Search API
As the Northern Hemisphere heads into its fall respiratory season, most European countries are seeing an end to the recent COVID-19 wave.
Consultation rates of patients presenting to general practitioners with respiratory illness/acute respiratory infection increased in several countries in September 2023 but remained similar to the low levels observed in the same period last year.
The European Centre for Disease Prevention and Control (ECDC) reported for week #36, there was a decrease in COVID-19 cases at the EU/EEA level, which is an inversion of the increasing trend observed in the previous weeks.
Of 19 countries reporting data as of September 14, 2023, twelve reported decreases in COVID-19 cases.
And the impact on severe disease and mortality remained limited in these European countries.
Of countries reporting COVID-19-related hospitalizations and deaths, one reported an increase in hospital admissions, and one reported an increase in fatalities.
Globally, as of September 13, 2023, the World Health Organization (WHO) reported there have been 770,563,467 confirmed cases of COVID-19, including 6,957,216 deaths since the pandemic began in late 2019.
As of September 5, 2023, a total of 13,500,135,157 vaccine doses have been administered.
The WHO has Listed twelve different COVID-19 vaccines during the pandemic.
CSL Seqirus today announced new data will be presented at the European Scientific Working Group on Influenza Conference taking place from 17-20 September 2023 in Spain.
Data from various studies, including real-world evidence, economic and cost-effectiveness modeling, and clinical studies across seasonal and pandemic influenza, will be shared as posters and oral presentations.
"At CSL Seqirus, public health protection is at our core and is what drives us to safeguard at-risk communities against seasonal and pandemic influenza threats," said Raja Rajaram, M.D., Head of Global Medical Strategy, CSL Seqirus, in a press release on September 15, 2023.
"Delivering on our promise means continually gathering and analyzing data to assess the value and impact of our vaccines, and we look forward to sharing results from many of these studies at ESWI that emphasize the national, regional, and global impact."
Influenza is a contagious seasonal respiratory disease that may cause severe illness and life-threatening complications in some people.
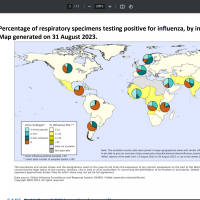
On September 4, 2023, the World Health Organization published Influenza Update N° 453, indicating that influenza detections remained low globally, with activity in many Southern Hemisphere and Oceania countries.
As of September 15, 2023, the new flu and Respiratory Syncytial Virus seasons have begun to impact people in the United States.
An updated overview of influenza vaccines targeting the 2023-2024 flu viruses is posted at Precision Vaccinations.

About 4 billion people eagerly await a vaccine to prevent and/or treat herpes virus infections, a global Sexually Transmitted Disease (STD).
According to the World Health Organization, an estimated 3.7 billion people have herpes simplex virus type 1 (HSV-1) infection, the leading cause of oral herpes.
Additionally, an estimated 491 million people have herpes simplex virus type 2 (HSV-2) infection, the main cause of genital herpes.
Recently, a worldwide leader in mRNA technologies launched a phase 1/2 clinical trial to address this massive market.
Moderna Inc.'s mRNA-1608 is an HSV-2 therapeutic vaccine candidate conducting a study (mRNA-1608-P101) with 300 healthy adults with recurrent HSV-2 Genital Herpes.
This randomized, observer-blind, controlled, dose-ranging study launched in the United States on September 6, 2023, and is forecasted to be completed on June 4, 2025.
Moderna has selected study locations in Alabama, Arizona, and California. The email contact is [email protected]. Or visit Trial Search for additional clinical studies.
With mRNA-1608, Moderna aims to induce a strong antibody response with neutralizing and effector functionality combined with cell-mediated immunity.
On February 18, 2022, Moderna stated that it expects an HSV-2 vaccine could provide cross-protection against HSV-1.
As of September 15, 2023, there is no herpes vaccine candidate approved by the U.S. Food and Drug Administration.

