Search API
Since Ebola virus disease (EVD) impacts everyone in an outbreak area, another agency has approved a preventive vaccine for young children.
Merck today announced that the European Commission (EC) approved an expanded indication for the ERVEBO® vaccine for active immunization of individuals one year of age or older to protect against EVD caused by Zaire ebolavirus.
ERVENO was previously approved for use in the European Union for individuals 18 or older.
The U.S. FDA recently issued similar approval for young children.
ERVEBO is approved in the European Union, United Kingdom, United States, Canada, Switzerland, and ten countries in Africa.
ERVEBO is a live recombinant viral vaccine with a vesicular stomatitis virus backbone that protects people from Zaire ebolavirus. This vaccine does not protect people from the Sudan ebolavirus or the Marburg virus.
Dr. Eliav Barr, senior vice president, head of global clinical development, and chief medical officer, Merck Research Laboratories, commented in a press release on September 7, 2023, “When outbreaks of Ebola virus disease occur, they can quickly become a public health crisis. We are proud to play a role, alongside the global public health community, in helping to prepare for potential outbreaks of Zaire ebolavirus.”
In January 2021, Merck confirmed an agreement with UNICEF to establish the world’s first global Ebola vaccine stockpile to support future Zaire ebolavirus (EBOV) outbreak preparedness and response efforts.
Over 500,000 doses of ERVEBO have been delivered to the stockpile, administered by the International Coordinating Group on Vaccine Provision.
The initial EBOV case was confirmed in 1976 in the African countries of South Sudan and the Democratic Republic of Congo. Recent data suggest EBOV outbreaks may originate from human-to-human transmission instead of spillover events.
The U.S. Centers for Disease Control and Prevention published a list of EBOV Cases and Outbreaks as of August 2023.

While there are no approved human vaccines for Lyme disease, results from a new study show a booster dose can produce a strong immune response in children, adolescents, and adults.
France-based Valneva SE and Pfizer Inc. today announced positive pediatric and adolescent immunogenicity and safety data for their Lyme disease vaccine candidate, VLA15, when given as a booster.
The VLA15-221 Phase 2 clinical trial showed a strong anamnestic antibody response for all serotypes in pediatric (5 to 11 years of age) and adolescent participants (12 to 17 years of age), as well as in adults (18 to 65 years of age), one month after administration of a booster dose (month 19).
Depending on the primary vaccination schedule (month 0-2-6 or month 0-6), participants seroconverted after the booster dose, yielding seroconversion rates of 95.3% and 94.6% for all outer surface protein A (OspA) serotypes in all age groups, respectively.
Additionally, OspA antibody titers were significantly higher one month after the booster dose compared to one month after the primary schedule, with 3.3- to 3.7-fold increases (Geometric Mean Fold Rises) in adults, 2.0- to 2.7-fold increases in adolescents and 2.3- to 2.5-fold increases in children for all serotypes.
“Protection against Lyme disease is important for anyone who lives or spends time outdoors in areas where Lyme disease is endemic. This data from the VLA15-221 study is vital to improve our understanding of how vaccination may help to protect both adults and children from this potentially devastating disease,” commented Annaliesa Anderson, Ph.D., Senior Vice President and Head of Vaccine Research and Development at Pfizer, in a press release on September 7, 2023.
These results follow six-month antibody persistence data in children and adults reported for the VLA15-221 study in December 20222 and positive immunogenicity and safety data reported in April 20223.
Pfizer aims to submit a Biologics License Application to the U.S. Food and Drug Administration and a Marketing Authorisation Application to the European Medicines Agency in 2026, subject to positive Phase 3 data.

Moderna, Inc. today announced that clinical trial data from its research assay confirm its updated COVID-19 vaccine, which is pending approval by the U.S. Food and Drug Administration, generates an 8.7-fold increase in neutralizing antibodies in humans against BA.2.86 (Pirola), a new variant under monitoring.
