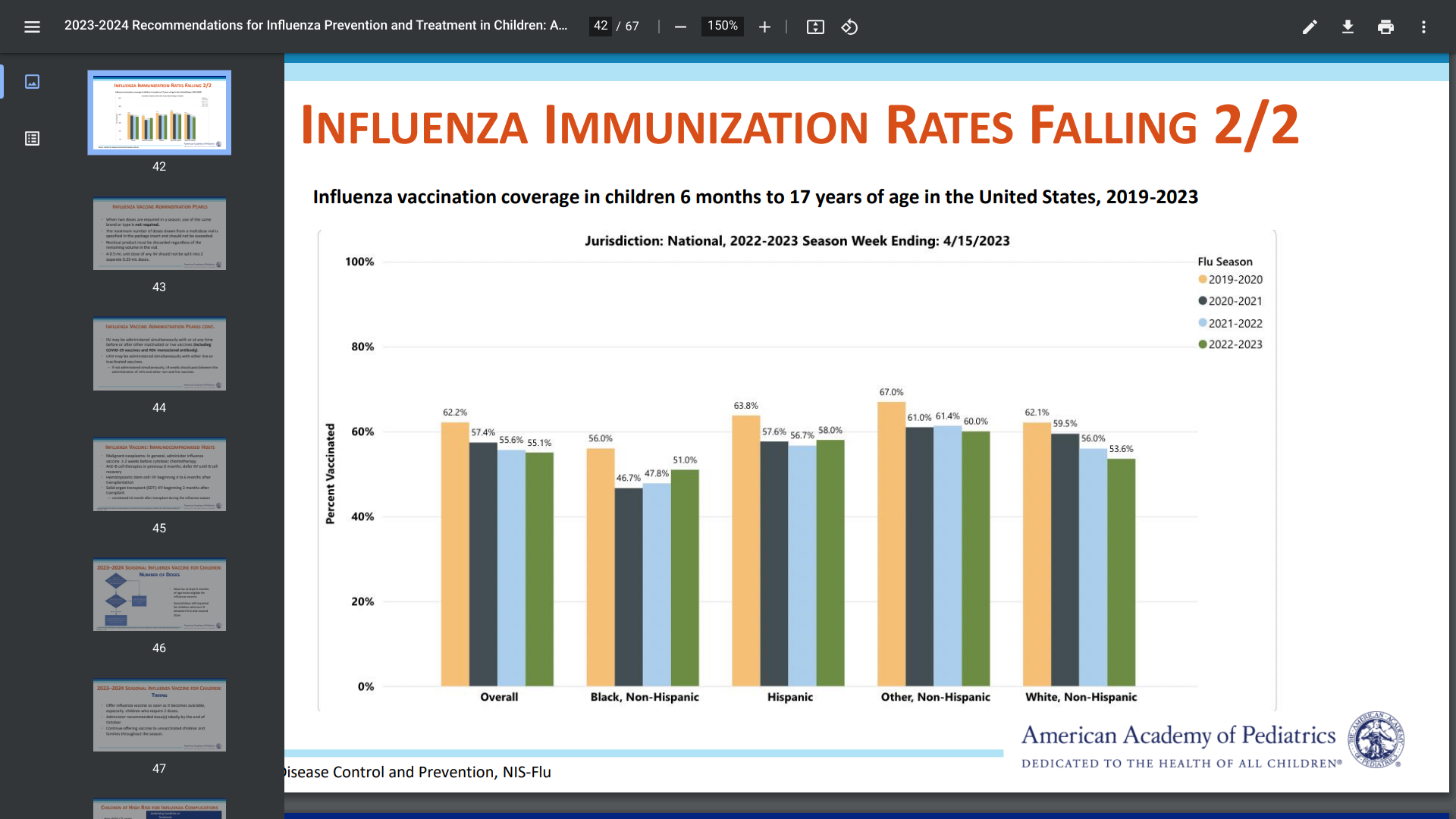
According to the Florida Department of Health's week #34 report, 21 new dengue virus cases were reported last week.
On August 28, 2023, twenty new dengue cases were reported this week in persons who had international travel. There are now 244 travel-associated dengue cases reported in Florida this year.
These dengue-infected visitors primarily came from Cuba (165) and Brazil (13).
Additionally, and more concerning, one new locally acquired dengue was reported last week in Miami-Dade County. In 2023, 16 cases of locally acquired dengue have been reported this year, with Miami-Dade confirmed 12 patients.
This data is positive news compared to 2022 when 68 locally acquired dengue cases were reported.
Furthermore, it appears the malaria outbreak in the Sarasota area has subsided. No cases of locally acquired malaria were reported last week.