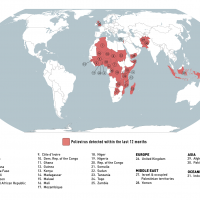
The Ministry of Health, Welfare and Sport of the Netherlands recently notified the World Health Organization (WHO) of a laboratory-confirmed human case of infection with a swine-origin influenza A(H1N1) variant virus.
This is the first human swine flu case reported in the Netherlands this year.
As of September 7, 2023, there were no symptomatic contacts of this case; the person has recovered, and no further detections have been reported.
Therefore, the likelihood of community-level spread among humans and/or international disease spread through humans is considered 'low,' says the WHO.
Zoonotic influenza infections are caused by germs, bacteria, parasites, and fungi, says the U.S. Centers for Disease Control and Prevention (CDC). These germs cause illnesses in people, birds, and animals, ranging from mild to severe infections.
When a swine influenza virus is detected in a person, it is called a "variant influenza virus."
As of September 14, 2023, the CDC says annual 'flu shots' do not prevent zoonotic influenza infections such as swine flu (H3N2) or avian influenza (Bird Flu).

The European Medicines Agency (EMA) today announced its Human Medicines Committee (CHMP) has recommended authorizing an adapted Spikevax vaccine targeting the Omicron XBB.1.5 subvariant.
In its decision to recommend the authorization on September 14, 2023, the CHMP considered all the available data on Spikevax and its other adapted vaccines.
Known as Spikevax XBB.1.5, this vaccine prevents COVID-19 in adults and children from six months of age.
In line with previous recommendations by EMA and the European Centre for Disease Prevention and Control, adults and children from 5 years of age who require vaccination should have a single dose, irrespective of their COVID-19 vaccination history.
Children from 6 months to 4 years of age may have one or two doses depending on whether they have completed a primary vaccination course or have had COVID-19.
The CHMP also considered data from a study in which adults were given Spikevax XBB.1.5 as a booster.
The study showed that the vaccine produced an immune response against the Omicron XBB.1.5 subvariant, as measured by a rise in antibodies against this strain.
The vaccine also produced an immune response against several other strains of the betacornavirus that causes COVID-19, including the currently circulating Omicron XBB.1.16 subvariant.
The EMA has sent the CHMP’s recommendation to the European Commission for an EU-wide legally binding decision.
SpikeVax has recently been approved in Canada and the United States.

Moderna Inc.'s respiratory vaccine franchise is targeting an approximately $30 billion annual market, comprised of a $15 billion COVID-19 market, a $10 billion RSV market, and a $6 billion flu market, with the potential for growth with more effective vaccines.
According to Modern'a press release on September 13, 2023, the Company's respiratory products sales in 2027 are expected to be in the range of $8 billion to $15 billion, depending on certain variables.
"With today's positive Phase 3 flu results, along with previous results in COVID and RSV, we are now three for three on advancing respiratory disease programs to positive Phase 3 data," commented Stéphane Bancel, Chief Executive Officer of Moderna, in a press release.
Modera's press release specifically highlighted its Seasonal Influenza Vaccine business today.
The mRNA-1010 vaccine candidate has demonstrated an acceptable safety and tolerability profile across all clinical trials to date, including three Phase 3 trials (P301, P302, P303), and independent data and safety monitoring boards have raised no safety concerns.
In an interim analysis of the P303 study,mRNA-1010 met all co-primary endpoints across all four A and B strains (A/H1N1, A/H3N2, influenza B/Yamagata, B/Victoria).
Higher HAI geometric mean titers and seroconversion rates were observed for all four strains compared to a licensed comparator (Fluarix).
And local and systemic solicited adverse reactions were similar to those reported in previous mRNA-1010 studies.
Furthermore, improved immunogenicity was observed across age groups and, importantly, was seen in older adults.
mRNA-1010 also elicited higher HAI titers against A/H1N1, A/H3N2, B/Victoria, and comparable titers to B/Yamagata compared to Fluzone HD in a separate Phase 1/2 head-to-head study.
As the previous P302 efficacy study has not accrued its target case numbers by the end of the most recent season, the Company would need to enroll in a second season to accrue enough cases.
In light of P303 meeting all its primary endpoints, the Company has decided not to enroll in a second season in the P302 study.
The Company continues to advance a portfolio of influenza vaccine candidates that include additional HA antigens for broader coverage of circulating influenza A strains (mRNA-1011 and mRNA-1012) and candidates that incorporate both HA and neuraminidase antigens to target multiple proteins involved in the influenza virus lifecycle to reduce the potential of viral antigenic escape (mRNA-1020 and mRNA-1030).
As of early September 2023, over 100 million influenza vaccines have been distributed in the U.S., preparing for the 2023-2024 flu season.