The U.S. Centers for Disease Control indicates that the highly mutated BA.2.86 variant may be more capable of causing infection in people who previously had COVID-19 or were vaccinated with previous vaccines.
"These results demonstrate that our updated COVID-19 vaccine generates a strong human immune response against the highly mutated BA.2.86 variant. Taken together with our previously communicated results showing a similarly effective response against EG.5 and FL.1.5.1 variants, these data confirm that our updated COVID-19 vaccine will continue to be an important tool for protection as we head into the fall vaccination season," said Stephen Hoge, M.D., President of Moderna, in a press release on September 6, 2023.
Public health authorities are vigilantly monitoring the BA.2.86 variant, a highly mutated strain of the SARS-CoV-2 betacoronavirus with over 30 mutations as compared to prior Omicron strains.
Moderna's clinical trial data around its updated COVID-19 vaccine's effectiveness against BA.2.86 have been shared with regulators and submitted for peer review publication.

The Centers for Disease Control and Prevention (CDC) today announced it is preparing for co-circulating influenza virus, SARS-CoV-2, and respiratory syncytial virus (RSV) during the 2023-2024 season.
The CDC stated on September 5, 2023, that vaccines can provide life-saving protection against all three viral respiratory diseases.
Furthermore, the CDC recommends these vaccines for older adults, who are at a higher risk of severe illness from these diseases.
For example, the CDC recently published a Health Advisory about increased RSV activity in the Southeastern United States, including Georgia and Florida.
During a Clinician Outreach and Communication Activity (COCA) Call on September 19, 2023, CDC presenters will provide updates about the latest recommendations and clinical considerations for administering influenza, COVID-19, and RSV vaccines to seniors this year.
This COCA Webinar link is https://www.zoomgov.com/j/1606020154, ID: 160 602 0154, and Passcode: 493303.
According to the CDC, clinicians play a vital role in ensuring that older adults protect themselves by encouraging them to stay up to date on vaccinations.

GSK and the YMCA today announced they are hosting GSK's Sideline RSV "Community Conversations" event series to raise awareness about respiratory syncytial virus (RSV) infection in older adults.
This is an essential event since the U.S. CDC issued a Health Advisory on September 5, 2023, regarding the 2023-2024 RSV season.
The events will occur at four YMCA locations in Chicago, Los Angeles, New York City, and Phoenix, joined by GSK's campaign spokesperson Earvin "Magic" Johnson Jr., to help spark important conversations about RSV.
Kicking off in September 2023 and running through RSV Awareness Month in October, the private events will include perspectives from medical professionals and patients and convene public health leaders, media, and 60+ YMCA members.
"As a dedicated public health advocate, I'm excited to be able to visit older adults in their communities to talk about their risk for RSV infection," said Johnson in a press release.
"Like many of us, and being over 60 myself, I used to be unaware of RSV's effect on older adults."
"Now, I'm passionate about educating others and encouraging them to understand their risks and be proactive about their health."
For older adults, data suggest an increased risk for severe RSV infection that can lead to hospitalization.
RSV can exacerbate certain underlying medical conditions, such as chronic obstructive pulmonary disease, asthma, and chronic heart failure, and can lead to severe outcomes, such as pneumonia, hospitalization, and death.
As of September 6, 2023, two RSV vaccines were approved by the U.S. FDA.
To learn more, visit www.SidelineRSV.com.
Earvin Johnson played for Michigan State in college in 1979, then for the Los Angeles Lakers, winning numerous championships.