In 2023, seven cases of locally acquired malaria have been reported.
While the U.S. CDC has not issued a travel advisory for these mosquito-carry diseases, various alerts have been issued by Escambia, Jefferson, Nassau, Orange, Polk, St. Johns, Walton, Broward, Manatee, Miami-Dade, and Sarasota counties.
In total, the CDC has confirmed 642 dengue cases in 38 jurisdictions this year, including San Juan, Puerto Rico with 41 cases.
From a disease prevention perspective, one dengue vaccine is approved for use in the U.S. The Dengvaxia® live attenuated tetravalent chimeric vaccine requires pre-admission testing.
The World Health Organization (WHO) recently announced the initiation of the second round of polio vaccinations in six provinces within the Islamic Emirate of Afghanistan.
The WHO's goal is to ensure every child in the selected provinces.
These vaccinations are essential since five wild poliovirus type 1 cases were confirmed in Afghanistan in August 2023.
Although the number of positive samples in Afghanistan increased in 2023 compared to the year 2022, this was partly due to more intensive surveillance in the country, stated the WHO on August 25, 2023.
The WHO's International Travel and Health also recommends that all travelers to polio-affected areas be fully vaccinated against polio.
Residents (and visitors for more than four weeks) from infected areas should receive an additional dose of OPV or inactivated polio vaccine within four weeks to 12 months of travel.
As of September 2023, the Global Polio Eradication Initiative is focusing on reaching every last child in Afghanistan with vaccines, strengthening surveillance, and maintaining political commitment, financial resources, and technical support at all levels.
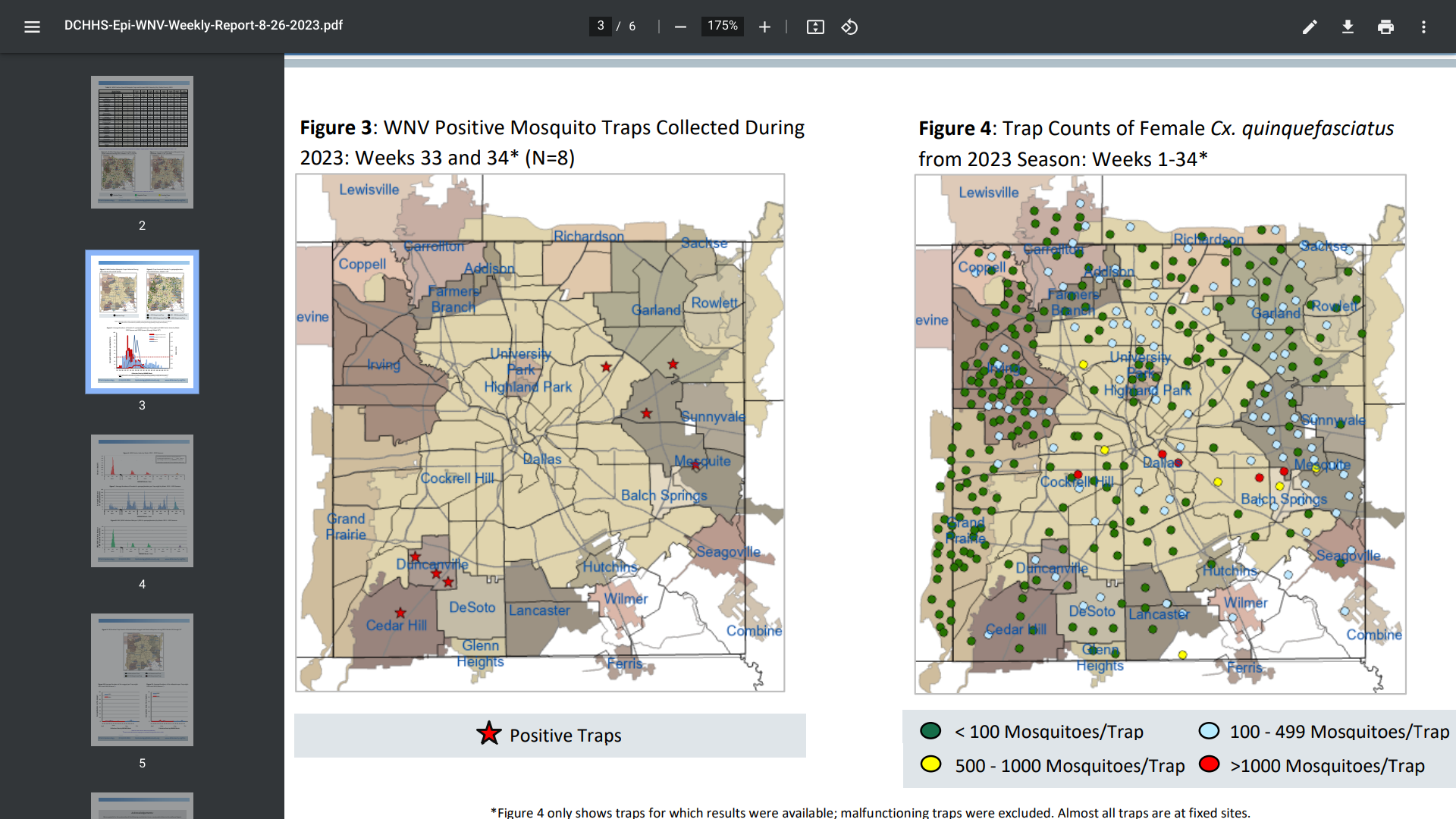
The Dallas County Health and Human Services (DCHHS) recently reported additional Dengue and West Nile virus (WNV) cases, as Aedes albopictus and Aedes aegypti mosquitoes are currently circulating in the area.