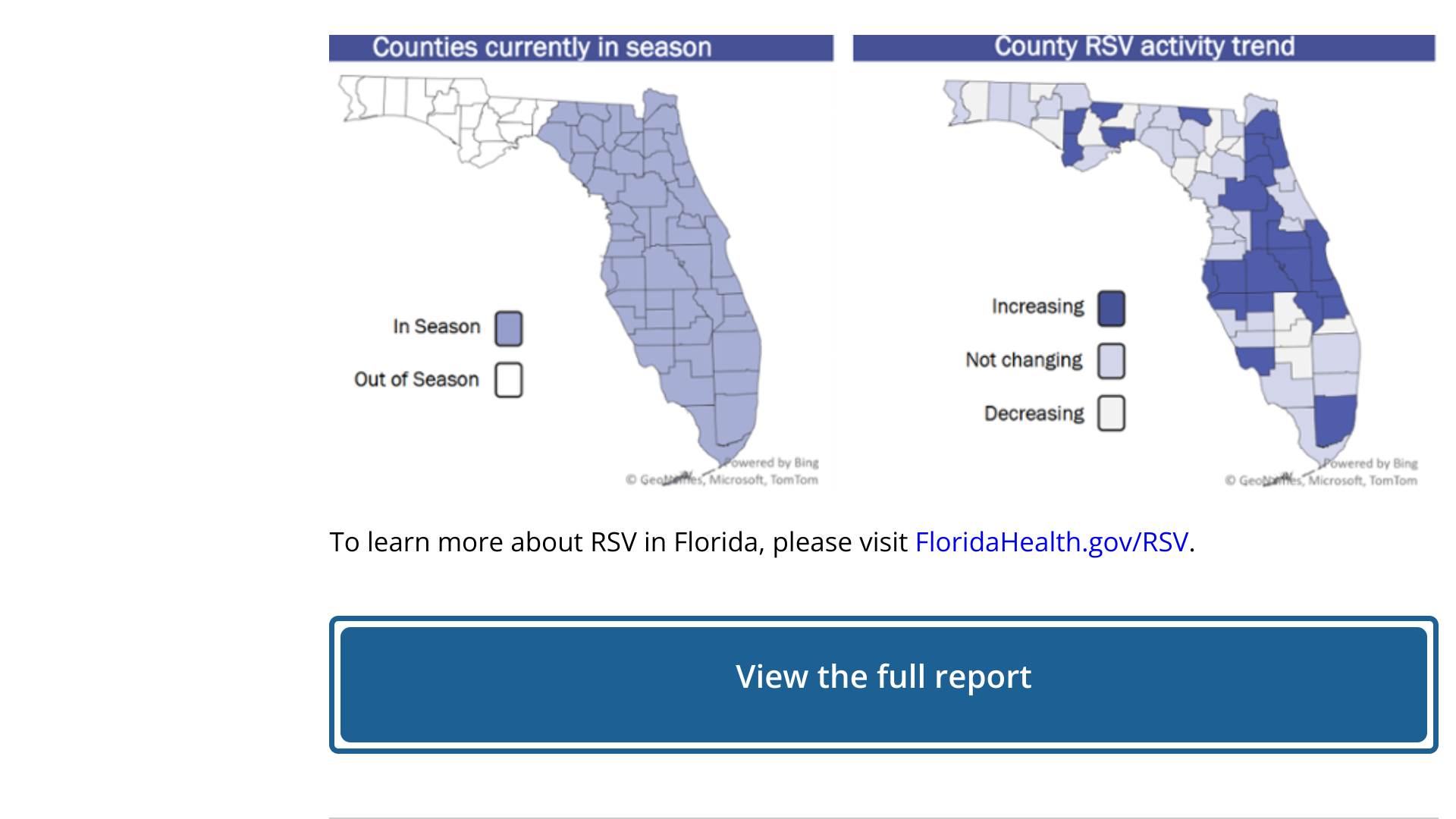
The Centers for Disease Control and Prevention (CDC) today announced a Health Alert Network Health Advisory CDCHAN-00498 to notify clinicians and caregivers about increased respiratory syncytial virus (RSV) activity in the Southeastern United States.
Historically, such regional increases have predicted the beginning of the RSV season in the U.S.
Across both prepandemic and pandemic years, RSV circulation began in Florida, said the CDC on September 5, 2023.