As of Week #34, ending August 26, 2023, DCHHS reported that one hundred eighty-eight mosquito traps tested positive for WNV.
And 15 human WNV cases and one human death have been reported in 2023.
Additionally, four travel-related Dengue cases have been reported this year in Dallas.
"It's important for people to be aware that there are many diseases transmitted by mosquitoes found in Texas," said Texas DSHS Commissioner Jennifer Shuford, MD, MPH, in a July 2023 press release.
"Most of these diseases cause mild illness, but in rare instances, diseases like dengue or Zika can cause severe illness."
To service residents, DCHHS provides free or low-cost public health and social services to low-income residents of Dallas County.
Philip Huang, MD, MPH, DCHHS's Director, says Dallas County has been at the forefront of the local response to major public health issues and emergencies, including Dengue, Mpox, West Nile, and Zika viruses.
Throughout the U.S., West Nile virus has been detected in 36 states, mainly in the southwest.
And the state of Florida has reported both travel-related and locally acquired dengue cases in 2023.
Influenza remains a serious threat to children due to its potential to cause severe morbidity and mortality during the 2023-2024 flu season, says the U.S. Centers for Disease Control and Prevention (CDC).
The CDC has scheduled a Clinician Outreach and Communication Activity call on August 31, 2023, to update Pediatric Providers regarding the 2023-2024 influenza recommendations.
The CDC says annual flu shots are the most effective way to prevent influenza illness and associated complications. And clinicians play a critical role in immunizing children, their family members, and caregivers.
As of late August 2023, over 100 million flu shots have been distributed in the U.S.
The webinar/call is from 2:00 PM – 3:00 PM ET on August 31, 2023, and is available from this CDC webpage.

The European Medicines Agency (EMA) today announced the Human Medicines Committee (CHMP) has recommended authorizing an adapted Comirnaty vaccine targeting the Omicron XBB.1.5 subvariant.
COVID-19 vaccines are being adapted to better match the circulating SARS-CoV-2 variants.
On August 30, 2023, the EMA stated the Comirnaty Omicron XBB.1.5 vaccine is to prevent COVID-19 in adults and children from 6 months of age.
In its decision to recommend the authorization, the CHMP considered all available data on Comirnaty and its other adapted vaccines, including safety, efficacy, and immunogenicity data.
In addition, the CHMP assessed new laboratory data showing a strong response of the adapted vaccine against XBB.1.5 and related strains of the betacoronavirus that causes COVID-19.
The EMA will now send the CHMP’s recommendation to the European Commission for an EU-wide legally binding decision.
“As COVID-19 is expected to adopt a seasonal pattern, similar to other respiratory viruses, we remain committed to providing COVID-19 vaccines that are better matched to relevant circulating variants or sublineages to people worldwide to support vaccinations in the upcoming fall and winter season,” said Prof. Ugur Sahin, M.D., CEO and Co-founder of BioNTech, in a press release.
“Omicron XBB-related sublineages are antigenically distant from prior Omicron strains and continue to account for most COVID-19 cases globally. The updated COVID-19 vaccine aims to improve protection against severe illness and hospitalization further.”
In line with previous recommendations by EMA and the European Centre for Disease Prevention and Control, adults and children from 5 years of age who require vaccination should have a single dose, irrespective of their COVID-19 vaccination history.
Children from 6 months to 4 years of age may have one or three doses depending on whether they have completed a primary vaccination course or have had COVID-19.
The EMA stated that more data on emerging variants are expected, and the Committee will assess these data when available.
Pfizer and BioNTech have also filed an application with the U.S. Food and Drug Administration requesting approval of their Omicron XBB.1.5-adapted monovalent COVID-19 vaccine and expect a decision in the coming days. The companies have submitted data for the updated COVID-19 vaccine to other regulatory authorities worldwide.
As with the other eleven WHO-Listed COVID-19 vaccines, national authorities determine how to use vaccines in national vaccination campaigns, considering factors such as infection and hospitalization rates, the risk to vulnerable people, and vaccine availability.

Alzamend Neuro, Inc. today announced that it has submitted an investigational new drug (IND) application to the U.S. Food and Drug Administration (FDA) for the initiation of study AL001-BD01, a Phase IIA clinical study of AL001 for Bipolar Disorder (BD).
Alzamend believes this program may qualify for the 505(b)(2) pathway for FDA approval, which is available to new formulations of an approved drug.
Once the IND is cleared by the FDA, Alzamend intends to initiate the Phase IIA study to determine relative increased lithium levels in the brain compared to a marketed lithium salt in BD patients, based on published mouse studies that predict that lithium can be given at lower doses for equivalent therapeutic benefit when treated with AL001.
Alzamend's goal is to replace a 300 mg TID lithium carbonate dose for the treatment of BD with a 240 mg TID AL001 lithium equivalent, which represents a daily decrease of 20% of lithium given to a patient.
"This IND submission represents a key milestone for Alzamend as we continue to advance our proprietary pipeline," commented Stephan Jackman, Chief Executive Officer of Alzamend, in a press release on August 30, 2023.
"Being able to develop a next-generation lithium product (AL001) that would not routinely require therapeutic drug monitoring (TDM) could positively impact the 7 million Americans afflicted with BD."
Lithium is a commonly prescribed drug for manic episodes in BD type 1 and maintenance therapy of BD in patients with a history of manic episodes.
AL001 is a novel lithium-delivery system that can potentially deliver the benefits of marketed lithium salts while mitigating or avoiding currently experienced toxicities associated with lithium.
Results from Alzamend's recently completed Phase IIA multiple-ascending dose study of AL001 in Alzheimer's and healthy patients identified a maximum tolerated dose ("MTD"), as assessed by an independent safety review committee.
This MTD, providing lithium at a lithium carbonate equivalent dose of 240 mg 3-times daily, is designed to be unlikely to require TDM.
Moreover, this dose is designed to distribute more lithium to the brain but at lower systemic exposure, resulting in an improved safety profile compared to currently marketed lithium salts, thereby avoiding clinical disadvantages.
BD, previously known as manic depression, is a mood disorder with three types, according to the U.S. NIH.
In the U.S., about 3% of the population is estimated to be affected by BD at some point in their life.
BD is among the top 20 causes of disability worldwide, leading to substantial societal costs. Other mental health issues, such as anxiety and substance use disorders, are commonly associated with BD.
The FDA has not approved an Alzheimer's Disease or BD vaccine candidate as of August 30, 2023.

The World Health Organization (WHO) today announced that from September 2022 to August 12, 2023, three laboratory-confirmed cases of Middle East respiratory syndrome coronavirus (MERS-CoV), including two deaths, were reported by the Ministry of Health of the Kingdom of Saudi Arabia (KSA).
Close contacts for the three cases from Riyadh, Asser, and Makkah Al Mukarramah regions were followed up by the Ministry of Health, and no secondary cases were identified.
Of the three male cases, two had a history of contact with dromedary camels, and all three patients had a history of consumption of raw camel milk in the 14 days before the onset of symptoms.
The WHO wrote on August 29, 2023, that it expects that additional cases of MERS-CoV viral respiratory infections will be reported from the Middle East and/or other countries where MERS-CoV is circulating in dromedaries and that patients will continue to be exported to other countries by individuals who were exposed to the virus.
For example, a MERS outbreak was confirmed in the Republic of Korea in 2015.
Since the beginning of 2023, and as of late July 2023, one MERS-CoV case was reported by UAE authorities.
Furthermore, human-to-human transmission of MERS-CoV has been confirmed in healthcare settings.
WHO stated it does not advise special screening at points of entry regarding this event, nor does it recommend applying any travel or trade restrictions.
Since the first report of MERS-CoV in KSA in 2012, human infections have been reported from 27 countries in all six WHO regions.
Of the reported 2605 cases, most MERS-CoV cases (2196; 84%) have been reported from KSA. Approximately 36% of patients with MERS have died.
A study published by the International Journal of Infectious Diseases on March 29, 2023, confirms MERS-CoV remains a threat to global health security. as variants continue circulating in humans and camels.
No vaccine or specific treatment is currently available, although several MERS-CoV-specific vaccines and therapeutics are in development as of late August 2023.