The Florida Department of Health segmented reports into five RSV regions, each with its own RSV season.
As of week #35, September 2, 2023, Florida's RSV activity had increased admission, positivity, and emergency room rates, but no current outbreaks.
In anticipation of the onset of the 2023-2024 RSV season, the CDC encourages clinicians to prepare to implement new RSV prevention options.
A new monoclonal antibody product, Beyfortus™ (Nirsevimab-alip), protects infants and some young children at higher risk for severe RSV disease. Its availability in the U.S. is expected in October 2023.
Also, two new RSV vaccines are available to protect older adults from severe RSV disease: RSVPreF3 (Arexvy, GSK) or RSVpreF (Abrysvo, Pfizer), based on shared clinical decision-making between the healthcare provider and the patient.
Furthermore, the CDC recommends healthcare personnel, childcare providers, and staff at long-term care facilities should stay home and not go to work when they have a fever or symptoms of respiratory infection to reduce the spread of respiratory infections, including RSV.
The Coalition for Epidemic Preparedness Innovations (CEPI) and the International Vaccine Institute (IVI) recently renewed a multi-year collaboration to accelerate the development of vaccines against emerging infectious diseases.
Under the renewed CEPI-IVI Implementing Partnership Agreement announced on September 1, 2023, IVI will provide technical services for CEPI-funded projects, leveraging IVI’s expertise and capabilities at their headquarters in Korea.
And help accelerate vaccine development against pathogens with epidemic or pandemic potential, and in support of CEPI’s 100 Days Mission, a goal to reduce the time taken to develop safe and effective vaccines against pathogens with pandemic potential to 100 days.
To this end, IVI will support CEPI across a variety of projects, including providing strategic, technical, and scientific support for clinical development and manufacturing of vaccines, standards, and assay development, and capacity-building initiatives that strengthen local resources to help establish or improve research preparedness and emergency evidence generation for future outbreaks.
CEPI's 5-year plan lays out a $3.5 billion roadmap to compress vaccine development timelines to 100 days, develop a broadly protective vaccine against COVID-19 and other betacoronaviruses, and create a "library" of vaccine candidates for use against known and unknown pathogens.
CEPI has also invested in new platform technologies for rapid vaccine development against unknown pathogens (Disease X).
Before the recent pandemic, beginning in 2027, CEPI focused on developing vaccines against the Ebola, Lassa, MERS, Nipah, Rift Valley Fever, and Chikungunya viruses.
CEPI's plan is available at https://endpandemics.cepi.net.

The epidemiology of human papillomavirus (HPV) in men is less known than in women, according to research in a study published in The Lancet Global Health.
In early September 2023, a group of researchers published a global systematic review and meta-analysis assessing the prevalence of genital HPV infection in the male population.
They identified 5,685 publications from database searches, of which 65 studies were included from 35 countries.
The pooled prevalence was 31% (95% CI 27–35) for any HPV.
And 21% (18–24) for HR-HPV.
HPV-16 was the most prevalent HPV genotype (5%, 95% CI 4–7), followed by HPV-6 (4%, 3–5).
HPV prevalence was high in young adults, reaching a maximum between 25 and 29 years.
Pooled prevalence estimates were similar for the UN Sustainable Development Goal geographical regions of Europe and Northern America, Sub-Saharan Africa, Latin America and the Caribbean, and Oceania.
However, the estimates for Eastern and South-Eastern Asia were half that of the other regions.
Our findings show that HPV prevalence is high in men over the age of 15 years and support that sexually active men, regardless of age, are an important reservoir of HPV genital infection, wrote these researchers.
These estimates emphasize the importance of incorporating men in comprehensive HPV prevention strategies to reduce HPV-related morbidity and mortality in men and ultimately eliminate cervical cancer and other HPV-related diseases in women.
As of September 5, 2023, there are various HPV vaccines available.
This study was not funded by industry.